Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Commercial Scale-Up
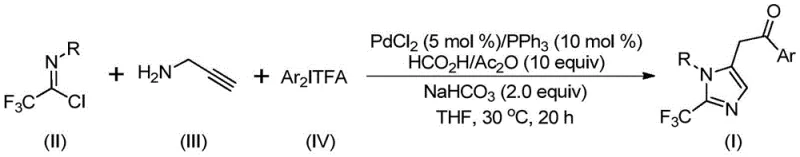
The pharmaceutical and agrochemical industries continuously demand robust synthetic routes for fluorinated heterocycles, particularly those incorporating the trifluoromethyl group due to its profound impact on metabolic stability and lipophilicity. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical limitations in existing literature. This technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. Unlike conventional approaches that often struggle with harsh conditions or unstable reagents, this innovation operates under mild conditions at 30°C, achieving high reaction efficiency and exceptional substrate compatibility. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this methodology represents a significant leap forward in process safety and cost-effectiveness, enabling the design of diversified imidazole derivatives essential for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on synthons like trifluorodiazoethane. While effective in certain contexts, these traditional reagents pose severe safety hazards due to their explosive nature and thermal instability, creating substantial bottlenecks in commercial scale-up of complex pharmaceutical intermediates. Furthermore, alternative methods using trifluoroethylimide acid halides have not been widely explored or optimized, leaving a gap in practical, scalable applications for industrial manufacturing. The reliance on dangerous diazo compounds necessitates specialized equipment and rigorous safety protocols, drastically inflating operational costs and extending lead times for high-purity API intermediates. Additionally, many existing protocols suffer from narrow substrate scope, failing to tolerate diverse functional groups required for the synthesis of advanced medicinal chemistry candidates, thereby limiting their utility in generating diverse chemical libraries for biological screening.
The Novel Approach
The patented methodology circumvents these challenges by employing a sophisticated palladium-catalyzed carbonylation series reaction that merges trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts in a single pot. This novel approach eliminates the need for hazardous diazo reagents, replacing them with stable, commercially accessible precursors that simplify supply chain logistics and reduce raw material costs. The reaction proceeds efficiently in common aprotic solvents like tetrahydrofuran (THF) at a mild temperature of 30°C, demonstrating remarkable functional group tolerance across a wide range of aryl substituents including methyl, tert-butyl, halogens, and nitro groups. By integrating a carbon monoxide surrogate system generated in situ from formic acid and acetic anhydride, the process achieves high atom economy and minimizes the need for high-pressure gas handling infrastructure. This streamlined workflow not only enhances operator safety but also facilitates the rapid synthesis of varied 1,5-disubstituted imidazole compounds, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a complex yet elegant sequence of organometallic steps initiated by base-promoted intermolecular carbon-nitrogen bond formation. Initially, the reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine intermediate, which subsequently undergoes isomerization to set the stage for cyclization. The palladium catalyst then mediates the aminopalladation of the alkyne moiety, forming a key alkenyl palladium species that isomerizes further into a more stable alkyl palladium intermediate. This critical intermediate captures carbon monoxide released from the formic acid/acetic anhydride mixture, undergoing carbonylation to yield an acyl palladium complex. The cycle concludes with the oxidative addition of the diaryliodonium salt to generate a high-valent tetravalent palladium species, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst. Understanding this detailed catalytic cycle is crucial for R&D teams aiming to optimize reaction parameters for specific substrate classes and ensure consistent impurity profiles during production.
Impurity control in this system is inherently managed by the high selectivity of the palladium catalyst and the mild reaction conditions which suppress side reactions common in high-temperature processes. The use of specific ligands like triphenylphosphine alongside palladium chloride ensures precise control over the coordination sphere of the metal center, minimizing the formation of homocoupling byproducts or unreacted starting materials. Furthermore, the compatibility of the reaction with various electron-withdrawing and electron-donating groups on the aryl rings of both the imidoyl chloride and the iodonium salt allows for the predictable synthesis of targeted analogs without extensive purification burdens. This level of mechanistic understanding supports the development of robust quality control strategies, ensuring that the resulting high-purity OLED material or pharmaceutical intermediate meets stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and mixing protocols outlined in the patent to maximize yield and minimize waste. The process begins with the careful preparation of the reaction mixture in a Schlenk tube or equivalent reactor, ensuring an inert atmosphere to protect the palladium catalyst from oxidation. Detailed standard operating procedures regarding the addition order of reagents, specifically the timing of the CO surrogate addition relative to the catalyst activation, are critical for reproducibility. For a comprehensive guide on the exact molar ratios, solvent volumes, and workup procedures including silica gel treatment and column chromatography conditions, please refer to the standardized synthesis steps provided below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid mixture, trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt in an organic solvent like THF.
- Stir the reaction mixture at 30°C for 16 to 24 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers transformative benefits by shifting the supply chain away from volatile, high-cost diazo reagents toward stable, commodity-grade chemicals. The reliance on trifluoroethylimidoyl chloride and diaryliodonium salts, which are derived from widely available aromatic amines and boronic acids, ensures a resilient supply base that is less susceptible to market fluctuations or geopolitical disruptions. This stability is paramount for supply chain heads responsible for maintaining continuous production lines for critical active pharmaceutical ingredients. Moreover, the simplified post-processing workflow, which involves standard filtration and chromatography rather than complex distillation or cryogenic separations, significantly reduces downstream processing costs and energy consumption. These factors collectively contribute to a more sustainable and economically viable manufacturing model that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous specialty reagents like trifluorodiazoethane directly lowers the bill of materials, while the use of inexpensive palladium chloride and triphenylphosphine keeps catalyst costs manageable. The mild reaction temperature of 30°C drastically reduces energy expenditure compared to high-temperature reflux conditions typically required for heterocycle synthesis. Additionally, the high yields reported across diverse substrates minimize raw material waste and reduce the burden on waste treatment facilities, leading to substantial overall cost savings in the production of fluorinated intermediates.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are commercially available off-the-shelf or easily synthesized from bulk commodities, manufacturers can secure long-term supply contracts with multiple vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling production facilities to respond rapidly to changes in demand without compromising product quality. This reliability is essential for maintaining just-in-time inventory levels and ensuring uninterrupted delivery of key building blocks to downstream formulation partners.
- Scalability and Environmental Compliance: The protocol has been successfully demonstrated at the gram level with clear pathways for expansion to kilogram and metric ton scales, supported by the use of standard organic solvents like THF that are well-understood in industrial settings. The avoidance of high-pressure carbon monoxide gas cylinders in favor of liquid surrogates simplifies facility requirements and enhances workplace safety compliance. Furthermore, the efficient atom economy and reduced generation of hazardous byproducts facilitate easier adherence to increasingly strict environmental regulations regarding solvent emissions and chemical waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, safety profiles, and scalability potential. Understanding these nuances helps technical teams make informed decisions about adopting this technology for their specific project needs.
Q: What are the key safety advantages of this new synthesis method compared to traditional routes?
A: Traditional methods often rely on hazardous trifluorodiazoethane. This patented process utilizes stable trifluoroethylimidoyl chlorides and diaryliodonium salts, significantly reducing explosion risks and handling hazards associated with diazo compounds.
Q: What yields can be expected for diverse substrates using this palladium-catalyzed protocol?
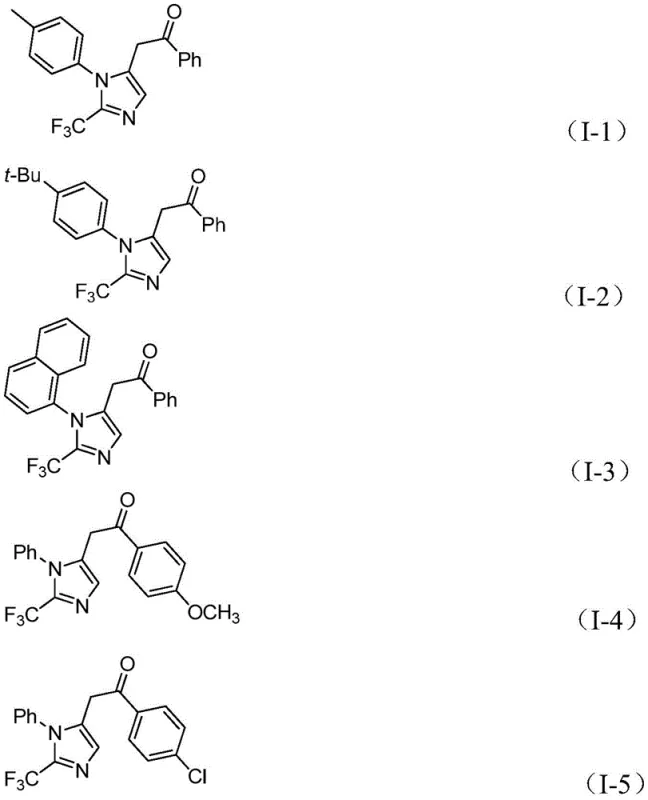
A: The patent demonstrates excellent substrate compatibility with isolated yields ranging from 65% to 97% across various electron-donating and electron-withdrawing groups on both the amine and iodonium salt components.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method operates at mild temperatures (30°C) using commercially available catalysts and solvents. The patent explicitly states the process can be expanded to gram-level synthesis, indicating strong potential for metric ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like this palladium-catalyzed carbonylation to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-trifluoromethyl imidazole intermediate meets the highest standards required for global pharmaceutical applications. Our commitment to quality and consistency makes us the preferred partner for companies seeking to innovate their drug discovery pipelines with reliable, high-performance building blocks.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your long-term supply chain goals with precision and expertise.
