Revolutionizing Phthalide Manufacturing: Direct Oxidation Technology for Commercial Scale-Up
The chemical manufacturing landscape for critical fine chemical intermediates is undergoing a significant transformation driven by the need for atom-economic processes and reduced environmental footprints. A pivotal development in this sector is detailed in patent CN108794439B, which discloses a highly selective preparation method for phthalide, a vital building block in the synthesis of pharmaceuticals and agrochemicals. This innovation shifts the paradigm from traditional multi-step hydrogenation routes to a streamlined direct oxidation process using o-xylene as the primary substrate. By leveraging a synergistic combination of transition metal catalysts and solid acid promoters, this technology achieves high conversion rates while minimizing the formation of stubborn byproducts that typically plague conventional synthesis methods. For R&D directors and procurement strategists, understanding this technological leap is essential for securing a competitive edge in the supply of high-purity pharmaceutical intermediates. The ability to produce phthalide directly from o-xylene not only simplifies the reaction pathway but also aligns with modern green chemistry principles, offering substantial potential for cost optimization and supply chain resilience in the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of phthalide has relied heavily on the hydrogenation of phthalic anhydride, a route that, while established, suffers from inherent inefficiencies and economic drawbacks. The conventional process necessitates the prior oxidation of o-xylene to phthalic anhydride, followed by a distinct and energy-intensive hydrogenation step to close the lactone ring. This multi-stage approach introduces significant operational complexity, requiring separate reactor setups and stringent control over hydrogen pressure and temperature to manage safety risks. Furthermore, the hydrogenation step is prone to generating undesirable byproducts such as o-toluic acid and o-methylcyclohexanecarboxylic acid, which complicate downstream purification and reduce overall yield. The reliance on noble metal catalysts in some traditional variations further exacerbates cost pressures, as these materials are subject to volatile market pricing and require complex recovery systems to prevent loss. Consequently, the cumulative material and energy consumption of the phthalic anhydride hydrogenation method creates a substantial burden on manufacturing budgets, limiting the flexibility of supply chains to respond to fluctuating demand for high-purity pharmaceutical intermediates without incurring prohibitive costs.
The Novel Approach
In stark contrast to the legacy hydrogenation routes, the novel methodology presented in the patent data utilizes a direct catalytic oxidation strategy that fundamentally restructures the synthesis pathway for improved efficiency. By employing o-xylene directly as the starting material, this approach bypasses the isolation of phthalic anhydride entirely, effectively merging the oxidation and cyclization steps into a single, cohesive operation. The core of this innovation lies in the precise selection of a solid acid catalyst, which facilitates the in-situ esterification of the intermediate ortho-hydroxymethyl benzoic acid, driving the reaction directly toward the ring-closed phthalide product. This telescoping of reaction steps not only drastically reduces the physical footprint required for production but also minimizes the thermal energy input needed for intermediate handling and separation. The result is a process characterized by exceptional atom economy, where the majority of the starting material is converted into the desired product rather than waste streams. For supply chain leaders, this translates to a more robust manufacturing protocol that is less susceptible to the bottlenecks associated with multi-step processing, thereby enhancing the reliability of supply for critical fine chemical intermediates used in the production of anticoagulants and fungicides.
Mechanistic Insights into Transition Metal and Solid Acid Catalyzed Oxidation
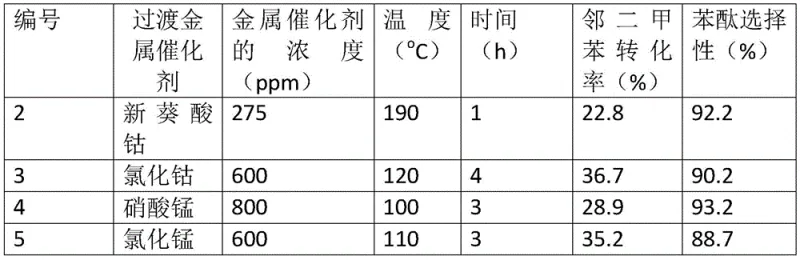
The chemical elegance of this synthesis route is underpinned by a sophisticated dual-catalyst mechanism that orchestrates the selective oxidation of methyl groups on the o-xylene molecule. The transition metal component, typically selected from cobalt or manganese salts such as cobalt isooctanoate or manganese naphthenate, acts as the primary driver for the activation of molecular oxygen. These metal centers facilitate the radical chain reactions necessary to oxidize one of the methyl groups on the o-xylene ring to a hydroxymethyl group and subsequently to a carboxylic acid functionality, forming the critical ortho-hydroxymethyl benzoic acid intermediate. The concentration of these transition metals is meticulously optimized, often ranging from 10ppm to 2000ppm, to ensure sufficient catalytic activity without promoting over-oxidation to phthalic acid or other degradation products. This precise control over the oxidation state is crucial for maintaining high selectivity, as it prevents the formation of fully oxidized byproducts that would otherwise contaminate the final phthalide stream and necessitate expensive purification protocols.
Complementing the oxidation function, the solid acid catalyst plays a pivotal role in the intramolecular cyclization step that defines the formation of the lactone ring. Materials such as Amberlyst-15, HY zeolites, or H-ZSM-5 provide the necessary acidic protons to catalyze the esterification between the newly formed carboxylic acid group and the adjacent hydroxymethyl group. This acid-catalyzed dehydration occurs in situ, effectively pulling the equilibrium toward the desired phthalide product and preventing the accumulation of the open-chain acid intermediate. The use of heterogeneous solid acids offers distinct advantages over liquid acids, including ease of separation from the reaction mixture and the potential for catalyst reuse, which further contributes to the economic viability of the process. By harmonizing the oxidative power of the transition metal with the cyclization capability of the solid acid, the system achieves a high degree of chemo-selectivity, ensuring that the reaction proceeds cleanly to phthalide with minimal generation of structural isomers or polymeric tars.
How to Synthesize Phthalide Efficiently
Implementing this advanced synthesis route requires a systematic approach to reactor configuration and parameter control to maximize the benefits of the dual-catalyst system. The process begins with the preparation of a homogeneous reaction mixture where o-xylene is combined with the transition metal catalyst at specific concentrations, typically optimized around 400-600ppm for cobalt-based systems to balance activity and cost. Following this, the solid acid catalyst is introduced into the autoclave, with loading amounts carefully calibrated between 1% to 100% of the substrate mass depending on the specific acidity and surface area of the chosen material. The reaction is then conducted under a controlled oxygen atmosphere, with pressures maintained between 0.1MPa and 2MPa to ensure adequate oxygen solubility without creating unsafe operating conditions. Temperature control is equally critical, with the reaction window spanning from 80°C to 190°C, allowing operators to tune the reaction rate and selectivity based on the specific kinetic profile of the catalyst batch. Detailed standard operating procedures for scaling this chemistry from laboratory benchtop to commercial production volumes are essential for maintaining consistency and safety.
- Prepare the reaction system by mixing o-xylene substrate with a transition metal catalyst such as cobalt isooctanoate at a concentration of 10-2000ppm.
- Add a solid acid catalyst like Amberlyst-15 or H-ZSM-5 to the mixture, ensuring the mass ratio is optimized between 1% to 100% relative to the substrate.
- Conduct the oxidation reaction in a sealed autoclave at temperatures between 80°C to 190°C under an oxygen pressure of 0.1 to 2MPa for 0.5 to 10 hours.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this direct oxidation technology offers compelling advantages that directly address the core concerns of cost management and supply continuity in the fine chemicals sector. By eliminating the intermediate isolation of phthalic anhydride and the subsequent hydrogenation step, manufacturers can significantly reduce the overall processing time and energy consumption associated with phthalide production. This simplification of the process flow reduces the capital expenditure required for plant infrastructure, as fewer distinct reaction units and separation columns are needed to achieve the same output volume. Furthermore, the reliance on o-xylene, a widely available commodity chemical, as the sole carbon source mitigates the supply risks associated with sourcing specialized hydrogenation feedstocks. The removal of noble metal catalysts from the equation also shields buyers from the volatility of precious metal markets, replacing them with more stable and abundant transition metal salts. These factors combine to create a manufacturing profile that is inherently more cost-effective and resilient, providing a stable foundation for long-term supply agreements.
- Cost Reduction in Manufacturing: The economic benefits of this novel route are primarily derived from the drastic simplification of the synthetic pathway, which removes entire unit operations from the production line. By avoiding the hydrogenation step, facilities save on the substantial costs associated with high-pressure hydrogen handling, specialized hydrogenation reactors, and the safety systems required to manage flammable gases. Additionally, the high selectivity of the solid acid catalyzed cyclization minimizes the loss of raw materials to byproducts, thereby improving the overall mass balance and reducing the cost per kilogram of the final active pharmaceutical ingredient intermediate. The use of heterogeneous solid acids also facilitates easier catalyst recovery and potential regeneration, further lowering the recurring operational expenses related to catalyst consumption. These cumulative efficiencies result in a leaner cost structure that allows for more competitive pricing without compromising on the quality or purity specifications required by downstream pharmaceutical manufacturers.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of o-xylene as the primary feedstock, a chemical that is produced on a massive global scale for the petrochemical industry. Unlike specialized intermediates that may be subject to supply constraints from a limited number of vendors, o-xylene is readily accessible from multiple sources, reducing the risk of raw material shortages disrupting production schedules. The robustness of the solid acid catalysts, which are less sensitive to poisoning and deactivation compared to some homogeneous systems, ensures consistent campaign lengths and reduces the frequency of unplanned shutdowns for catalyst replacement. This operational reliability translates directly into more predictable lead times for customers, enabling procurement managers to plan their inventory levels with greater confidence. The ability to maintain continuous production runs with minimal intervention enhances the overall agility of the supply chain, allowing suppliers to respond more rapidly to spikes in demand for phthalide derivatives used in critical medical applications.
- Scalability and Environmental Compliance: The environmental profile of this direct oxidation method aligns perfectly with the increasingly stringent regulatory requirements facing the chemical industry today. The atom-economic nature of the reaction means that less waste is generated per unit of product, reducing the burden on waste treatment facilities and lowering the costs associated with environmental compliance and disposal. The absence of heavy metal contaminants often associated with traditional oxidation processes simplifies the purification of the final product, ensuring that it meets the rigorous purity standards demanded by the pharmaceutical sector without extensive downstream processing. Furthermore, the scalability of the process is enhanced by the use of standard oxidation equipment that is common in fine chemical plants, facilitating a smoother transition from pilot scale to multi-ton commercial production. This ease of scale-up ensures that supply can be expanded rapidly to meet market growth without the need for exotic or hard-to-source engineering solutions, securing a sustainable long-term supply of high-quality phthalide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this direct oxidation technology for phthalide synthesis. These insights are derived directly from the technical disclosures and experimental data provided in the patent literature, offering a clear understanding of the process capabilities. By clarifying these points, we aim to assist technical decision-makers in evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers focus on the practical implications of catalyst selection, reaction conditions, and the comparative advantages over legacy methods.
Q: What are the primary advantages of the direct oxidation method over phthalic anhydride hydrogenation?
A: The direct oxidation method described in patent CN108794439B eliminates the need for a separate hydrogenation step, significantly reducing process complexity and energy consumption while improving atom economy.
Q: Which catalysts are essential for achieving high selectivity in this phthalide synthesis route?
A: High selectivity is achieved through a dual-catalyst system comprising transition metals like cobalt or manganese salts combined with solid acid catalysts such as Amberlyst-15 or zeolites.
Q: How does this technology impact the impurity profile of the final phthalide product?
A: By avoiding the hydrogenation of phthalic anhydride, this route minimizes the formation of byproducts like o-toluic acid and o-methylcyclohexanecarboxylic acid, resulting in a cleaner crude product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalide Supplier
As the global demand for high-purity pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses both technical expertise and scalable capacity is paramount for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the direct oxidation of o-xylene to deliver superior quality phthalide to our international clients. Our team of expert chemists and engineers has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on stringent purity specifications. We operate state-of-the-art rigorous QC labs that monitor every batch for impurities and consistency, guaranteeing that the material you receive is perfectly suited for the synthesis of complex drug substances. Our commitment to technical excellence allows us to adapt quickly to specific customer needs, providing a level of service that goes beyond simple commodity supply.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By understanding your current supply chain challenges, our technical procurement team can provide specific COA data and route feasibility assessments that demonstrate the tangible value of switching to our optimized manufacturing processes. Whether you are looking to secure a long-term supply agreement or need support in scaling a new development candidate, we are ready to collaborate closely with your organization. Contact us today to discuss how our advanced phthalide synthesis capabilities can enhance your product pipeline and drive efficiency in your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
