Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The rapid evolution of medicinal chemistry demands efficient access to fluorinated heterocycles, particularly those containing the 1,2,3-triazole motif, which serves as a robust bioisostere in numerous drug candidates. A groundbreaking development in this field is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a simpler, base-promoted protocol that eliminates the need for hazardous azides and expensive copper catalysts. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this methodology offers a compelling value proposition by streamlining synthesis while enhancing safety profiles. The ability to construct these complex scaffolds under mild conditions using readily available starting materials addresses critical bottlenecks in the supply chain for high-purity OLED material and API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. As illustrated in the comparative reaction schemes, these legacy pathways present significant operational challenges. The primary concern is the inherent instability and toxicity of organic azides, which pose severe explosion risks during handling and storage, thereby complicating safety protocols in manufacturing facilities. Furthermore, the reliance on copper catalysts introduces a downstream burden; residual heavy metals must be rigorously removed to meet stringent regulatory standards for pharmaceutical ingredients, often requiring additional purification steps like scavenging resins or extensive chromatography. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, making conventional routes less attractive for large-scale commercial production where efficiency and safety are paramount.
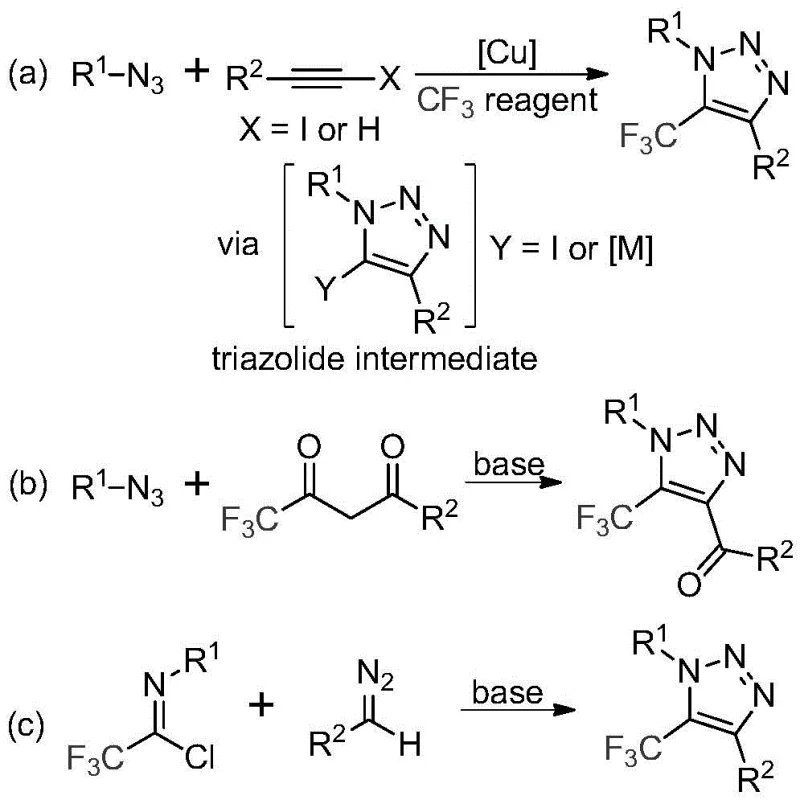
The Novel Approach
In stark contrast to the hazardous traditional routes, the invention described in CN113121462B introduces a metal-free, base-promoted synthesis that utilizes cheap and readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials. This innovative strategy bypasses the use of explosive azides entirely, substituting them with safer diazo surrogates that react efficiently under mild thermal conditions. The process operates effectively in common aprotic solvents like acetonitrile at temperatures ranging from 50°C to 70°C, typically optimizing around 60°C. By eliminating transition metals, the method inherently produces a cleaner crude reaction profile, drastically simplifying the workup procedure. This approach not only mitigates safety risks associated with azide handling but also removes the economic and logistical burden of metal removal, positioning it as a superior choice for cost reduction in electronic chemical manufacturing and pharmaceutical intermediate production.
Mechanistic Insights into Base-Promoted Cyclization
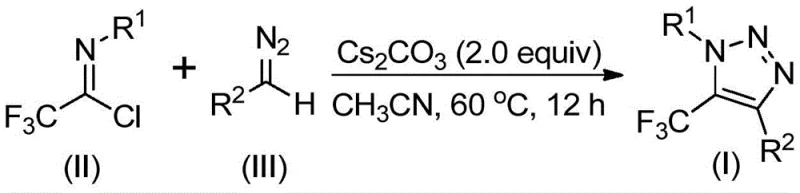
The mechanistic pathway of this transformation is both elegant and efficient, driven by the basicity of cesium carbonate which acts as a promoter rather than a stoichiometric reagent in a catalytic-like fashion relative to the hazardous reagents avoided. The reaction likely initiates with a base-promoted intermolecular nucleophilic addition-elimination process where the diazo compound attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This key step facilitates the formation of a crucial carbon-carbon bond, setting the stage for ring closure. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific cyclization mode is thermodynamically favorable in this system and leads directly to the formation of the five-membered 1,2,3-triazole ring bearing the trifluoromethyl group at the 5-position. The use of 4Å molecular sieves in the reaction mixture further drives the equilibrium forward by sequestering moisture, ensuring high conversion rates and minimizing hydrolysis side products.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation methods. Because the trifluoromethyl group is pre-installed on the imidoyl chloride starting material, there is no risk of random radical abstraction or non-selective fluorination that often plagues late-stage functionalization strategies. The regioselectivity is inherently controlled by the structure of the imidoyl chloride and the diazo compound, ensuring that the trifluoromethyl group ends up exclusively at the 5-position of the triazole ring. This high degree of predictability simplifies the purification process, as the formation of regioisomers is virtually non-existent. Consequently, the final product exhibits a clean impurity profile, which is critical for meeting the rigorous quality specifications required for high-purity pharmaceutical intermediates and agrochemical active ingredients.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot plant operations. The standard protocol involves charging a reaction vessel with cesium carbonate, 4Å molecular sieves, trifluoroethylimidoyl chloride, and the specific diazo compound in an organic solvent such as acetonitrile. The mixture is then heated to 60°C and stirred for approximately 12 hours, although the window allows for 8 to 16 hours depending on the specific substrate reactivity. Upon completion, the reaction mixture is filtered to remove solids, concentrated, and the residue is purified via standard silica gel column chromatography. This straightforward workflow minimizes unit operations and equipment requirements, facilitating rapid technology transfer.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, diazo compound, and 4Å molecular sieves in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 50-70°C (optimally 60°C) and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere chemical yield. The elimination of copper catalysts and explosive azides fundamentally alters the cost structure and risk profile of the manufacturing process. By removing the need for specialized hazard containment equipment required for azides and the expensive scavenging materials needed for metal removal, the overall production cost is significantly reduced. Furthermore, the starting materials—trifluoroethylimidoyl chlorides and diazo compounds—are commercially available or easily synthesized from bulk commodities, ensuring a stable and resilient supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the exclusion of transition metal catalysts. In traditional processes, the cost of the catalyst itself, combined with the downstream processing required to reduce metal residues to ppm levels, constitutes a significant portion of the manufacturing budget. This metal-free protocol obviates these costs entirely. Additionally, the use of cesium carbonate as a benign base instead of exotic ligands or organometallic bases further lowers raw material expenses. The simplified workup, which avoids complex extraction or chelation steps, reduces solvent consumption and labor hours, leading to substantial cost savings in the overall production of complex polymer additives or pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for hazardous or highly specialized reagents. This method utilizes robust, shelf-stable starting materials that are widely sourced in the global chemical market. The avoidance of organic azides removes a major logistical hurdle, as shipping and storing explosive materials require special permits and infrastructure, often causing delays. By shifting to a safer chemical platform, manufacturers can streamline logistics, reduce insurance premiums, and ensure consistent delivery schedules, thereby reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production stoppages due to regulatory compliance issues.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, the environmental footprint of a synthesis route is increasingly critical. This method aligns with green chemistry principles by avoiding toxic heavy metals and hazardous explosives. The waste stream is significantly cleaner, consisting primarily of inorganic salts and organic solvents that are easier to treat and dispose of compared to heavy metal-contaminated waste. This simplifies environmental compliance and reduces waste treatment costs. Moreover, the reaction conditions (60°C, atmospheric pressure) are mild and easily scalable from gram to kilogram quantities without requiring high-pressure reactors or cryogenic cooling, facilitating the commercial scale-up of complex pharmaceutical intermediates with minimal capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners and licensees.
Q: What are the safety advantages of this new triazole synthesis method compared to traditional copper-catalyzed routes?
A: Traditional methods often rely on toxic and explosive organic azides and require transition metal catalysts like copper. This novel protocol utilizes stable diazo compounds and imidoyl chlorides under metal-free conditions, significantly reducing safety hazards and eliminating the need for costly heavy metal removal steps.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states that the method is easily scalable to the gram level and beyond. The use of inexpensive reagents like cesium carbonate and common solvents like acetonitrile, combined with mild reaction temperatures (60°C), makes it highly suitable for commercial scale-up.
Q: What is the substrate scope for R1 and R2 groups in this triazole formation?
A: The method demonstrates broad functional group tolerance. R1 can be alkyl, substituted or unsubstituted aryl (e.g., phenyl, 4-Me-Ph, 4-Cl-Ph). R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for diverse structural modifications essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
The technological advancements detailed in CN113121462B underscore the immense potential of metal-free heterocycle synthesis in modern drug discovery and material science. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative pathways and have integrated similar advanced synthetic capabilities into our CDMO portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole meets the highest international standards for pharmaceutical and agrochemical applications.
We invite global partners to leverage our expertise for their next project. Whether you require custom synthesis of novel triazole derivatives or process optimization for existing routes, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and product quality.
