Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in existing synthetic methodologies. Quinazolinones are privileged scaffolds found in numerous drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory activities. The strategic introduction of a trifluoromethyl group further enhances these properties by improving metabolic stability, lipophilicity, and bioavailability. This patent presents a novel iron-catalyzed cyclization strategy that utilizes readily available isatins and trifluoroethylimidoyl chlorides, offering a sustainable and economically viable pathway for producing these high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons like anthranilamide or isatoic anhydride with trifluoroacetic anhydride or ethyl trifluoroacetate. These traditional pathways are often plagued by severe reaction conditions that require stringent temperature control and inert atmospheres, increasing operational complexity and safety risks. Furthermore, the starting materials such as trifluoroacetic anhydride can be corrosive and expensive, driving up the overall cost of goods. Many of these legacy methods also suffer from narrow substrate scope, failing to tolerate sensitive functional groups, which limits their utility in late-stage functionalization or diversity-oriented synthesis. Low yields and difficult purification processes further exacerbate the inefficiency, making large-scale production economically challenging for procurement teams managing tight budgets.
The Novel Approach
In stark contrast, the methodology described in CN111675662B utilizes a cheap and earth-abundant iron catalyst to drive the cyclization of isatin derivatives with trifluoroethylimidoyl chlorides. This approach significantly simplifies the synthetic route by employing commercially available and stable starting materials. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. As illustrated in the general reaction scheme below, this method achieves high conversion rates under air atmosphere, eliminating the need for expensive glovebox techniques. The broad applicability allows for the synthesis of various derivatives, including those with halogen, alkyl, and nitro substituents, providing medicinal chemists with a versatile toolbox for drug discovery.
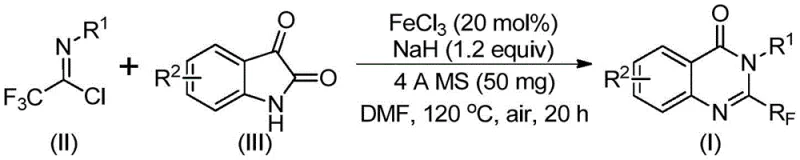
The versatility of this new protocol is further demonstrated by the successful synthesis of specific examples such as compounds (I-1) through (I-5), which include known bioactive scaffolds. The ability to access these structures with high efficiency and purity represents a significant leap forward in process chemistry. By shifting away from precious metal catalysts and harsh fluorinating agents, this method aligns perfectly with green chemistry principles while delivering substantial cost reductions in pharmaceutical intermediates manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this innovation lies in the unique role of ferric chloride (FeCl3) combined with sodium hydride (NaH) and 4A molecular sieves. The reaction mechanism initiates with the deprotonation of the isatin nitrogen by sodium hydride, generating a nucleophilic species that attacks the imidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the iron catalyst facilitates a decarbonylation event, which is the rate-determining step, leading to the formation of the quinazolinone ring system. The presence of 4A molecular sieves is crucial for scavenging moisture and potentially sequestering byproducts, thereby driving the equilibrium towards the desired product. This synergistic effect between the base and the Lewis acidic iron center ensures high turnover numbers and excellent selectivity.
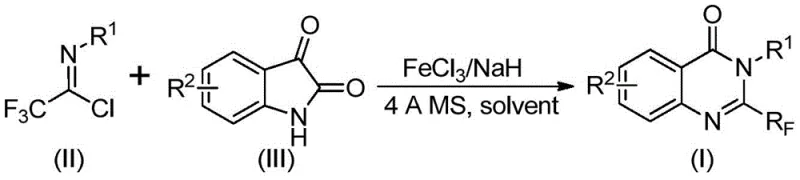
From an impurity control perspective, the use of DMF as the solvent provides excellent solubility for both polar and non-polar intermediates, minimizing the formation of insoluble oligomers that often plague heterogeneous reactions. The two-stage heating profile, starting at 40°C and ramping to 120°C, allows for controlled initiation of the C-N bond formation before triggering the energetically demanding cyclization step. This thermal gradient helps suppress side reactions such as hydrolysis of the imidoyl chloride or decomposition of the sensitive trifluoromethyl group. Consequently, the crude reaction profiles are cleaner, reducing the burden on downstream purification units and ensuring that the final API intermediates meet stringent purity specifications required by regulatory bodies.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management. The patent outlines a robust protocol where ferric chloride is used at 20 mol% loading, balancing catalytic activity with cost efficiency. Sodium hydride is employed in slight excess to ensure complete deprotonation of the isatin substrate. The reaction is typically conducted in DMF, although other aprotic solvents like acetonitrile or dioxane can be evaluated depending on solubility requirements. The detailed standardized synthesis steps, including precise addition rates and quenching procedures, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent such as DMF.
- Stir the mixture at 40°C for approximately 10 hours to initiate the reaction under air atmosphere.
- Heat the reaction mixture to 120°C and continue stirring for an additional 20 hours to complete the cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this iron-catalyzed process offers compelling economic and logistical benefits. The primary driver for cost optimization is the replacement of expensive precious metal catalysts or specialized fluorinating reagents with commodity chemicals like ferric chloride and sodium hydride. These materials are globally sourced with stable pricing, insulating the supply chain from the volatility often seen with rhodium, palladium, or iridium markets. Additionally, the reaction operates under air rather than strict inert gas conditions, significantly reducing the capital expenditure required for specialized reactor infrastructure and lowering utility costs associated with nitrogen or argon purging.
- Cost Reduction in Manufacturing: The elimination of costly catalysts and the use of cheap, bulk-available starting materials directly lower the bill of materials. The simplified workup procedure, which involves basic filtration and standard chromatography, reduces solvent consumption and labor hours compared to multi-step protection-deprotection sequences. This streamlined process flow translates to a lower cost per kilogram, enhancing the margin potential for high-volume production campaigns without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals mitigates the risk of supply disruptions. Isatins and aromatic amines (precursors to imidoyl chlorides) are produced by numerous global suppliers, ensuring a competitive vendor landscape. The robustness of the reaction conditions means that minor variations in raw material quality can be tolerated without catastrophic yield loss, providing a buffer against supply chain fluctuations and ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is designed for scalability, having been demonstrated from gram to multi-gram levels with consistent results. The use of iron, a non-toxic metal, simplifies waste stream management and reduces the environmental footprint associated with heavy metal disposal. This aligns with increasingly strict environmental regulations, avoiding the need for expensive wastewater treatment protocols required for copper or palladium residues, thereby facilitating smoother regulatory approvals for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for this synthesis?
A: The use of ferric chloride (FeCl3) offers significant cost advantages as it is an abundant and inexpensive base metal compared to precious metal catalysts. Furthermore, the protocol demonstrates excellent functional group tolerance and operates under relatively mild conditions, reducing energy consumption and safety risks associated with harsh reagents.
Q: Can this method accommodate diverse substituents on the isatin and imidoyl chloride substrates?
A: Yes, the patented method exhibits broad substrate scope. It successfully tolerates various substituents including alkyl, halogen, methoxy, and nitro groups at ortho-, meta-, and para-positions on the aryl rings, allowing for the synthesis of a wide library of derivatives for SAR studies.
Q: Is the purification process complex for these quinazolinone derivatives?
A: The post-treatment is straightforward and relies on standard industry practices. The reaction mixture is simply filtered to remove solids, mixed with silica gel, and purified using conventional column chromatography, making it highly suitable for scale-up without requiring specialized separation equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex heterocyclic scaffolds. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest industry standards. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate use in drug substance manufacturing.
We invite you to collaborate with us to leverage this advanced iron-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your development timelines and reduce your overall manufacturing costs.
