Scalable Synthesis of Novel Indole Derivatives for High-Purity PAK Inhibitor Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic routes for next-generation kinase inhibitors, particularly those targeting the p21-activated kinases (PAKs) which play a critical role in tumor progression and cell migration. Patent CN113831325A introduces a significant advancement in this domain by disclosing a series of novel indole derivatives defined by general formula (I), which exhibit potent inhibitory activity against PAK4. This intellectual property represents a strategic opportunity for pharmaceutical manufacturers looking to diversify their oncology pipeline with compounds that offer improved selectivity over class I PAKs. The technical depth of this patent lies not only in the biological efficacy of the final molecules but also in the versatility of the synthetic methodology employed to construct the complex indole-pyrimidine scaffold. For R&D teams evaluating new entry points into the kinase inhibitor market, understanding the chemical space covered by this patent is essential for freedom-to-operate analyses and competitive intelligence. The disclosed compounds serve as high-value pharmaceutical intermediates that can be further elaborated into active pharmaceutical ingredients (APIs) for clinical development. As we delve into the technical specifics, it becomes clear that the innovation here supports the production of high-purity PAK inhibitor intermediates with a level of structural diversity that was previously difficult to achieve using traditional heterocyclic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex indole-based kinase inhibitors has been plagued by significant challenges related to regioselectivity and the harsh conditions required to form carbon-carbon bonds at the indole core. Conventional methods often rely on multiple protection and deprotection steps to manage the reactivity of the indole nitrogen, which drastically increases the step count and reduces the overall atom economy of the process. Furthermore, traditional cross-coupling reactions on indole substrates frequently suffer from low yields due to the formation of homocoupling byproducts or decomposition of the sensitive heterocyclic ring under strong basic conditions. These inefficiencies translate directly into higher manufacturing costs and longer cycle times, creating bottlenecks for procurement managers who are tasked with securing cost reduction in API manufacturing. The reliance on expensive transition metal catalysts without efficient recovery systems also poses environmental compliance issues, complicating the waste management strategy for large-scale production facilities. Additionally, the purification of intermediates generated through older methodologies often requires extensive chromatographic separation, which is not feasible for commercial scale-up of complex polymer additives or pharmaceutical intermediates. These legacy limitations necessitate a shift towards more convergent and catalytic approaches that can deliver the required molecular complexity with greater operational simplicity.
The Novel Approach
In contrast to these legacy constraints, the methodology outlined in CN113831325A leverages a highly efficient Sonogashira coupling strategy to construct the critical alkyne linkage between the indole core and the cyclohexyl-hydroxy moiety. This novel approach utilizes a palladium-copper catalytic system that operates under relatively mild thermal conditions, thereby preserving the integrity of sensitive functional groups such as the aminopyrimidine ring. 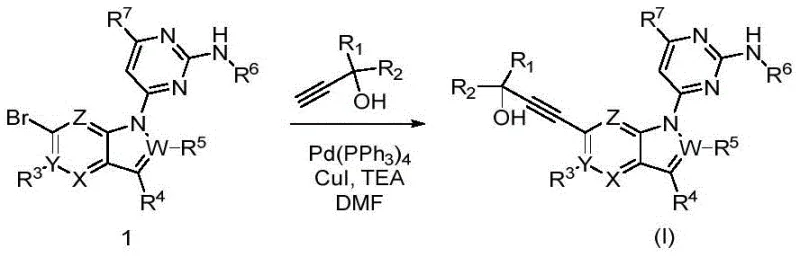 By streamlining the bond formation process, this route eliminates the need for excessive protecting group manipulations, effectively reducing the total number of unit operations required to reach the final target molecule. The use of commercially available alkynols and bromo-indole precursors allows for a modular synthesis where different substituents can be introduced late in the sequence, providing flexibility for medicinal chemistry optimization. This modularity is a key advantage for supply chain heads concerned with reducing lead time for high-purity kinase inhibitors, as it allows for the stocking of common intermediates that can be rapidly converted into various final analogs. The robustness of this catalytic cycle ensures consistent quality across batches, which is paramount for maintaining stringent purity specifications required in GMP environments. Ultimately, this synthetic design transforms a potentially cumbersome multi-step sequence into a manageable and scalable process suitable for industrial adoption.
By streamlining the bond formation process, this route eliminates the need for excessive protecting group manipulations, effectively reducing the total number of unit operations required to reach the final target molecule. The use of commercially available alkynols and bromo-indole precursors allows for a modular synthesis where different substituents can be introduced late in the sequence, providing flexibility for medicinal chemistry optimization. This modularity is a key advantage for supply chain heads concerned with reducing lead time for high-purity kinase inhibitors, as it allows for the stocking of common intermediates that can be rapidly converted into various final analogs. The robustness of this catalytic cycle ensures consistent quality across batches, which is paramount for maintaining stringent purity specifications required in GMP environments. Ultimately, this synthetic design transforms a potentially cumbersome multi-step sequence into a manageable and scalable process suitable for industrial adoption.
Mechanistic Insights into Pd-Catalyzed Sonogashira Coupling
The core chemical transformation enabling this technology is the palladium-catalyzed cross-coupling reaction, specifically the Sonogashira coupling, which joins a terminal alkyne with an aryl or vinyl halide. In the context of this patent, the reaction involves the coupling of a 6-bromo-1H-indole derivative with a substituted alkynol in the presence of tetrakis(triphenylphosphine)palladium(0) and copper(I) iodide. 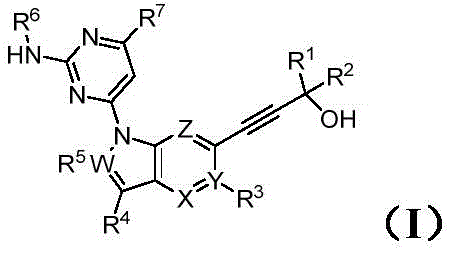 The mechanism proceeds through a catalytic cycle where the palladium species undergoes oxidative addition into the carbon-bromine bond of the indole substrate, forming an organopalladium intermediate. Simultaneously, the copper co-catalyst activates the terminal alkyne by forming a copper-acetylide species, which then transmetallates with the palladium complex. This intricate dance of ligands and metals ensures that the carbon-carbon bond is formed with high fidelity, minimizing the formation of undesired homocoupled alkyne byproducts that often plague uncatalyzed or poorly optimized reactions. The choice of base, typically triethylamine in this protocol, plays a crucial role in regenerating the active catalyst and neutralizing the acid byproduct generated during the coupling event. Understanding this mechanistic pathway is vital for R&D directors focusing on purity and impurity profiles, as deviations in catalyst loading or base strength can lead to trace metal contamination or incomplete conversion. The patent demonstrates that by fine-tuning these parameters, it is possible to achieve high conversion rates while maintaining the structural integrity of the complex heterocyclic system.
The mechanism proceeds through a catalytic cycle where the palladium species undergoes oxidative addition into the carbon-bromine bond of the indole substrate, forming an organopalladium intermediate. Simultaneously, the copper co-catalyst activates the terminal alkyne by forming a copper-acetylide species, which then transmetallates with the palladium complex. This intricate dance of ligands and metals ensures that the carbon-carbon bond is formed with high fidelity, minimizing the formation of undesired homocoupled alkyne byproducts that often plague uncatalyzed or poorly optimized reactions. The choice of base, typically triethylamine in this protocol, plays a crucial role in regenerating the active catalyst and neutralizing the acid byproduct generated during the coupling event. Understanding this mechanistic pathway is vital for R&D directors focusing on purity and impurity profiles, as deviations in catalyst loading or base strength can lead to trace metal contamination or incomplete conversion. The patent demonstrates that by fine-tuning these parameters, it is possible to achieve high conversion rates while maintaining the structural integrity of the complex heterocyclic system.
Beyond the primary coupling event, the patent details a sophisticated strategy for managing impurity profiles through careful control of reaction stoichiometry and workup procedures. The presence of the aminopyrimidine moiety introduces potential sites for side reactions, such as nucleophilic attack on the pyrimidine ring or coordination with the metal catalysts, which could deactivate the system. To mitigate this, the process employs specific solvent systems like dimethylformamide (DMF) that solubilize the polar intermediates while stabilizing the catalytic species. The purification strategy described involves aqueous quenching followed by extraction with ethyl acetate, which effectively removes inorganic salts and polar byproducts from the organic phase containing the desired indole derivative. Further purification via column chromatography ensures that the final product meets the rigorous standards required for biological evaluation and subsequent clinical use. This attention to detail in impurity control is a hallmark of a mature process designed for commercial viability, ensuring that the resulting high-purity OLED material or pharmaceutical intermediate is free from genotoxic impurities or residual heavy metals. For quality assurance teams, this level of process definition provides the necessary confidence to validate the method for GMP production.
How to Synthesize Novel Indole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the reaction conditions and safety protocols outlined in the intellectual property. The process begins with the preparation of the key bromo-indole intermediate, which serves as the electrophilic partner in the subsequent coupling reaction. Operators must ensure that all reagents, particularly the palladium catalyst and copper iodide, are handled under inert atmosphere conditions to prevent oxidation which could diminish catalytic activity. The detailed standardized synthesis steps involve precise temperature control during the heating phase to drive the reaction to completion without degrading the thermally sensitive components. Following the reaction, the workup procedure is critical for isolating the product in high yield, involving careful pH adjustments and solvent exchanges to maximize recovery. It is important to note that while the patent provides exemplary embodiments, scaling this chemistry requires engineering assessments to manage heat transfer and mixing efficiency in larger vessels. The following guide summarizes the critical operational phases derived from the patent examples to assist technical teams in replicating this high-value chemistry.
- Prepare the bromo-indole intermediate by reacting 6-bromoindole with cesium carbonate and 2-amino-4-chloropyrimidine in DMF at elevated temperatures.
- Execute the Sonogashira coupling reaction using tetrakis(triphenylphosphine)palladium(0) and copper(I) iodide catalysts with an alkynol substrate.
- Purify the final indole derivative product through aqueous workup and column chromatography to achieve high purity specifications suitable for biological testing.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement and supply chain teams focused on optimizing the cost structure of oncology drug development. The primary advantage lies in the use of readily available starting materials, such as 6-bromoindole and substituted chloropyrimidines, which are produced at scale by multiple global suppliers, thereby mitigating single-source supply risks. This abundance of raw materials ensures enhanced supply chain reliability, allowing manufacturers to secure long-term contracts and stabilize pricing against market volatility. Furthermore, the convergence of the synthesis, where complex fragments are joined late in the sequence, reduces the inventory burden of carrying multiple advanced intermediates, freeing up working capital for other strategic initiatives. The elimination of exotic reagents or cryogenic conditions also simplifies the facility requirements, meaning that production can be outsourced to a broader range of contract manufacturing organizations without needing specialized infrastructure. These factors collectively contribute to significant cost reduction in manufacturing, making the final API more competitive in price-sensitive markets. By streamlining the process flow, companies can also accelerate their time-to-market, capturing value sooner in the product lifecycle.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the need for expensive protecting group strategies and reduces the total number of isolation steps, which directly lowers labor and material consumption costs. By utilizing a catalytic coupling reaction with high atom economy, the process minimizes waste generation, leading to substantial cost savings in waste disposal and solvent recovery operations. The high yields reported in the patent examples suggest that less raw material is required per kilogram of final product, further driving down the cost of goods sold. Additionally, the robustness of the reaction conditions reduces the likelihood of batch failures, ensuring consistent production output and avoiding the financial penalties associated with reprocessing or scrapped batches. This economic efficiency makes the technology particularly attractive for generic drug manufacturers seeking to enter the kinase inhibitor market with a competitive cost basis.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the starting materials ensures that the supply chain is resilient to disruptions that might affect specialized or custom-synthesized reagents. Since the key building blocks are widely traded, procurement managers can easily qualify alternative vendors to ensure continuity of supply in case of logistical issues or geopolitical instability. The modular nature of the synthesis also allows for flexible production scheduling, where different analogs can be produced on the same equipment line with minimal changeover time. This agility is crucial for responding to fluctuating demand signals from clinical trials or commercial launches. Moreover, the stability of the intermediates allows for safer storage and transportation, reducing the risk of degradation during transit and ensuring that materials arrive at the manufacturing site in optimal condition for processing.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as filtration, extraction, and distillation that are easily transferred from laboratory to commercial scale. The avoidance of hazardous reagents like strong lithiating agents or pyrophoric catalysts enhances workplace safety and simplifies regulatory compliance regarding environmental health and safety standards. The solvent system, primarily based on DMF and ethyl acetate, is well-understood in terms of recovery and recycling, supporting sustainability goals and reducing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only satisfies corporate social responsibility mandates but also future-proofs the supply chain against tightening environmental regulations. Consequently, this technology represents a sustainable path forward for the commercial production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indole derivative synthesis technology. These answers are derived directly from the technical disclosures within the patent documentation to provide accurate guidance for stakeholders. Understanding these nuances is critical for making informed decisions about licensing, partnership, or internal development of this chemistry. The responses cover aspects ranging from catalytic mechanisms to supply chain considerations, ensuring a comprehensive overview of the technology's capabilities. Stakeholders are encouraged to review these points carefully to assess the fit of this technology within their existing portfolios.
Q: What is the primary catalytic mechanism used in this indole derivative synthesis?
A: The synthesis primarily relies on a Palladium-catalyzed Sonogashira cross-coupling reaction, utilizing Pd(PPh3)4 and CuI to couple bromo-indole intermediates with alkynols efficiently.
Q: How does this novel route address impurity profiles compared to conventional methods?
A: By employing specific nucleophilic substitution conditions with cesium carbonate prior to coupling, the route minimizes side reactions at the indole nitrogen, resulting in a cleaner impurity profile and simplified downstream purification.
Q: Are the starting materials for this PAK inhibitor intermediate commercially available?
A: Yes, key starting materials such as 6-bromoindole and various substituted pyrimidines are readily available commodity chemicals, which significantly reduces supply chain risk and raw material procurement lead times.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the novel indole derivatives disclosed in CN113831325A for the development of next-generation PAK inhibitors. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these patented laboratory methods into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical supply to full market launch. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation for comprehensive impurity profiling. Our dedication to quality assurance guarantees that every batch of indole derivatives we produce adheres to the highest international standards, providing you with the reliability needed for successful regulatory filings. Partnering with us means gaining access to a wealth of process chemistry knowledge that can optimize your supply chain and reduce your overall development risks.
We invite you to engage with our technical procurement team to discuss how we can support your specific requirements for high-purity PAK inhibitor intermediates. Whether you need a Customized Cost-Saving Analysis for your current route or require assistance with technology transfer, our experts are ready to provide tailored solutions. We encourage you to request specific COA data and route feasibility assessments to verify our capabilities against your project milestones. By collaborating with NINGBO INNO PHARMCHEM, you secure a supply partner dedicated to your success in the competitive oncology therapeutics market. Let us help you navigate the complexities of fine chemical manufacturing so you can focus on delivering life-saving medicines to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
