Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to anti-HIV drugs such as Delavirdine. As illustrated in the diverse pharmacological landscape shown below, the ability to efficiently functionalize the indole nitrogen is critical for drug discovery and process development. Patent CN112898192B introduces a groundbreaking preparation method for N-acyl indole compounds that addresses long-standing challenges in synthetic efficiency and operational safety. This technology leverages a sophisticated palladium-catalyzed carbonylation cyclization strategy, transforming simple 2-alkynyl anilines and aryl iodides into complex N-acyl indoles in a streamlined, one-pot procedure. For R&D directors and process chemists, this represents a significant leap forward, offering a robust pathway to access high-value pharmaceutical intermediates with exceptional purity and yield profiles.
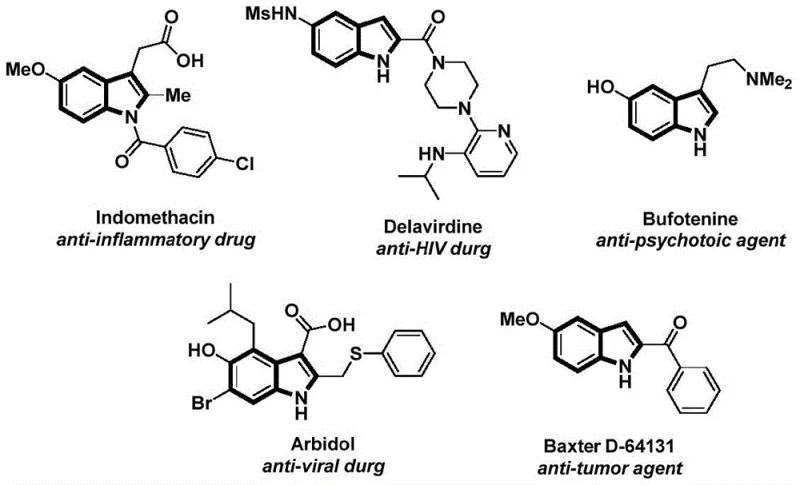
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of N-acyl indoles has often relied on multi-step sequences involving the protection and deprotection of the indole nitrogen, or the use of harsh acylating agents that generate significant waste streams. Conventional carbonylation reactions frequently require the handling of toxic carbon monoxide gas under high pressure, posing severe safety risks and necessitating specialized equipment that increases capital expenditure. Furthermore, older methodologies often suffer from poor atom economy and limited substrate tolerance, particularly when sensitive functional groups are present on the aromatic rings. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and inflated costs for high-purity intermediates. The reliance on stoichiometric amounts of hazardous reagents also complicates waste disposal and environmental compliance, making these legacy processes increasingly untenable for modern green chemistry standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112898192B utilizes a solid carbon monoxide substitute, 1,3,5-tricarboxylic acid phenol ester (TFBen), which safely releases CO in situ under mild thermal conditions. This innovation eliminates the need for high-pressure gas cylinders, drastically simplifying the reactor setup and enhancing operational safety for plant personnel. The reaction proceeds through a tandem sequence where palladium catalysis facilitates both the carbonylation and the subsequent cyclization, effectively merging two distinct transformations into a single operational unit. As depicted in the general reaction scheme below, this approach allows for the direct conversion of readily available 2-alkynyl anilines and aryl iodides into the target N-acyl indoles with remarkable efficiency. The process operates at a moderate temperature of 60°C in acetonitrile, demonstrating excellent compatibility with a wide range of substituents including halogens, alkoxy groups, and alkyl chains.
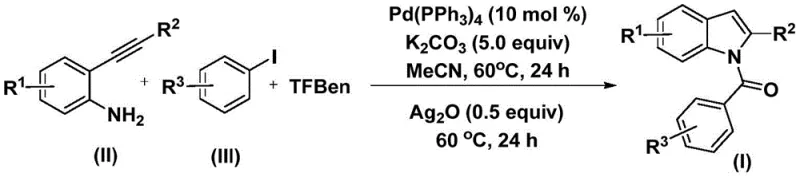
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, initiating with the oxidative addition of the aryl iodide to the active Pd(0) species generated from the tetrakis(triphenylphosphine)palladium precursor. This forms a reactive aryl-palladium(II) intermediate, which subsequently undergoes migratory insertion of carbon monoxide derived from the thermal decomposition of TFBen. The resulting acyl-palladium species is then intercepted by the amine functionality of the 2-alkynyl aniline, forming an amide linkage through a reductive elimination or nucleophilic attack sequence. Crucially, the addition of silver oxide in the second stage serves a dual purpose: it acts as an oxidant to regenerate the active palladium catalyst and promotes the intramolecular cyclization of the alkyne moiety to close the indole ring. This carefully orchestrated sequence ensures high conversion rates and minimizes the formation of side products, providing a clean reaction profile that simplifies downstream purification.
From an impurity control perspective, the use of specific ligands and the controlled release of CO are pivotal in suppressing homocoupling of the aryl iodide or polymerization of the alkyne. The reaction conditions specified in the patent, such as the precise molar ratio of potassium carbonate (5.0 equivalents) and the staged addition of silver oxide (0.5 equivalents), are optimized to maintain the catalytic cycle's integrity over the 48-hour duration. Experimental data indicates that varying the electronic nature of the substituents on the aryl rings has a minimal impact on the overall yield, with electron-donating groups like methoxy and electron-withdrawing groups like fluorine both tolerated well. This robustness suggests that the catalytic system is highly resilient, capable of handling the structural diversity required in the synthesis of complex API intermediates without requiring extensive re-optimization for each new substrate.
How to Synthesize N-Acyl Indole Compounds Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to the specific protocol outlined in the patent to ensure maximum yield and reproducibility. The process begins with the careful weighing of reagents, specifically maintaining the stoichiometric balance between the palladium catalyst, the base, and the CO source. The reaction is typically conducted in a Schlenk tube or a sealed vessel under an inert atmosphere to prevent catalyst deactivation by oxygen. Following the initial heating phase, the timely addition of silver oxide is critical to trigger the cyclization step, and the mixture must be maintained at 60°C for the full duration to drive the reaction to completion. Detailed standardized operating procedures for this synthesis are provided in the section below.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial coupling and carbonylation.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant reduction of raw material costs, as the starting materials—2-alkynyl anilines and aryl iodides—are commodity chemicals available from multiple global suppliers, reducing dependency on single-source vendors. The elimination of high-pressure carbon monoxide infrastructure not only lowers capital investment but also reduces insurance premiums and regulatory compliance costs associated with handling toxic gases. Furthermore, the high reaction efficiency and yields reported, reaching up to 82% in optimized examples, translate directly into improved material throughput and reduced waste disposal fees. This efficiency gain allows for a more predictable production schedule, minimizing the risk of stockouts for critical intermediates.
- Cost Reduction in Manufacturing: The replacement of gaseous CO with a solid surrogate like TFBen removes the need for specialized high-pressure reactors and gas handling systems, leading to drastic capital expenditure savings. Additionally, the use of a common solvent like acetonitrile and commercially available catalysts ensures that the variable costs per kilogram of product remain competitive. The simplified work-up procedure, involving filtration and standard column chromatography, further reduces labor and processing time compared to traditional multi-step syntheses.
- Enhanced Supply Chain Reliability: By utilizing widely available building blocks, manufacturers can diversify their supplier base, mitigating the risk of supply disruptions caused by geopolitical issues or production outages at specific facilities. The robustness of the reaction conditions means that production can be scaled up from gram to ton scale with minimal process adjustments, ensuring a continuous and reliable flow of materials to downstream API manufacturing sites. This stability is crucial for maintaining just-in-time inventory levels and meeting strict delivery deadlines.
- Scalability and Environmental Compliance: The mild reaction temperature of 60°C and the absence of hazardous gas emissions make this process inherently safer and more environmentally friendly. The reduced generation of toxic byproducts aligns with increasingly stringent environmental regulations, facilitating easier permitting for new production lines. The scalability is further supported by the homogeneous nature of the reaction mixture, which allows for efficient heat and mass transfer in larger reactors, ensuring consistent product quality across different batch sizes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acyl indole synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN112898192B, providing a reliable foundation for decision-making. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What are the key advantages of this N-acyl indole synthesis method?
A: The method described in patent CN112898192B offers high reaction efficiency, excellent substrate compatibility, and utilizes cheap, readily available starting materials like 2-alkynyl anilines and aryl iodides.
Q: What catalyst system is used for this carbonylation reaction?
A: The process employs a palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, along with potassium carbonate as a base and silver oxide as an oxidant/promoter.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild conditions (60°C) in common solvents like acetonitrile, uses stable solid reagents instead of toxic CO gas, and demonstrates high yields, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team of expert process chemists has extensively evaluated the methodology described in patent CN112898192B and confirmed its potential for robust commercial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to perform comprehensive impurity profiling, guaranteeing that every batch of N-acyl indole delivered adheres to the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your R&D timeline and your bottom line.
