Advanced Chiral Resolution for Chloramphenicol: Scaling High-Purity API Production
The pharmaceutical industry continuously seeks methods to enhance the efficiency of antibiotic manufacturing, particularly for broad-spectrum agents like Chloramphenicol. Patent CN1322711A presents a groundbreaking approach to the preparation of optically active Chloramphenicol base, addressing the long-standing inefficiency of traditional synthetic routes. Historically, the production of this critical antibiotic has been plagued by the generation of useless enantiomers, specifically the (1S, 2S) isomer, which offers no therapeutic value and constitutes significant chemical waste. This patent discloses a novel methodology utilizing RS-(±)-2-amino-3-hydroxy-1-p-nitrophenylacetone hydrochloride as a key starting material. By implementing a sophisticated sequence of chiral resolution and configuration inversion, the process effectively eliminates the formation of the dextrorotatory byproduct. This technical advancement not only improves the overall yield of the biologically active (1R, 2R) enantiomer but also aligns with modern green chemistry principles by minimizing waste generation. For R&D directors and procurement specialists, understanding this shift from linear synthesis to dynamic kinetic resolution is essential for evaluating supply chain robustness and cost structures in the generic antibiotic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Chloramphenicol, often originating from p-nitroacetophenone or ethylbenzene, involve a lengthy sequence of nitration, oxidation, bromination, and condensation steps. As illustrated in the historical process flow, these conventional methods suffer from a fundamental stereochemical bottleneck. During the condensation and reduction phases, the reaction typically produces a racemic mixture or a specific diastereomer that requires extensive separation. The most critical drawback is the inevitable formation of the (1S, 2S)-2-amino-1-(4-nitrophenyl)-1,3-propanediol isomer, commonly referred to as the right-amine or useless isomer. In standard industrial practice, for every ton of useful Chloramphenicol base produced, approximately one ton of this useless isomer is generated as waste. This 1:1 ratio of waste-to-product imposes a severe burden on downstream processing, requiring complex separation techniques to isolate the active levo-form while disposing of the dextro-form. Furthermore, the disposal of this chemically active waste introduces significant environmental compliance costs and safety hazards, complicating the regulatory approval process for manufacturing facilities.
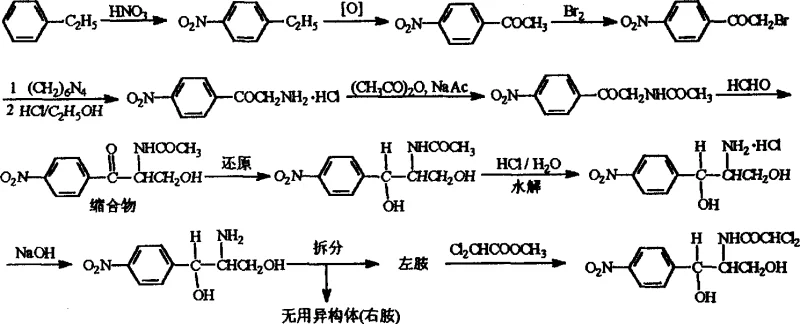
The Novel Approach
In stark contrast to the wasteful conventional pathways, the novel approach detailed in the patent re-engineers the synthesis at the chiral center formation stage. Instead of accepting the racemic outcome, this method introduces a resolution step early in the sequence using the intermediate RS-(±)-2-amino-3-hydroxy-1-p-nitrophenylacetone hydrochloride. The innovation lies in the dual capability of the process to not only separate the enantiomers but also to invert the configuration of the unwanted isomer. By employing specific chiral resolving agents, such as cyclic phosphoric acids or carboxylic acid derivatives, the process selectively crystallizes the desired enantiomer. Crucially, the mother liquor containing the unwanted isomer is not discarded; instead, it undergoes a configuration inversion reaction. This transforms the useless (S,S) or (R,R) precursors into the useful configuration, theoretically allowing for 100% atom economy regarding the chiral carbon skeleton. This shift from a linear, wasteful process to a cyclic, restorative process represents a paradigm shift in antibiotic intermediate manufacturing, offering substantial advantages in raw material utilization and waste reduction.
Mechanistic Insights into Chiral Resolution and Configuration Inversion
The core of this technological breakthrough relies on the precise interaction between the racemic amino keto hydrochloride substrate and the chiral resolving agents. The patent specifies the use of chiral cyclic phosphoric acids or chiral carboxylic acids, which are often derived from the Chloramphenicol base itself to ensure stereochemical compatibility. When the racemic mixture is treated with a resolving agent of a specific configuration, such as the (4R, 5R) or (4S, 5S) cyclic phosphoric acid derivatives, diastereomeric salts are formed. These salts possess different physical properties, particularly solubility, allowing for their separation via fractional crystallization. The mechanism involves the formation of a rigid crystalline lattice that preferentially incorporates one enantiomer over the other. For instance, using a resolving agent derived from the left-amine (R,R-configuration) facilitates the precipitation of the R-(-)-2-amino-3-hydroxy-1-p-nitrophenyl-1-acetone hydrochloride. The optical purity of the isolated salt is critical, with the patent indicating that optical purity can exceed 90% through optimized crystallization conditions involving polar solvents like methanol or ethanol at controlled temperatures ranging from 15°C to 50°C.
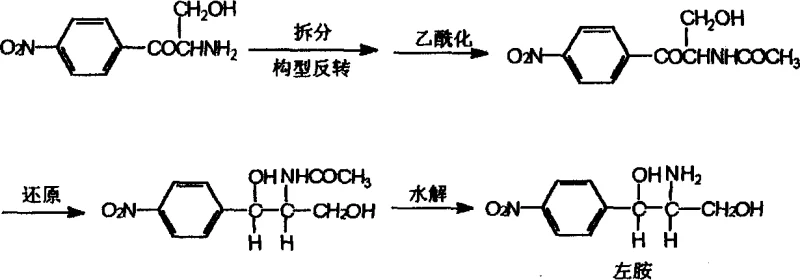
Following the initial separation, the process addresses the remaining isomer through a configuration inversion mechanism. This step is vital for maximizing yield and eliminating waste. The unwanted isomer, which would traditionally be discarded, is subjected to conditions that invert its stereochemistry at the chiral center. This is achieved by manipulating the pH and utilizing specific resolving agent salts that facilitate the epimerization or inversion of the amino ketone structure. Once inverted, this material re-enters the resolution cycle as the desired enantiomer. Subsequent steps involve dichloroacetylation or acetylation followed by reduction with aluminum isopropoxide. The reduction step is particularly sensitive, requiring strict control to maintain the stereochemical integrity established during the resolution. The use of aluminum isopropoxide ensures the selective reduction of the ketone group to the alcohol without affecting the nitro group or the amide functionality,最终 yielding the optically active Chloramphenicol base with high stereochemical purity. This mechanistic control ensures that the final product meets the stringent impurity profiles required for pharmaceutical applications.
How to Synthesize Optically Active Chloramphenicol Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the resolving agents and the control of crystallization parameters. The process begins with the hydrolysis of protected precursors to generate the free amino ketone hydrochloride, which is then subjected to the resolution cycle. Detailed standard operating procedures for the stoichiometry of resolving agents, solvent ratios, and temperature gradients are essential for reproducibility. The patent outlines specific examples using various cyclic phosphoric acid derivatives, demonstrating the flexibility of the method across different resolving agent structures. For a comprehensive understanding of the operational parameters, including specific molar ratios and reaction times, please refer to the standardized synthesis guide below.
- Prepare the racemic amino keto hydrochloride substrate and mix with a chiral resolving agent such as cyclic phosphoric acid derivatives.
- Conduct fractional crystallization to separate the diastereomeric salts, isolating the desired S-(+) or R-(-) enantiomer.
- Perform configuration inversion on the mother liquor or unwanted isomer to convert it into the useful enantiomer, followed by dichloroacetylation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this resolution and inversion technology offers transformative benefits for procurement managers and supply chain directors. The primary advantage lies in the drastic reduction of raw material costs associated with waste. By converting the useless isomer into the active product, the effective yield of the process is nearly doubled compared to traditional methods where the byproduct is discarded. This efficiency translates directly into lower cost of goods sold (COGS) and reduced dependency on volatile raw material markets. Furthermore, the elimination of large volumes of chemical waste simplifies the environmental compliance landscape. Facilities no longer need to invest heavily in the treatment and disposal of the (1S, 2S) isomer, leading to significant operational savings and a reduced carbon footprint. This aligns with the increasing global demand for sustainable manufacturing practices in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the theoretical 100% utilization of the chiral intermediate. In traditional synthesis, half of the material value is lost to the useless enantiomer. By recovering and inverting this material, the process effectively reduces the raw material consumption per kilogram of final API. Additionally, the resolving agents described in the patent can be recovered and recycled from the mother liquor, further lowering the recurring cost of reagents. This creates a closed-loop system that minimizes variable costs and stabilizes production economics against fluctuations in the price of starting materials like p-nitroacetophenone derivatives.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of waste management and the availability of specialized reagents. This simplified route reduces the number of distinct chemical inputs required and minimizes the generation of hazardous byproducts that might face regulatory shipping restrictions. The ability to source resolving agents that can be synthesized from the product stream itself adds a layer of supply chain security, reducing reliance on external specialty chemical vendors. This self-sufficiency ensures that production schedules are less vulnerable to external supply shocks, guaranteeing more reliable delivery timelines for downstream pharmaceutical formulators.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste handling challenges. This technology mitigates that risk by designing waste out of the process at the molecular level. The reduction in total effluent volume and the lower toxicity profile of the waste stream facilitate easier permitting and expansion of manufacturing capacity. Facilities can scale up from pilot batches to multi-ton commercial production without proportionally increasing their environmental liability. This scalability is crucial for meeting the growing global demand for generic antibiotics while adhering to increasingly strict environmental, social, and governance (ESG) standards imposed by international regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral resolution technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the method for industry stakeholders. Understanding these details is key for R&D teams evaluating technology transfer and procurement teams assessing vendor capabilities.
Q: How does this patent address the issue of useless isomers in Chloramphenicol production?
A: The patent introduces a resolution and configuration inversion strategy that converts the traditionally useless (1S, 2S) isomer into the biologically active (1R, 2R) form, theoretically achieving 100% utilization of the starting material.
Q: What resolving agents are suitable for this chiral separation process?
A: The method utilizes specific chiral cyclic phosphoric acids or chiral carboxylic acids derived from Chloramphenicol base itself, ensuring high stereochemical compatibility and recovery rates.
Q: Is this process scalable for industrial manufacturing of antibiotics?
A: Yes, the process avoids complex asymmetric synthesis catalysts and relies on standard crystallization and acetylation steps, making it highly suitable for large-scale commercial production with reduced environmental burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloramphenicol Supplier
The technical potential of the resolution and configuration inversion route described in patent CN1322711A represents a significant opportunity for optimizing the production of high-purity Chloramphenicol. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated chemistry to the market. Our engineering team is adept at translating laboratory-scale resolution protocols into robust, GMP-compliant manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Chloramphenicol base meets the exacting standards of the global pharmaceutical industry. Our commitment to quality ensures that the optical purity and impurity profiles of our products are consistently verified, providing our partners with the confidence needed for regulatory filings.
We invite procurement leaders and technical directors to collaborate with us on optimizing their supply chains for antibiotic intermediates. By leveraging our expertise in chiral synthesis and process optimization, we can help your organization achieve significant efficiency gains. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals. Let us partner to engineer a more efficient and sustainable supply chain for essential medicines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
