Advanced Palladium-Catalyzed Carbonylation for Scalable 3-Arylquinolin-2(1H)-one Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 3-Arylquinolin-2(1H)-one Production
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical backbones for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a highly efficient preparation method for 3-arylquinolin-2(1H) ketone derivatives. These derivatives are not merely academic curiosities; they are pivotal structures found in a vast array of therapeutic agents, including antibiotics, antiplatelet drugs, and antitumor medications. The innovation lies in the strategic use of benzisoxazole, which functions dually as a nitrogen source and a formyl source, thereby streamlining the synthetic pathway. This approach addresses long-standing challenges in heterocycle synthesis by offering a operationally simple protocol with excellent functional group tolerance. For R&D teams focused on developing reliable pharmaceutical intermediate supplier networks, understanding this technology is crucial for securing high-purity building blocks.
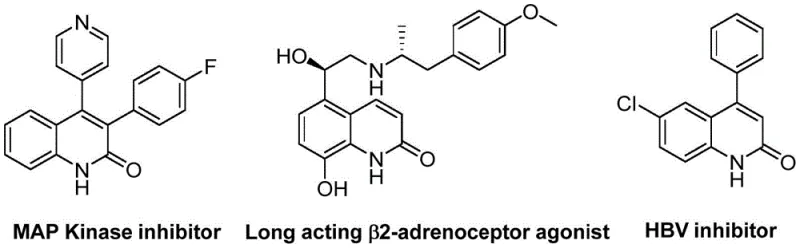
Quinolin-2(1H)one derivatives represent a privileged scaffold in medicinal chemistry, underpinning the efficacy of numerous drugs targeting endothelin receptors and angiotensin II receptors. Historically, accessing these cores required multi-step sequences involving harsh reagents or unstable intermediates. The methodology described in the patent leverages transition metal catalysis to forge these bonds with precision. By utilizing a palladium catalyst system combined with molybdenum hexacarbonyl as a solid carbon monoxide surrogate, the process avoids the safety hazards associated with handling gaseous CO. This technological leap facilitates the production of high-purity OLED material precursors and pharmaceutical intermediates alike, ensuring that supply chains remain uninterrupted by hazardous material restrictions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing the quinolinone core, such as the Vilsmeier-Haack, Knorr, and Friedlander reactions, have served the industry for decades but suffer from inherent limitations that impact cost reduction in API manufacturing. These classical methods often necessitate the use of corrosive reagents like phosphorus oxychloride or strong acids, which generate substantial acidic waste streams requiring expensive neutralization and disposal protocols. Furthermore, these routes frequently exhibit poor atom economy and limited substrate scope, particularly when sensitive functional groups are present on the aromatic rings. The reliance on pre-functionalized anilines or beta-keto esters can also drive up raw material costs, as these precursors may require their own complex synthesis. Consequently, manufacturers face challenges in scaling these processes while maintaining environmental compliance and economic viability.
The Novel Approach
In stark contrast, the novel palladium-catalyzed aminocarbonylation route offers a transformative solution by merging simplicity with high efficiency. As illustrated in the reaction scheme below, the direct coupling of benzisoxazole with benzyl chloride compounds proceeds under relatively mild thermal conditions. This method eliminates the need for pre-installed carbonyl groups, as the carbon monoxide is introduced in situ from molybdenum hexacarbonyl. The versatility of this approach is evidenced by its ability to accommodate a wide range of substituents, including halogens, alkoxy groups, and cyano groups, without significant loss in yield. For procurement managers, this translates to a more flexible sourcing strategy where diverse analogues can be accessed from a common platform, reducing the need for multiple distinct synthetic lines.
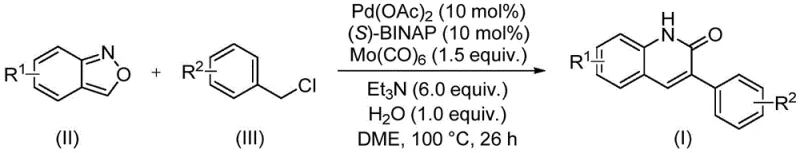
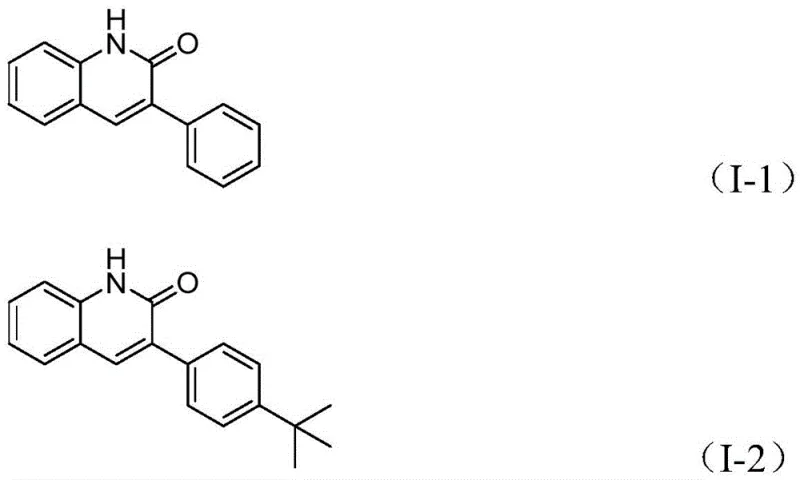
The practical utility of this new method is further highlighted by the specific examples provided in the patent data, which demonstrate exceptional yields across different electronic environments. For instance, substrates bearing electron-donating groups like tert-butyl or methoxy, as well as electron-withdrawing groups like cyano or chloro, all afford the desired products in yields exceeding 90% in many cases. This broad scope is critical for medicinal chemists who need to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies. The ability to synthesize compounds like I-1 through I-5 with such consistency ensures that the transition from discovery to development is seamless, supporting the commercial scale-up of complex polymer additives or drug candidates with minimal process re-engineering.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The success of this transformation relies on a sophisticated catalytic cycle initiated by the oxidative addition of the benzyl chloride to the active palladium(0) species generated in situ. The chiral ligand (S)-BINAP plays a pivotal role not only in stabilizing the palladium center but potentially in influencing the stereochemical outcome if chiral centers were involved, although the primary focus here is on the efficient formation of the heterocyclic ring. Following oxidative addition, the insertion of carbon monoxide—released from the decomposition of Mo(CO)6—forms an acyl-palladium intermediate. This step is crucial as it introduces the carbonyl functionality required for the lactam ring closure. The subsequent nucleophilic attack by the nitrogen species derived from the ring-opening of benzisoxazole leads to the formation of the amide bond, completing the quinolinone skeleton.
Impurity control is a paramount concern for any GMP-compliant manufacturing process, and this mechanism offers distinct advantages in that regard. The use of benzisoxazole as a masked nitrogen/formyl source minimizes the formation of side products typically associated with free amine acylation, such as over-acylation or polymerization. The reaction conditions, specifically the use of triethylamine as a base and DME as a solvent, are optimized to suppress competing pathways like homocoupling of the benzyl chloride. Furthermore, the high selectivity observed in the experimental data suggests that the catalytic system effectively discriminates between the desired carbonylation pathway and potential beta-hydride elimination side reactions. This intrinsic selectivity reduces the burden on downstream purification, allowing for simpler workup procedures that align with green chemistry principles.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent ratios and reaction parameters to maximize yield and purity. The protocol dictates a specific molar ratio of palladium catalyst to ligand to ensure optimal turnover numbers, while the excess of benzyl chloride drives the reaction to completion. The reaction is typically conducted in a sealed vessel to retain volatile components and maintain the necessary pressure for carbonylation. Detailed operational guidelines regarding temperature ramping and quenching procedures are essential for reproducibility. For a comprehensive guide on executing this synthesis, please refer to the standardized steps outlined below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)-one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound benefits for supply chain resilience and cost management. The primary advantage stems from the utilization of commodity chemicals as starting materials; benzisoxazoles and benzyl chlorides are widely available from global chemical suppliers, mitigating the risk of raw material shortages. This accessibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond swiftly to market demands. Moreover, the simplified reaction setup, which does not require specialized high-pressure equipment for gaseous CO, lowers the barrier to entry for contract manufacturing organizations (CMOs) looking to offer this capability.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous reagents traditionally used for formylation. By using molybdenum hexacarbonyl as a solid CO source, the need for specialized gas handling infrastructure is removed, resulting in substantial capital expenditure savings. Additionally, the high yields reported (often above 90%) mean that less raw material is wasted per kilogram of product, directly improving the cost of goods sold (COGS). The simplified purification process further contributes to cost efficiency by reducing solvent consumption and labor hours associated with complex chromatographic separations.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic intermediates is a major vulnerability in the pharmaceutical supply chain. This method relies on robust, commercially available building blocks that can be sourced from multiple vendors, thereby diversifying supply risk. The stability of the reagents allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to maintain strategic stockpiles. This reliability is critical for ensuring continuous production schedules and meeting strict delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this homogeneous catalytic system is well-suited for scale-up due to its straightforward kinetics. The reaction operates at moderate temperatures (100°C), which is easily achievable in standard stainless steel reactors without requiring extreme cooling or heating utilities. From an environmental standpoint, the atom economy is superior to classical methods, generating less waste. The ability to recycle solvents like DME and the reduced generation of acidic byproducts align with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals for new manufacturing sites.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their production portfolios. The following questions address common inquiries regarding catalyst loading, substrate compatibility, and purification strategies. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, eliminating the need for separate toxic formylating agents and simplifying the reaction stoichiometry significantly.
Q: What is the typical yield range for this palladium-catalyzed method?
A: The patented process demonstrates high reaction efficiency, with isolated yields ranging from 68% to 97% across various substrates, including those with electron-withdrawing and electron-donating groups.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the method utilizes commercially available starting materials and standard purification techniques like column chromatography, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
As the demand for complex heterocyclic intermediates continues to grow, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale optimization to full-scale manufacturing. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-arylquinolin-2(1H)-one meets the highest industry standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering consistent results.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By working together, we can optimize your supply chain and accelerate the development of life-saving therapies based on this versatile quinolinone scaffold.
