Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
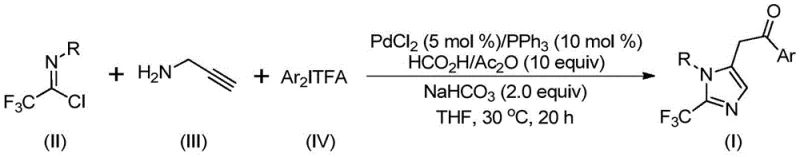
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds, particularly those incorporating fluorine atoms which significantly enhance metabolic stability and bioavailability. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical challenges in modern medicinal chemistry. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing inexpensive and commercially available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. The significance of this innovation lies in its ability to construct the imidazole core under remarkably mild conditions, specifically at 30°C, thereby preserving sensitive functional groups often required in advanced drug candidates. As a leading entity in fine chemical manufacturing, we recognize this pathway as a pivotal advancement for producing high-purity pharmaceutical intermediates efficiently.
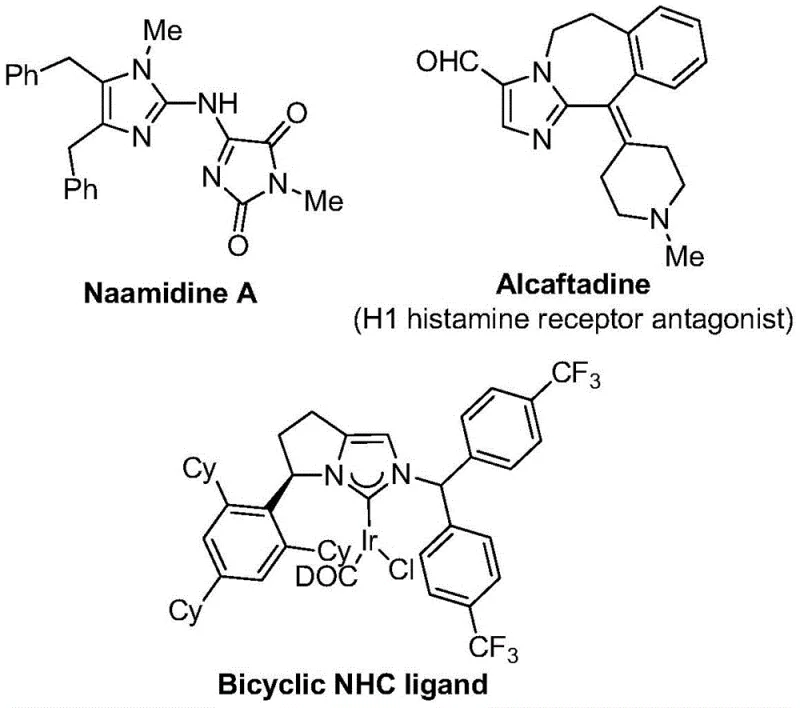
Imidazole compounds represent a cornerstone class of nitrogen-containing five-membered heterocycles, ubiquitous in the molecular frameworks of biologically active agents ranging from antihistamines like Alcaftadine to complex natural products like Naamidine A. The strategic introduction of a trifluoromethyl group into these heterocyclic systems offers profound advantages, including improved electronegativity and lipophilicity, which are essential parameters for optimizing drug-receptor interactions. However, traditional synthetic routes often rely on hazardous reagents or harsh conditions that limit their utility in large-scale manufacturing. The novel approach detailed in this patent circumvents these limitations by employing a safe and efficient carbonylation strategy. This method not only simplifies the operational workflow but also broadens the scope of accessible derivatives, making it an invaluable tool for reliable pharmaceutical intermediate suppliers aiming to diversify their portfolio with high-value fluorinated building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functionalities has been fraught with significant technical hurdles and safety concerns. Conventional literature methods predominantly utilize synthons such as trifluorodiazoethane, which, while effective, pose substantial risks due to their explosive nature and instability during storage and handling. Furthermore, alternative approaches involving trifluoroethylimide acid halides have seen limited application despite their potential, largely due to a lack of efficient catalytic systems capable of activating these substrates under mild conditions. These legacy processes often require elevated temperatures, strong bases, or toxic solvents, which complicate downstream purification and increase the environmental footprint of the manufacturing process. For procurement managers and supply chain heads, these factors translate into higher operational costs, stringent safety protocols, and potential bottlenecks in the consistent supply of critical API precursors.
The Novel Approach
In stark contrast, the methodology disclosed in CN111423381A presents a paradigm shift by utilizing a palladium-catalyzed carbonylation series reaction that operates at a gentle 30°C. This innovative route combines trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts in a one-pot transformation that efficiently constructs the 2-trifluoromethyl imidazole core. The use of diaryliodonium salts as arylating agents is particularly advantageous, as they serve as stable and versatile sources of aryl groups that facilitate the oxidative addition step in the catalytic cycle. By replacing dangerous diazo compounds with stable imidoyl chlorides and leveraging the power of transition metal catalysis, this method achieves high reaction efficiency and excellent substrate compatibility. This technological leap enables the synthesis of diversified substituted imidazoles with trifluoromethyl groups, providing a scalable solution for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for process development. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the reactive centers for cyclization. The palladium catalyst, generated in situ from PdCl2 and triphenylphosphine, then engages in an aminopalladation of the alkyne moiety, forming a key alkenyl palladium intermediate. Through a subsequent isomerization event, this evolves into an alkyl palladium species, setting the stage for the critical carbonylation step. The carbon monoxide required for this insertion is conveniently released from the decomposition of the formic acid and acetic anhydride mixture added to the reaction system, eliminating the need for high-pressure CO gas cylinders and enhancing laboratory safety.
Following the carbonylation, an acyl palladium intermediate is formed, which then undergoes oxidative addition with the diaryliodonium salt. This step generates a high-valent tetravalent palladium intermediate, a rare and highly reactive species that facilitates the final carbon-carbon bond formation. The catalytic cycle concludes with a reductive elimination step, releasing the final 2-trifluoromethyl substituted imidazole compound and regenerating the active palladium catalyst. This intricate cascade demonstrates exceptional chemoselectivity, tolerating a wide array of functional groups including halogens, nitro groups, and ethers on both the R and Ar aromatic rings. Such mechanistic robustness ensures that the impurity profile remains manageable, a critical factor for meeting the stringent purity specifications required in the production of high-purity OLED materials or pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it highly attractive for contract development and manufacturing organizations (CDMOs). The standard protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, and the CO source mixture in an aprotic organic solvent, preferably tetrahydrofuran (THF). To this mixture, the three key coupling partners—trifluoroethylimidoyl chloride, propargylamine, and the diaryliodonium salt—are added. The reaction proceeds at a constant temperature of 30°C for a duration of 16 to 24 hours, ensuring complete conversion of the starting materials. Post-reaction processing is straightforward, involving filtration to remove inorganic salts followed by silica gel column chromatography to isolate the pure product. For a detailed breakdown of the standardized operating procedures and specific molar ratios, please refer to the guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Heat the mixture at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of global supply chains and procurement strategies. The reliance on cheap and readily available starting materials, such as aromatic amines for preparing the imidoyl chlorides and commercially sourced propargylamine, significantly de-risks the raw material supply chain. Unlike specialized fluorinating reagents that may have long lead times or single-source dependencies, the precursors for this method are commodity chemicals available from multiple vendors worldwide. This abundance ensures supply continuity and mitigates the risk of production stoppages due to raw material shortages. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, contributing to a more sustainable and cost-effective manufacturing process that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by the use of palladium chloride, which is relatively inexpensive compared to other noble metal catalysts, and the avoidance of high-pressure equipment. By generating carbon monoxide in situ from formic acid and acetic anhydride, the process eliminates the capital expenditure associated with CO gas infrastructure and safety monitoring systems. Additionally, the high reaction yields reported across various substrates minimize waste generation and maximize atom economy, leading to substantial cost savings in raw material utilization. These factors collectively drive down the cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain reliability. The tolerance for diverse functional groups means that a single platform technology can be used to produce a wide library of derivatives without needing to re-optimize conditions for each new analog. This flexibility allows manufacturers to respond rapidly to changing market demands or custom synthesis requests from clients. Moreover, the scalability of the method, demonstrated from milligram to gram scales in the patent data, suggests a clear path to multi-kilogram or ton-scale production. This scalability ensures that suppliers can meet bulk orders consistently, reducing lead times for high-purity pharmaceutical intermediates and strengthening partnerships with downstream drug developers.
- Scalability and Environmental Compliance: Environmental compliance is increasingly becoming a decisive factor in vendor selection, and this method scores highly in this regard. The use of THF as a solvent, while requiring recovery systems, is standard in the industry, and the absence of toxic heavy metal waste or explosive reagents simplifies waste treatment protocols. The simple workup procedure involving filtration and chromatography reduces the volume of aqueous waste generated compared to traditional extraction-heavy processes. As regulatory pressures mount on the chemical industry to reduce its environmental footprint, adopting such cleaner synthetic routes provides a strategic advantage. It positions manufacturers as responsible partners capable of delivering complex molecules while adhering to strict environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
To further clarify the technical nuances and commercial implications of this synthesis method, we have compiled a set of frequently asked questions based on the patent specifications. These insights are derived directly from the experimental data and mechanistic descriptions provided in the intellectual property documentation. They are intended to assist technical decision-makers in evaluating the fit of this technology for their specific project requirements. Whether you are concerned about catalyst loading, substrate scope, or purification strategies, the following answers provide a solid foundation for understanding the capabilities of this palladium-catalyzed system.
Q: What are the optimal reaction conditions for this Pd-catalyzed synthesis?
A: The patent specifies a mild temperature of 30°C with a reaction time of 16 to 24 hours. The preferred solvent is tetrahydrofuran (THF), utilizing PdCl2 and PPh3 as the catalytic system with sodium bicarbonate as the base.
Q: Does this method tolerate diverse functional groups on the aryl rings?
A: Yes, the methodology demonstrates excellent substrate compatibility. It successfully accommodates electron-donating groups like methyl and tert-butyl, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl substituents on both the imidoyl chloride and the iodonium salt components.
Q: Is this process suitable for large-scale industrial production?
A: The patent explicitly states that the method can be expanded to the gram level and potentially for industrial large-scale production. The use of cheap, readily available starting materials and simple post-processing (filtration and chromatography) supports scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we are committed to translating cutting-edge academic research into tangible commercial solutions for the global pharmaceutical and fine chemical industries. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods like the one described in CN111423381A can be successfully implemented on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch. Our dedication to excellence makes us a trusted partner for companies seeking high-purity pharmaceutical intermediates that meet the demanding standards of regulatory agencies worldwide.
We invite you to explore the potential of this advanced synthesis technology for your next drug discovery or process development project. By collaborating with us, you gain access to our deep technical expertise and flexible manufacturing capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your timeline to market. Let us be your strategic partner in navigating the complexities of modern chemical synthesis and securing a reliable supply of critical building blocks.
