Scalable Synthesis of Novel Six-Membered Ring Carbapenem Intermediates for Advanced Antibiotic Production
Scalable Synthesis of Novel Six-Membered Ring Carbapenem Intermediates for Advanced Antibiotic Production
The pharmaceutical landscape is constantly evolving to combat resistant bacterial strains, driving the need for innovative antibiotic scaffolds. Patent CN101367815B introduces a significant advancement in this field by disclosing a series of six-membered ring methanamide substituted sulfhydryl pyrrolidine carbapenem compounds. These novel structures, represented by general formula (I), exhibit potent antibacterial activity against a wide range of pathogens, including resistant organisms, while maintaining superior chemical stability compared to earlier generations of carbapenems. For R&D directors and procurement specialists seeking reliable carbapenem intermediate suppliers, understanding the synthetic accessibility and structural advantages of these compounds is crucial for securing the supply chain of next-generation anti-infectives.
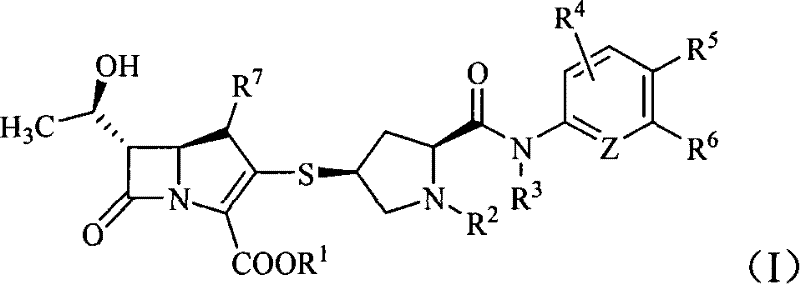
The core innovation lies in the specific substitution pattern on the pyrrolidine ring, where a six-membered ring carboxamide group is introduced. This modification not only enhances the biological profile but also offers distinct processing advantages during manufacturing. The patent details various substituents (R1 through R7 and Z) that allow for fine-tuning the pharmacokinetic properties, making these intermediates highly versatile for developing diverse drug candidates. As the industry moves towards more stable and effective beta-lactam antibiotics, these intermediates represent a critical building block for future commercial products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of carbapenem antibiotics often faces significant hurdles related to chemical instability and complex purification processes. Many prior art methods rely on harsh reaction conditions that can degrade the sensitive beta-lactam ring, leading to lower yields and increased impurity profiles. Furthermore, the introduction of side chains onto the carbapenem nucleus frequently requires multiple protection and deprotection steps, which elongates the production timeline and increases waste generation. For supply chain heads, these inefficiencies translate into higher costs and potential bottlenecks in meeting the demand for essential antibiotics. The sensitivity of the thioether linkage in many conventional derivatives also poses challenges during scale-up, requiring stringent control over reaction parameters to prevent oxidation or decomposition.
The Novel Approach
The methodology outlined in CN101367815B addresses these challenges through a streamlined synthetic strategy that prioritizes stability and modularity. By utilizing specific protecting groups and optimized coupling conditions, the new approach minimizes the exposure of the reactive beta-lactam core to detrimental environments. The use of readily available aniline derivatives for the side chain construction allows for a modular assembly line, facilitating cost reduction in pharmaceutical manufacturing by simplifying the sourcing of raw materials. This novel route ensures that the final intermediates possess high purity and consistent stereochemistry, which are critical quality attributes for regulatory approval. The ability to produce these compounds with robust yields makes them an attractive option for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Amide Coupling and Deprotection Strategies
The synthesis relies heavily on efficient amide bond formation between the pyrrolidine carboxylic acid derivative and various aniline substrates. In the first preferred method, 1,1'-carbonyldiimidazole (CDI) is employed as the activating agent in anhydrous tetrahydrofuran. This activation step generates a reactive acyl imidazole intermediate, which subsequently reacts with the amine nucleophile to form the desired amide linkage with high fidelity. The choice of CDI is strategic, as it avoids the formation of difficult-to-remove urea byproducts often associated with other coupling reagents, thereby simplifying downstream purification. Following the coupling, the removal of protecting groups is achieved using aluminum chloride in nitromethane, a condition specifically selected to cleave the ester protecting group on the carbapenem nucleus without compromising the integrity of the newly formed amide bond or the sensitive beta-lactam ring.
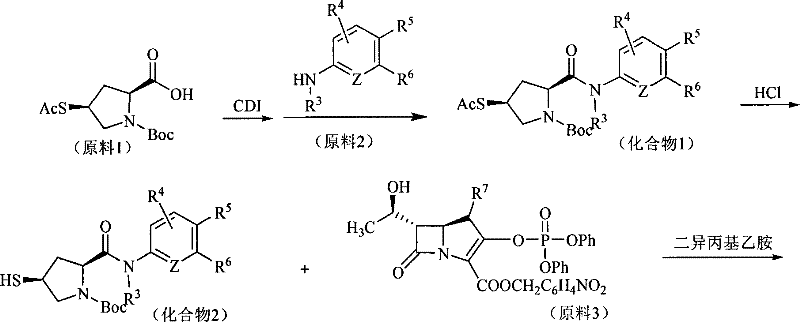
Impurity control is paramount in the production of beta-lactam intermediates, and the described mechanism incorporates specific steps to mitigate common degradation pathways. The use of low temperatures during the coupling and deprotection phases helps suppress epimerization at the chiral centers, ensuring the correct stereochemical configuration essential for biological activity. Additionally, the final hydrogenation step using palladium on charcoal serves a dual purpose: it removes benzyl-type protecting groups and reduces any potential nitro-group impurities that might arise from the nitromethane solvent or reagents. This comprehensive approach to reaction design ensures that the final product meets stringent purity specifications, reducing the burden on quality control laboratories and enhancing the overall reliability of the manufacturing process.
How to Synthesize Six-Membered Ring Carbapenem Intermediates Efficiently
The synthesis of these high-value intermediates involves a sequence of precise chemical transformations that must be carefully controlled to ensure safety and efficacy. The process begins with the activation of the pyrrolidine precursor, followed by coupling with the specific aniline derivative, and concludes with global deprotection to reveal the active carboxylic acid functionality. Each step requires strict adherence to anhydrous conditions and temperature controls to maintain the stability of the intermediates. The detailed standardized synthesis steps provided in the patent offer a clear roadmap for replicating these results in a GMP environment, ensuring that the process is robust enough for technology transfer.
- Activation of the pyrrolidine carboxylic acid precursor using coupling agents like CDI or DCC under anhydrous conditions.
- Coupling the activated acid with the appropriate aniline derivative to form the amide bond, followed by selective deprotection.
- Final deprotection of the carbapenem core using aluminum chloride or catalytic hydrogenation to yield the target active compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits in terms of operational efficiency and risk mitigation. The reliance on commodity chemicals such as CDI, DCC, and standard solvents like ethyl acetate and tetrahydrofuran means that the supply chain is less vulnerable to shortages of exotic reagents. This accessibility translates directly into enhanced supply chain reliability, as manufacturers can source materials from multiple vendors without compromising the quality of the final intermediate. Furthermore, the modular nature of the synthesis allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuations in market demand for different antibiotic variants.
- Cost Reduction in Manufacturing: The elimination of complex transition metal catalysts in the initial coupling steps significantly lowers the cost of goods sold. By avoiding expensive noble metals until the final deprotection stage, where they can be easily recovered and recycled, the process achieves substantial cost savings. Additionally, the high yields reported in the examples reduce the amount of raw material required per kilogram of finished product, further driving down production costs. The simplified purification protocols also decrease solvent consumption and waste disposal fees, contributing to a more economically viable manufacturing model.
- Enhanced Supply Chain Reliability: The use of stable intermediates throughout the synthesis reduces the risk of batch failures due to decomposition during storage or transport. This stability is a critical factor for maintaining continuous production lines and meeting delivery deadlines. The robustness of the chemical steps ensures that the process is less sensitive to minor variations in reaction conditions, which is essential for maintaining consistency across different production batches and sites. This reliability is key for reducing lead time for high-purity pharmaceutical intermediates in a competitive global market.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing unit operations that are easily transferred from pilot plant to commercial scale. The avoidance of highly toxic reagents and the use of recyclable solvents align with modern environmental regulations, reducing the ecological footprint of the manufacturing process. The efficient atom economy of the coupling reactions minimizes waste generation, supporting sustainable chemistry initiatives. These factors combined make the process highly attractive for long-term commercial partnerships focused on green manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbapenem intermediates. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for making informed decisions about integrating these compounds into your development pipeline.
Q: What distinguishes these carbapenem intermediates from existing market options?
A: These compounds feature a unique six-membered ring methanamide substitution on the pyrrolidine side chain, offering improved chemical stability and broad-spectrum activity against resistant organisms compared to traditional penem structures.
Q: Are the synthesis routes suitable for large-scale commercial production?
A: Yes, the patent describes robust methodologies utilizing standard reagents like CDI, DCC, and palladium catalysts, which are well-established in industrial settings, ensuring high purity and consistent quality for commercial scale-up.
Q: What are the primary applications of these intermediates?
A: They serve as critical precursors for next-generation carbapenem antibiotics designed to treat severe infectious diseases, including those caused by Gram-positive and Gram-negative bacteria, with potential for both injectable and oral formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the laboratory to the marketplace. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, providing you with the confidence needed to advance your clinical programs. Our expertise in beta-lactam chemistry positions us as a strategic partner capable of navigating the complexities of carbapenem synthesis.
We invite you to engage with our technical procurement team to discuss how we can support your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into optimizing your supply chain for these advanced intermediates. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver consistent, high-quality materials. Let us help you secure a reliable supply of these critical building blocks for your next-generation antibiotic formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
