Advanced Manufacturing of Drospirenone Intermediates via Streamlined Epoxide Condensation
Advanced Manufacturing of Drospirenone Intermediates via Streamlined Epoxide Condensation
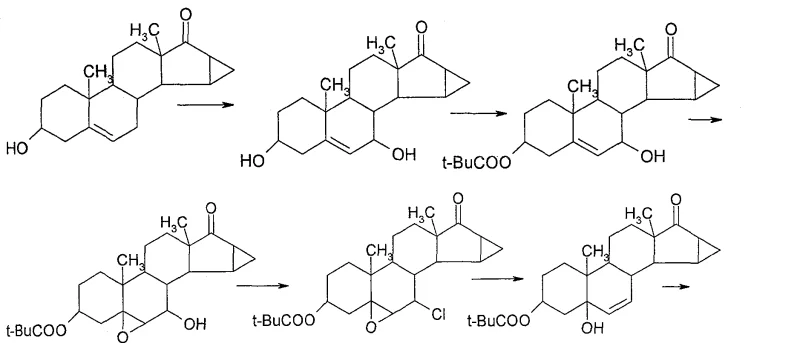
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of high-value steroidal active pharmaceutical ingredients (APIs), particularly for next-generation contraceptives like Drospirenone. Patent CN100480256C presents a significant technological advancement in this domain, detailing a novel method for synthesizing Drospirenone that addresses critical bottlenecks found in legacy manufacturing processes. By utilizing 3β,5-dihydroxy-6β,7β,15β,16β-dimethylene-5β-androstan-17-one as a strategic starting scaffold, this invention bypasses the notoriously difficult regioselective functionalization steps that have historically plagued production efficiency. The core innovation lies in the construction of the characteristic 17α-spirolactone ring through a sequence involving 17-position epoxidation, followed by nucleophilic ring-opening condensation, decarboxylation, and final oxidation. This approach not only simplifies the synthetic topology but also aligns perfectly with the stringent quality and cost requirements demanded by modern reliable drospirenone intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Drospirenone has been hindered by complex multi-step sequences that suffer from poor atom economy and challenging purification profiles. As illustrated in the prior art referenced within the patent documentation, early routes such as those described in EP51143 and DE19633685 attempted to introduce the 7-hydroxyl group at a late stage, resulting in significantly low yields due to steric hindrance and competing elimination reactions. Furthermore, alternative pathways like DE2652761 struggled with the specific introduction of the 6,7-cyclopropane ring, often leading to mixtures of isomers that required extensive and costly chromatographic separation. These conventional methodologies imposed severe constraints on cost reduction in steroidal API manufacturing, as the cumulative loss of material at each low-yielding step drastically inflated the cost of goods sold (COGS). The reliance on harsh conditions to force these transformations also raised safety concerns and complicated waste stream management, making these routes less attractive for commercial scale-up of complex steroid intermediates in a regulated GMP environment.
The Novel Approach
In stark contrast to the fragmented strategies of the past, the methodology disclosed in CN100480256C adopts a convergent strategy that leverages a highly functionalized starting material to minimize downstream processing. By commencing with a substrate that already possesses the critical 6β,7β,15β,16β-dimethylene cyclopropane architecture, the synthesis effectively decouples the difficult ring-formation chemistry from the lactone construction. The new route focuses on transforming the C17 ketone into a spiro-epoxide, which serves as an electrophilic handle for the subsequent attachment of the lactone precursor. This strategic pivot eliminates the need for aggressive conditions to install the cyclopropane ring, thereby preserving the integrity of the sensitive steroid backbone. The result is a streamlined process flow that enhances overall throughput and provides a more predictable impurity profile, which is essential for any high-purity drospirenone production campaign aiming for regulatory approval.
Mechanistic Insights into Epoxide-Mediated Lactone Construction
The heart of this synthetic innovation is the elegant utilization of sulfur ylide chemistry to generate a 17-spiro-epoxide intermediate (Compound II). In this transformation, dimethyl sulfoxide (DMSO) and potassium tert-butoxide generate a reactive sulfonium ylide in situ, which attacks the C17 carbonyl group of the starting androstan-17-one. This nucleophilic addition creates a betaine intermediate that collapses to form the strained three-membered oxirane ring. The formation of this epoxide is crucial because it activates the C17 position for nucleophilic attack while simultaneously establishing the stereochemical foundation for the 17α-configuration required in the final API. Following epoxidation, the process employs a classic malonic ester synthesis strategy where the epoxide ring is opened by the enolate of diethyl malonate under basic conditions (sodium ethoxide). This ring-opening event extends the carbon chain at C17, installing the necessary two-carbon unit that will eventually cyclize to form the lactone. The precision of this alkylation step ensures that the side chain is attached with the correct regiochemistry, avoiding the formation of unwanted regioisomers that often complicate direct alkylation of ketones.
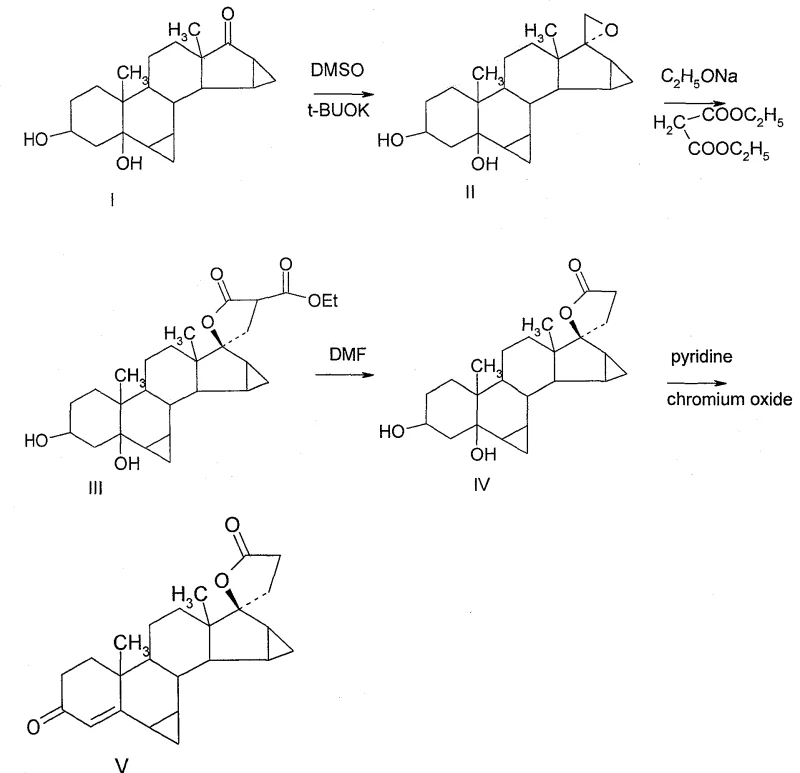
Following the condensation step, the intermediate undergoes hydrolytic decarboxylation in a polar aprotic solvent system (DMF/Water), which removes the extraneous carboxyl group and sets the stage for the final ring closure. The terminal step involves the oxidation of the secondary alcohol at the C3 position and the concurrent cyclization of the side chain ester to form the gamma-lactone ring. This is achieved using chromium trioxide in a pyridine-water mixture, a variation of the Jones oxidation tailored for sensitive substrates. The mechanism here likely involves the initial oxidation of the C3 alcohol to a ketone, followed by acid-catalyzed or thermal lactonization of the pendant ester chain onto the C17 position. Throughout this sequence, impurity control is maintained through rigorous monitoring via Thin Layer Chromatography (TLC), allowing operators to quench reactions precisely at completion to prevent over-oxidation or degradation of the cyclopropane moieties. This level of mechanistic control is vital for ensuring the reducing lead time for high-purity drospirenone intermediates by minimizing the need for repetitive recrystallizations.
How to Synthesize Drospirenone Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the exothermic epoxidation and the oxidative workup. The process begins with the generation of the sulfur ylide at elevated temperatures followed by cooling to sub-zero conditions to manage the addition to the steroid ketone. Subsequent steps involve standard reflux conditions for the condensation and decarboxylation, making them amenable to standard glass-lined reactor setups found in most fine chemical facilities. The detailed standardized operating procedures for each transformation, including specific solvent ratios and quenching protocols, are outlined below to ensure reproducibility and safety.
- Epoxidation of the 17-ketone using dimethyl sulfoxide and bromotrimethylsulfide under basic conditions to form the spiro-epoxide intermediate.
- Ring-opening condensation with diethyl malonate in the presence of sodium ethoxide to extend the carbon chain at the 17-position.
- Hydrolytic decarboxylation in DMF followed by oxidative cyclization using chromium trioxide to form the final gamma-lactone ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers tangible benefits that directly impact the bottom line and operational reliability. By shifting the complexity to the starting material selection rather than the processing steps, manufacturers can achieve a more consistent output quality, which reduces the risk of batch failures and supply disruptions. The use of commodity chemicals such as DMSO, diethyl malonate, and sodium ethoxide ensures that raw material sourcing remains stable and unaffected by the volatility often seen with specialized organometallic reagents. Furthermore, the workup procedures described, which rely heavily on water precipitation and filtration rather than complex extractive separations, significantly reduce solvent consumption and waste disposal costs. This alignment with green chemistry principles not only lowers environmental compliance burdens but also accelerates the throughput of the manufacturing suite, allowing for faster turnaround times on large-volume orders.
- Cost Reduction in Manufacturing: The elimination of low-yielding regioselective steps found in prior art translates directly into higher overall process mass intensity (PMI) efficiency. By avoiding the need for extensive chromatographic purification to separate cyclopropane isomers, the process drastically reduces the consumption of expensive silica gel and organic solvents. Additionally, the high specificity of the epoxide ring-opening reaction minimizes the formation of byproducts, meaning that more of the input raw material is converted into saleable product. This inherent efficiency allows for a substantial reduction in the cost per kilogram of the final intermediate, providing a competitive pricing advantage in the global market without compromising on margin.
- Enhanced Supply Chain Reliability: The robustness of this chemical sequence ensures that production schedules can be met with greater certainty. Since the reaction conditions are relatively mild and do not require exotic catalysts or ultra-low temperature cryogenic setups (beyond standard cooling), the risk of equipment-related delays is minimized. The starting material, derived from established precedents like USP4435327, is accessible through a mature supply network, mitigating the risk of single-source bottlenecks. This stability is crucial for long-term supply agreements where continuity of supply is paramount for downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing unit operations that are easily transferred from pilot plant to full commercial scale. The reliance on aqueous workups for product isolation simplifies the handling of mother liquors and facilitates solvent recovery systems. Moreover, by reducing the number of synthetic steps and avoiding heavy metal catalysts in the early stages, the overall environmental footprint of the manufacturing process is significantly lowered. This makes the route highly attractive for companies aiming to meet increasingly strict corporate sustainability goals and regulatory emissions standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential partners.
Q: What are the primary advantages of this synthesis route over prior art methods?
A: This method overcomes the low yield and poor specificity associated with introducing the 6,7-cyclopropane moiety in earlier routes by utilizing a pre-functionalized starting material, thereby streamlining the process to focus on efficient lactone ring construction.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrialization due to simple operational steps, the use of common reagents like DMSO and diethyl malonate, and straightforward workup procedures involving water precipitation and filtration.
Q: How is product purity maintained during the oxidation step?
A: Purity is controlled through careful temperature management during the chromium trioxide oxidation and rigorous purification via column chromatography, ensuring the removal of pyridine residues and side products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Drospirenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the pharmaceutical supply chain. Our technical team has thoroughly analyzed the methodology presented in CN100480256C and validated its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with international GMP standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Drospirenone intermediate we deliver meets the highest quality benchmarks required for API synthesis.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in steroidal chemistry and process optimization, we can help you secure a stable supply of high-quality intermediates while optimizing your total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
