Advanced Pd-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial API Manufacturing
Advanced Pd-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial API Manufacturing
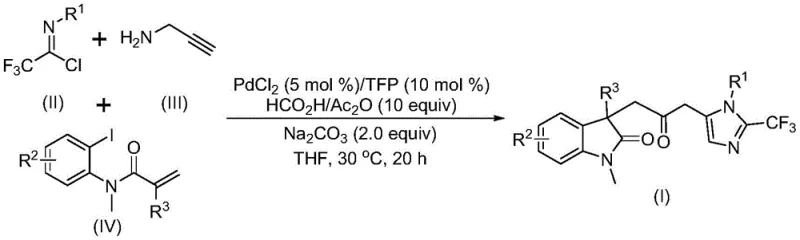
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, which serve as the core backbone for numerous bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a highly efficient, palladium-catalyzed multi-component reaction for synthesizing carbonyl-bridged biheterocyclic compounds. This technology addresses critical bottlenecks in the production of indolinone-imidazole hybrids, a class of structures renowned for their broad-spectrum biological activities and presence in various drug candidates. By leveraging a sophisticated cascade sequence that integrates Heck coupling, carbonylation, and cyclization into a single operational step, this invention offers a streamlined pathway for generating high-value pharmaceutical intermediates. The methodology is particularly notable for its ability to incorporate trifluoromethyl groups, a modification often crucial for enhancing the metabolic stability and lipophilicity of drug molecules, thereby providing a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in catalog diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems containing both indolinone and imidazole motifs has been fraught with synthetic challenges that hinder large-scale adoption. Traditional approaches typically rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that often suffers from poor atom economy and requires harsh reaction conditions that are incompatible with sensitive functional groups. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles have been employed, yet these methods frequently necessitate the use of stoichiometric oxidants and generate substantial amounts of chemical waste. Furthermore, conventional carbonylation strategies to install the bridging carbonyl moiety traditionally depend on the use of toxic carbon monoxide gas under high pressure, posing severe safety risks and requiring specialized, expensive reactor infrastructure that many contract manufacturing organizations lack. These limitations collectively result in prolonged development timelines, elevated production costs, and significant supply chain vulnerabilities for manufacturers seeking cost reduction in API manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN115353511A introduces a transformative one-pot, three-component coupling strategy that dramatically simplifies the synthetic landscape. This novel approach utilizes readily available starting materials—specifically trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamides—to construct the complex biheterocyclic framework in a single vessel. The reaction operates under remarkably mild conditions, proceeding efficiently at just 30°C, which minimizes energy consumption and thermal degradation of sensitive intermediates. Crucially, the process circumvents the need for gaseous carbon monoxide by employing an in-situ CO source generated from a mixture of formic acid and acetic anhydride, thereby enhancing operational safety and reducing regulatory burdens associated with hazardous gas handling. This streamlined protocol not only improves overall reaction efficiency but also expands the accessible chemical space, allowing for the rapid generation of diverse libraries of trifluoromethyl-substituted biheterocycles essential for modern drug discovery programs.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this transformation hinges on a meticulously orchestrated palladium-catalyzed cascade mechanism that seamlessly integrates multiple bond-forming events. The catalytic cycle is initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This species subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, forming a five-membered palladacycle that establishes the indolinone core. Following this cyclization, the pivotal carbonylation step occurs, where the alkyl-palladium intermediate inserts carbon monoxide—released from the formic acid/acetic anhydride additive—to yield an acyl-palladium complex. Concurrently, in a parallel pathway facilitated by the base, the trifluoroethylimidoyl chloride reacts with propargylamine to form a trifluoroacetamidine intermediate, which then undergoes isomerization. The final stage involves the nucleophilic attack of this amidine species onto the activated acyl-palladium center, followed by reductive elimination to release the final carbonyl-bridged product and regenerate the active palladium catalyst, ensuring high turnover numbers and catalytic efficiency.
From an impurity control perspective, the mechanistic design inherently suppresses common side reactions that plague multi-step syntheses. The use of tris(2-furyl)phosphine (TFP) as a ligand is critical, as it stabilizes the palladium center against aggregation while promoting the specific reactivity required for the carbonylation step over competing beta-hydride elimination pathways. The mild basicity of sodium carbonate ensures the deprotonation necessary for amidine formation without inducing hydrolysis of the sensitive imidoyl chloride or the newly formed amide bonds. Furthermore, the low reaction temperature of 30°C kinetically favors the desired cascade sequence over thermal decomposition or polymerization of the acrylamide components. This precise control over reaction parameters results in a clean crude reaction profile, significantly reducing the burden on downstream purification processes and enabling the isolation of high-purity pharmaceutical intermediates with minimal chromatographic effort.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding reagent quality and atmospheric control to ensure reproducibility and safety. The process begins with the preparation of the catalytic system, where palladium chloride and the TFP ligand are combined with sodium carbonate and the CO-generating mixture in an anhydrous organic solvent, preferably tetrahydrofuran (THF), which has been shown to provide superior solubility and reaction rates compared to acetonitrile or dioxane. Once the catalytic mixture is homogenized, the three key substrates—trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative—are introduced in precise molar ratios, typically maintaining a slight excess of the amine and acrylamide components to drive the equilibrium towards completion. The detailed standardized synthesis steps for this procedure are outlined below:
- Combine palladium chloride (5 mol%), TFP ligand (10 mol%), sodium carbonate (2.0 equiv), and the formic acid/acetic anhydride CO source in THF solvent.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, then filter and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to optimize sourcing strategies and mitigate production risks associated with complex heterocyclic intermediates. The shift from multi-step, hazardous processes to a single-pot, mild-temperature reaction fundamentally alters the cost structure of manufacturing these valuable scaffolds. By eliminating the need for high-pressure reactors and toxic gas infrastructure, capital expenditure requirements for production facilities are drastically reduced, allowing for more flexible deployment of manufacturing capacity. Moreover, the use of commodity chemicals such as propargylamine and simple acrylamide derivatives as starting materials ensures a stable and resilient supply chain, insulating production schedules from the volatility often seen with exotic or highly specialized reagents. This robustness translates directly into enhanced supply chain reliability and the ability to meet tight delivery windows for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the consolidation of multiple synthetic steps into a single operation, which significantly lowers labor costs, solvent consumption, and waste disposal fees. The avoidance of expensive transition metal scavengers, often required to remove residual palladium from products synthesized via traditional cross-coupling, further contributes to substantial cost savings. Additionally, the high atom economy of the multi-component reaction ensures that a greater proportion of raw material mass is incorporated into the final product, minimizing waste generation and maximizing yield per kilogram of input. These factors combine to create a highly cost-effective manufacturing route that offers significant margin improvements for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commercially available, off-the-shelf reagents eliminates the long lead times associated with custom synthesis of specialized building blocks. Since the reaction tolerates a wide range of functional groups, including halogens, nitro groups, and trifluoromethyl moieties, manufacturers can source diverse substrates from multiple global vendors without compromising reaction performance. This flexibility reduces the risk of supply disruptions caused by single-source dependencies. Furthermore, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, facilitating rapid technology transfer and scale-up without the need for specialized equipment procurement, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process has been validated at the gram scale with excellent yields, demonstrating a clear path towards kilogram and ton-scale production. The replacement of gaseous carbon monoxide with a liquid CO source simplifies safety protocols and reduces the environmental footprint of the operation by eliminating the risk of gas leaks. The use of THF as a solvent, which is easily recoverable and recyclable, aligns with green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. The simplified workup procedure, involving basic filtration and standard chromatography, minimizes the generation of aqueous waste streams, making this technology an environmentally sustainable choice for the long-term production of advanced fine chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method, providing clarity for R&D teams evaluating its feasibility for specific projects. These answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for process development planning. Understanding these nuances is essential for optimizing reaction parameters and predicting outcomes when applying this chemistry to novel substrate classes.
Q: What are the optimal reaction conditions for this carbonylation cascade?
A: The patent specifies using THF as the preferred solvent at a mild temperature of 30°C for 12 to 20 hours, utilizing PdCl2/TFP as the catalytic system.
Q: Does this method require toxic carbon monoxide gas?
A: No, the process utilizes a safe ex-situ CO source generated from a mixture of formic acid and acetic anhydride, eliminating the need for high-pressure CO cylinders.
Q: What is the substrate scope regarding the R1 and R3 groups?
A: The method demonstrates excellent compatibility with various substituents including alkyl, halogen (Cl, F, Br), methoxy, nitro, and trifluoromethyl groups on both aryl and benzyl moieties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multi-component technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control and safe handling of reactive intermediates, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug discovery pipelines with reliable quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can be tailored to your specific molecular targets. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this methodology for your specific portfolio. We encourage you to contact our technical procurement team today to request specific COA data for our existing catalog of biheterocyclic compounds or to discuss route feasibility assessments for your proprietary molecules, ensuring a partnership built on technical excellence and mutual growth.
