Scalable Green Synthesis of Diaryl Sulfones Using Iron-Based Ionic Liquids for Pharmaceutical Manufacturing
Scalable Green Synthesis of Diaryl Sulfones Using Iron-Based Ionic Liquids for Pharmaceutical Manufacturing
The global demand for high-purity diaryl sulfone derivatives continues to surge, driven by their critical role as structural scaffolds in active pharmaceutical ingredients (APIs) and agrochemical formulations. Patent CN113185434A introduces a transformative green synthesis methodology that addresses longstanding inefficiencies in constructing the sulfone bridge. This innovation leverages a three-component one-pot reaction involving beta-naphthol, aryltriazene, and DABCO(SO2)2, all catalyzed by a novel iron-containing Lewis acidic ionic liquid. By operating under mild thermal conditions at 90°C without the need for inert gas protection or external acidic additives, this process represents a significant leap forward in sustainable chemical manufacturing. For R&D directors and procurement specialists, this technology offers a compelling pathway to reduce complexity while maintaining rigorous quality standards essential for regulatory compliance in the pharmaceutical sector.
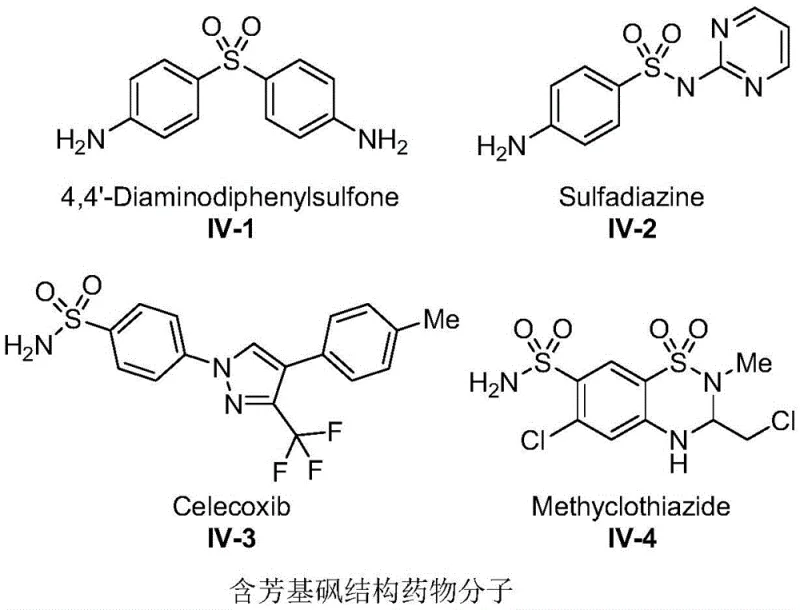
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for diaryl sulfones have historically been plagued by significant operational hazards and economic inefficiencies that burden modern supply chains. Conventional methodologies often rely on the direct use of gaseous sulfur dioxide, a toxic and corrosive reagent that necessitates specialized high-pressure containment systems and rigorous safety protocols, thereby inflating capital expenditure and operational overhead. Furthermore, many established protocols require stoichiometric amounts of strong acid promoters or expensive transition metal catalysts to activate the aryl source, leading to substantial waste generation and complicated downstream purification processes. The reliance on harsh reaction conditions often results in poor regioselectivity, generating difficult-to-remove impurities that compromise the purity profile of the final intermediate. These factors collectively contribute to extended lead times and increased environmental liability, making conventional sulfone synthesis increasingly untenable for cost-sensitive, large-scale commercial production environments.
The Novel Approach
The methodology disclosed in the patent data circumvents these historical bottlenecks by employing a solid sulfur dioxide surrogate, DABCO(SO2)2, which eliminates the handling risks associated with gaseous reagents. This innovative approach utilizes aryltriazene as a stable and readily available aryl source, which undergoes efficient activation in the presence of the iron-containing ionic liquid catalyst. The reaction proceeds smoothly in a one-pot fashion, merging the sulfonylation and coupling steps into a single operational unit, which drastically simplifies the workflow and reduces solvent consumption. By avoiding the need for external additives or inert atmosphere protection, the process enhances operational simplicity and reduces the dependency on specialized infrastructure. This streamlined protocol not only improves the overall atom economy but also facilitates easier scale-up, providing a robust foundation for the reliable supply of high-purity pharmaceutical intermediates required by multinational healthcare corporations.
Mechanistic Insights into FeCl4-Based Lewis Acid Catalysis
The core of this technological advancement lies in the unique properties of the iron-containing Lewis acidic ionic liquid, specifically variants such as IL 4 depicted in the structural analysis. This catalyst functions as a potent Lewis acid that effectively activates the aryltriazene species, facilitating the cleavage of the nitrogen-nitrogen bond and the subsequent generation of the reactive aryl diazonium intermediate in situ. The iron center within the ionic liquid matrix coordinates with the sulfur dioxide source, promoting the formation of the sulfonyl radical or cationic species necessary for the electrophilic aromatic substitution. This dual activation mechanism ensures high regioselectivity, directing the sulfonyl group specifically to the C1 position of the beta-naphthol ring system, which is crucial for maintaining the biological activity of downstream drug candidates. The ionic liquid environment stabilizes these reactive intermediates, preventing side reactions and decomposition pathways that typically lower yields in traditional solvent systems.
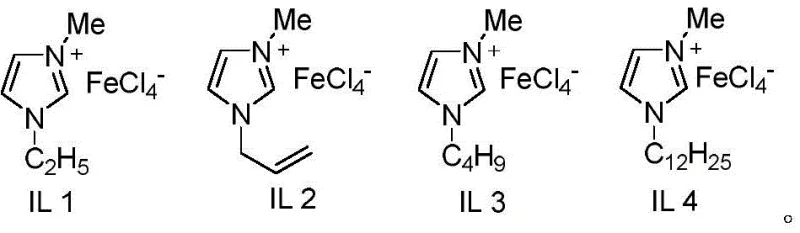
From an impurity control perspective, the use of this specific catalytic system minimizes the formation of polysulfonated byproducts and homocoupling artifacts that frequently contaminate sulfone syntheses. The mild acidity of the ionic liquid prevents the over-protonation of the naphthol substrate, which can lead to polymerization or tar formation under stronger acidic conditions. Additionally, the non-volatile nature of the ionic liquid allows for potential recycling strategies, further reducing the environmental footprint of the manufacturing process. For quality assurance teams, this means a cleaner crude reaction profile, which translates to fewer purification cycles and higher overall recovery of the target material. The mechanistic robustness of this system ensures consistent batch-to-batch reproducibility, a critical parameter for validating commercial manufacturing processes under Good Manufacturing Practice (GMP) guidelines.
How to Synthesize 1-Arylsulfonyl-2-naphthol Efficiently
Implementing this synthesis route requires precise adherence to the optimized reaction parameters identified during the patent development phase to ensure maximum yield and purity. The process involves the sequential addition of the iron-containing ionic liquid catalyst, beta-naphthol substrate, aryltriazene derivative, and the DABCO(SO2)2 sulfur source into a reaction vessel containing acetonitrile as the solvent. The mixture is then subjected to thermal heating at 90°C for a duration of approximately 14 hours to drive the reaction to completion. Following the reaction period, a straightforward aqueous workup involving extraction with ethyl acetate and drying over anhydrous magnesium sulfate isolates the crude product. Final purification is achieved through standard column chromatography techniques, yielding the target diaryl sulfone compound with high structural integrity. Detailed standardized synthesis steps are provided in the guide below.
- Charge a sealed tube with iron-containing ionic liquid IL 4 (20 mol%), beta-naphthol, aryltriazene, and DABCO(SO2)2 in acetonitrile solvent.
- Seal the reaction vessel and stir the mixture at 90°C for 14 hours to facilitate the sulfonylation reaction at the C1 position.
- Quench the reaction with water, extract with ethyl acetate, dry the organic phase, and purify via column chromatography to isolate the target diaryl sulfone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. The shift from hazardous gaseous reagents to stable solid surrogates significantly de-risks the logistics of raw material storage and transportation, ensuring uninterrupted production schedules even in regions with stringent safety regulations. The elimination of expensive noble metal catalysts in favor of abundant iron-based ionic liquids directly impacts the bill of materials, driving down the variable costs associated with each production batch. Furthermore, the simplified one-pot nature of the reaction reduces the requirement for multiple reactor vessels and extensive intermediate isolation steps, effectively increasing the throughput capacity of existing manufacturing facilities without the need for major capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly and hazardous reagents with commodity chemicals that are widely available in the global market. By utilizing DABCO(SO2)2 instead of pressurized sulfur dioxide cylinders, manufacturers eliminate the need for specialized gas handling equipment and the associated maintenance costs, leading to substantial operational savings. The catalyst, being an iron-based ionic liquid, is significantly cheaper than palladium or rhodium alternatives often used in cross-coupling reactions, and its stability allows for potential reuse, further amortizing the catalyst cost over multiple batches. Additionally, the high selectivity of the reaction minimizes waste disposal costs, as fewer byproducts are generated that require treatment, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of raw materials that possess high commercial availability and stable pricing structures, reducing vulnerability to market fluctuations. Aryltriazenes and beta-naphthol are established industrial commodities with multiple qualified suppliers worldwide, ensuring that production is not bottlenecked by single-source dependencies. The robustness of the reaction conditions, which do not require sensitive inert gas atmospheres or ultra-low temperatures, means that the process can be reliably transferred to contract manufacturing organizations (CMOs) in various geographic locations without compromising quality. This flexibility allows for a diversified supply base, mitigating risks associated with geopolitical instability or regional logistical disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the inherent safety and simplicity of the reaction design, which avoids exothermic runaway risks associated with gaseous reagents. The green chemistry principles embedded in this method, such as the use of a recyclable catalyst and the generation of minimal hazardous waste, align perfectly with increasingly strict environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and permitting timelines for new manufacturing lines, accelerating time-to-market for new drug candidates. The ability to operate at atmospheric pressure and moderate temperatures also lowers energy consumption, contributing to a reduced carbon footprint for the overall manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diaryl sulfone synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the advantages of using DABCO(SO2)2 over gaseous SO2 in this synthesis?
A: DABCO(SO2)2 serves as a solid, easy-to-handle sulfur dioxide surrogate, eliminating the safety hazards and specialized equipment required for handling toxic gaseous SO2, thereby simplifying the operational protocol and enhancing workplace safety.
Q: Why is the iron-containing ionic liquid preferred as a catalyst for this transformation?
A: The iron-containing Lewis acidic ionic liquid acts as a dual-function catalyst and solvent medium that is inexpensive, structurally stable, and environmentally benign, offering superior catalytic activity without the need for additional acidic promoters or inert gas protection.
Q: Can this method be applied to substrates with diverse electronic properties?
A: Yes, the patent data demonstrates broad substrate tolerance, successfully synthesizing derivatives with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups, indicating robust versatility for complex intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of this iron-catalyzed sulfonylation method and is fully prepared to support your project from gram-scale optimization to multi-ton commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diaryl sulfone intermediate delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative technology for your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green synthesis route can optimize your overall manufacturing budget. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-quality intermediates backed by deep technical expertise and a commitment to sustainable chemical manufacturing practices.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
