Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, due to their prevalence in bioactive molecular frameworks. Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current manufacturing processes. This technology leverages a metal-free oxidative cyclization strategy that eliminates the need for expensive transition metal catalysts and严苛 anhydrous conditions. By utilizing readily available starting materials such as 2-methylquinoline and trifluoroacetimidohydrazide, the process achieves exceptional conversion rates under mild thermal conditions. For R&D directors and procurement managers alike, this represents a paradigm shift towards more sustainable and cost-effective pharmaceutical intermediate production. The ability to synthesize diverse derivatives through simple substrate design further enhances the utility of this method in drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and operational complexity. Traditional protocols often rely on quinoline-2-carboxylic acid as a primary raw material, necessitating a tedious five-step reaction sequence to reach the final target. This multi-step approach not only accumulates impurities at each stage but also results in a dismal total yield of approximately 17%, rendering it economically unviable for large-scale applications. Furthermore, conventional methods frequently demand stringent reaction conditions, including strict anhydrous and oxygen-free environments, which significantly increase infrastructure costs and safety risks. The reliance on harsh reagents and complex purification steps creates a substantial barrier to entry for manufacturers aiming to produce these valuable heterocycles in bulk quantities.
The Novel Approach
In stark contrast, the methodology disclosed in CN113307790B offers a streamlined, one-pot solution that dramatically simplifies the synthetic landscape. By employing 2-methylquinoline and trifluoroacetimidohydrazide as direct precursors, the new route bypasses the need for pre-functionalized carboxylic acids. The core innovation lies in the use of tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP) to promote an efficient oxidative cyclization. This system operates effectively in polar aprotic solvents like DMSO at temperatures between 80°C and 100°C. The elimination of heavy metal catalysts not only reduces raw material costs but also simplifies downstream processing by removing the need for rigorous metal scavenging steps. This approach exemplifies how modern organic synthesis can achieve cost reduction in pharmaceutical intermediate manufacturing through intelligent catalyst design and process intensification.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The reaction mechanism involves a sophisticated interplay of radical and ionic pathways facilitated by the TBAI/TBHP system. Initially, the oxidant TBHP activates the methyl group of the 2-methylquinoline substrate, converting it in situ into a reactive 2-quinolinecarbaldehyde intermediate. This aldehyde then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone species. Subsequent oxidative iodination triggers an intramolecular electrophilic substitution, leading to ring closure and eventual aromatization to form the stable 1,2,4-triazole core. The presence of diphenylphosphinic acid is critical, acting as a promoter that stabilizes key intermediates and accelerates the cyclization kinetics. Understanding this mechanistic pathway allows chemists to fine-tune reaction parameters for optimal high-purity pharmaceutical intermediate output.
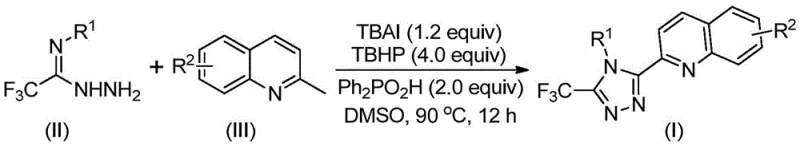
Impurity control is inherently superior in this metal-free system compared to transition-metal catalyzed alternatives. Without the risk of metal leaching or coordination complexes forming with the product, the impurity profile is dominated primarily by organic byproducts that are easily separated via standard chromatography. The tolerance of the reaction to various functional groups on the aryl ring (R1) and the quinoline backbone (R2) demonstrates remarkable robustness. Whether introducing electron-donating groups like methoxy or electron-withdrawing groups like nitro and halogens, the catalytic cycle maintains high efficiency. This broad substrate scope ensures that the process can be adapted for the commercial scale-up of complex pharmaceutical intermediates without requiring extensive re-optimization for each new derivative.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory research and pilot plant production. The procedure involves simply mixing the stoichiometric amounts of TBAI, TBHP aqueous solution, diphenylphosphinic acid, and the two main substrates in a suitable organic solvent such as DMSO. The reaction mixture is then heated to the specified range of 80-100°C and maintained for 8 to 14 hours. Post-reaction workup is straightforward, involving filtration and silica gel treatment followed by column chromatography. For detailed standard operating procedures and specific molar ratios optimized for different substrates, please refer to the guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented method offers transformative benefits that directly impact the bottom line. The shift away from precious metal catalysts to abundant organic salts like TBAI drastically reduces raw material volatility and cost. Additionally, the use of aqueous TBHP as an oxidant is safer and more economical than handling hazardous anhydrous peroxides. These factors combine to create a manufacturing process that is not only cheaper but also more resilient to supply chain disruptions common with specialty reagents. The simplified workflow means faster batch turnover times and reduced energy consumption per kilogram of product produced.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the necessity for costly metal removal resins and extensive analytical testing for residual metals. This qualitative shift in reagent selection leads to substantial cost savings in both material procurement and waste disposal. Furthermore, the high atom economy of the oxidative cyclization minimizes the generation of stoichiometric byproducts, reducing the load on waste treatment facilities. By streamlining the synthesis from five steps to a single pot, labor costs and equipment occupancy time are significantly decreased, enhancing overall production efficiency.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-methylquinoline and various aromatic amines required for the hydrazide, are commodity chemicals available from multiple global suppliers. This diversity in sourcing mitigates the risk of single-supplier dependency and ensures continuous production capability. The reaction's tolerance to moisture and oxygen means that storage and handling requirements for intermediates are less stringent, reducing the likelihood of batch failures due to environmental exposure. Consequently, lead times for high-purity pharmaceutical intermediates can be reliably shortened, supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: The absence of heavy metals aligns perfectly with increasingly strict environmental regulations regarding effluent discharge and product purity. Scaling this reaction from gram to kilogram or ton scale does not introduce new safety hazards associated with pyrophoric reagents or high-pressure hydrogenation. The use of DMSO, a high-boiling polar solvent, facilitates heat management in large reactors, ensuring safe exotherm control during the oxidation phase. This inherent safety profile makes the technology ideal for commercial scale-up in regulated markets where environmental compliance is a critical license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners and licensees.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring 5 steps and yielding only 17%, this patent describes a one-pot oxidative cyclization achieving yields up to 97% without heavy metal catalysts or anhydrous conditions.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses cheap, commercially available starting materials like 2-methylquinoline and avoids sensitive conditions, making it highly scalable for industrial production.
Q: What is the role of diphenylphosphinic acid in the reaction?
A: Diphenylphosphinic acid acts as a crucial additive that promotes the oxidative cyclization efficiency, significantly improving reaction yields compared to systems without it.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocycle synthesis in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole meets the highest international standards. Our commitment to quality assurance means that your supply chain remains robust and compliant with global regulatory requirements.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term growth and innovation goals.
