Advanced Palladium-Catalyzed Synthesis Of Isochroman Amides For Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis Of Isochroman Amides For Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN114539198A introduces a groundbreaking preparation method for amide compounds containing an (iso)chroman structure, addressing critical challenges in modern organic synthesis. This technology leverages a palladium-catalyzed cyclocarbopalladation and aminocarbonylation sequence, utilizing nitroaromatic hydrocarbons as a nitrogen source and molybdenum carbonyl as both a carbonyl source and a reducing agent. The significance of this innovation lies in its ability to streamline the production of high-value intermediates used in drug discovery and development. By replacing hazardous carbon monoxide gas with solid molybdenum carbonyl, the process enhances operational safety while maintaining high reaction efficiency. For global procurement teams and R&D directors, this patent represents a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering complex molecules with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of amide bonds relies heavily on the acylation of amines with carboxylic acids or their activated derivatives, which often generates substantial stoichiometric waste and requires harsh coupling reagents. Alternatively, transition metal-catalyzed carbonylation of halogenated aryl compounds with amines typically necessitates the use of high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized equipment that increases capital expenditure. Furthermore, conventional routes often struggle with substrate compatibility, particularly when sensitive functional groups are present on the aromatic rings, leading to side reactions and reduced purity profiles. These limitations create bottlenecks in cost reduction in pharmaceutical intermediates manufacturing, as the need for extensive purification and safety measures drives up the overall production cost. Additionally, the reliance on pre-functionalized amine starting materials can introduce supply chain vulnerabilities, as these precursors may not always be readily available in bulk quantities. Consequently, manufacturers face difficulties in achieving commercial scale-up of complex pharmaceutical intermediates without compromising on safety or economic feasibility.
The Novel Approach
In contrast, the methodology disclosed in CN114539198A offers a transformative solution by employing nitroarenes as inexpensive and stable nitrogen surrogates, effectively bypassing the need for pre-formed amines. The use of molybdenum carbonyl serves a dual purpose, acting as a solid source of carbon monoxide for the carbonylation step while simultaneously reducing the nitro group in situ, which simplifies the reaction setup and eliminates the handling of toxic gases. This approach allows for the direct construction of the (iso)chroman amide skeleton from simple iodoarene and nitroarene precursors under relatively mild thermal conditions. The reaction system demonstrates exceptional versatility, accommodating a wide range of substituents on both the iodoarene and nitroarene components without significant loss in yield. 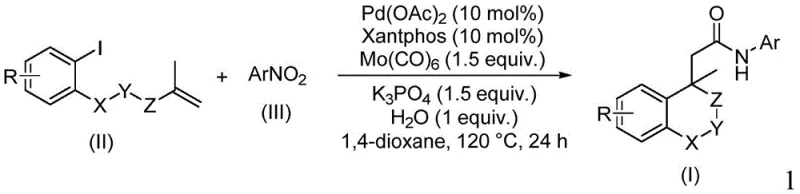
Mechanistic Insights into Pd-Catalyzed Cyclocarbopalladation
The core of this synthetic strategy involves a sophisticated palladium-catalyzed cascade that begins with the oxidative addition of the iodoarene to the Pd(0) species, followed by an intramolecular Heck-type cyclization to form the chroman ring system. The resulting sigma-alkylpalladium intermediate then undergoes carbon monoxide insertion, facilitated by the decomposition of molybdenum carbonyl, to generate an acyl-palladium complex. Subsequently, the nitroarene is reduced by the molybdenum species to an amine or hydroxylamine intermediate, which then attacks the acyl-palladium species to form the final amide bond and regenerate the catalyst. This intricate mechanism ensures high atom economy and minimizes the formation of by-products, which is crucial for maintaining high-purity pharmaceutical intermediates standards. The choice of the Xantphos ligand is critical, as its wide bite angle stabilizes the palladium center and promotes the reductive elimination step, thereby enhancing the overall turnover number of the catalyst. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters for optimal performance during technology transfer.
Impurity control is another vital aspect where this method excels, as the specific reaction conditions suppress common side reactions such as homocoupling of the iodoarene or over-reduction of the nitro group. The use of potassium phosphate as a base provides a buffered environment that prevents the degradation of sensitive functional groups like esters or ketones during the high-temperature reaction phase. Moreover, the compatibility with various substituents, including halogens and electron-withdrawing groups, ensures that the impurity profile remains manageable even with diverse substrate inputs. 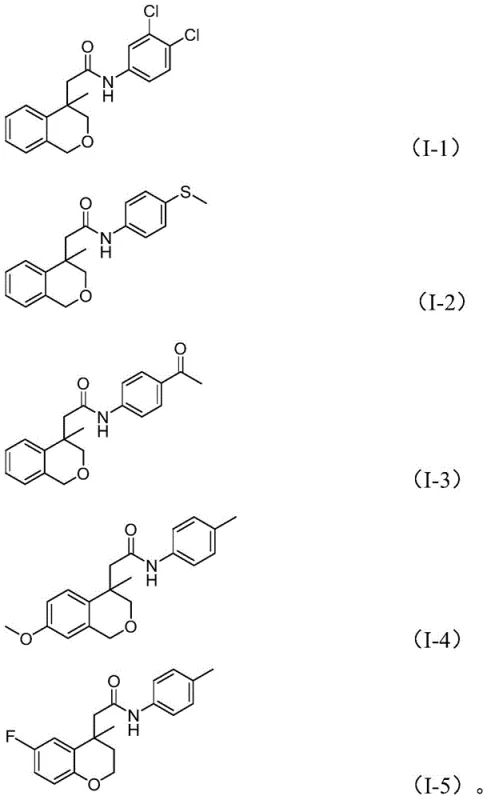
How to Synthesize Isochroman Amide Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and thermal conditions to maximize yield and minimize waste generation. The standard protocol involves charging a reaction vessel with palladium acetate, the Xantphos ligand, molybdenum carbonyl, potassium phosphate, and water, followed by the addition of the iodoarene and nitroarene substrates in 1,4-dioxane solvent. The mixture is then heated to 120°C and stirred for approximately 24 hours to ensure complete conversion of the starting materials into the desired amide product. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored for different substrate variations. Adhering to these parameters is essential for reproducing the high efficiency reported in the patent data and ensuring batch-to-batch consistency.
- Mix palladium acetate, Xantphos ligand, molybdenum carbonyl, potassium phosphate, water, iodoarene, and nitroarene in 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target amide compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for complex chemical building blocks. The elimination of high-pressure carbon monoxide gas cylinders significantly reduces safety compliance costs and infrastructure requirements, making the process more accessible for contract manufacturing organizations. Furthermore, the use of commercially available and inexpensive starting materials like nitroarenes and iodoarenes ensures a stable supply chain that is less susceptible to market fluctuations compared to specialized amine reagents. This stability translates into enhanced supply chain reliability, allowing manufacturers to plan production schedules with greater confidence and reduce lead time for high-purity pharmaceutical intermediates. The simplified workup procedure, involving filtration and column chromatography, also contributes to lower processing times and reduced solvent consumption, aligning with green chemistry principles.
- Cost Reduction in Manufacturing: The replacement of gaseous carbon monoxide with solid molybdenum carbonyl removes the need for expensive high-pressure reactors and associated safety monitoring systems, leading to significant capital and operational expenditure savings. Additionally, the use of nitroarenes as nitrogen sources avoids the cost premiums often associated with functionalized amine starting materials, further driving down the raw material bill. The high reaction efficiency and broad substrate tolerance mean that fewer batches are rejected due to quality issues, optimizing the overall cost of goods sold. These factors combined create a compelling economic case for adopting this methodology in large-scale production environments.
- Enhanced Supply Chain Reliability: Since the key raw materials such as iodoarenes and nitroarenes are commodity chemicals produced by multiple vendors globally, the risk of supply disruption is markedly reduced. This abundance allows procurement teams to negotiate better pricing and secure long-term contracts without fear of single-source dependency. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, ensuring continuity of supply even if one facility faces operational challenges. Such flexibility is invaluable for maintaining uninterrupted production lines for critical drug intermediates.
- Scalability and Environmental Compliance: The process operates in a homogeneous liquid phase using standard organic solvents, which facilitates straightforward scale-up from laboratory to pilot and commercial scales without encountering mass transfer limitations. The absence of toxic gas feeds simplifies environmental permitting and reduces the burden on waste treatment facilities, as the primary by-products are solid residues that can be managed more easily. This alignment with environmental regulations enhances the corporate sustainability profile of manufacturers adopting this technology, meeting the increasing demand for eco-friendly chemical processes from downstream pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. We encourage technical teams to review these points closely when assessing process viability.
Q: Why are nitroarenes preferred over traditional amines in this synthesis?
A: Nitroarenes serve as abundant, stable, and inexpensive nitrogen sources that eliminate the need for pre-functionalized amines, thereby simplifying the supply chain and reducing raw material costs significantly.
Q: What is the dual role of molybdenum carbonyl in this reaction?
A: Molybdenum carbonyl acts simultaneously as the carbonyl source for amide bond formation and as a reducing agent to convert nitro groups into amino intermediates, streamlining the reaction process.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates wide functional group tolerance, accommodating substituents such as halogens, alkoxy groups, and trifluoromethyl groups without compromising reaction efficiency or yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Amide Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of Isochroman Amide meets the highest international standards required by top-tier pharmaceutical companies. We understand the critical nature of supply continuity and cost efficiency, which is why we continuously invest in process optimization technologies that align with the latest patent advancements. Partnering with us means gaining access to a team of experts dedicated to solving complex synthesis challenges while maintaining competitive pricing structures.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. By collaborating with us, you can obtain specific COA data and route feasibility assessments that demonstrate how this novel synthesis method can benefit your supply chain. Let us help you navigate the complexities of chemical manufacturing and secure a reliable source for your critical intermediates today.
