Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
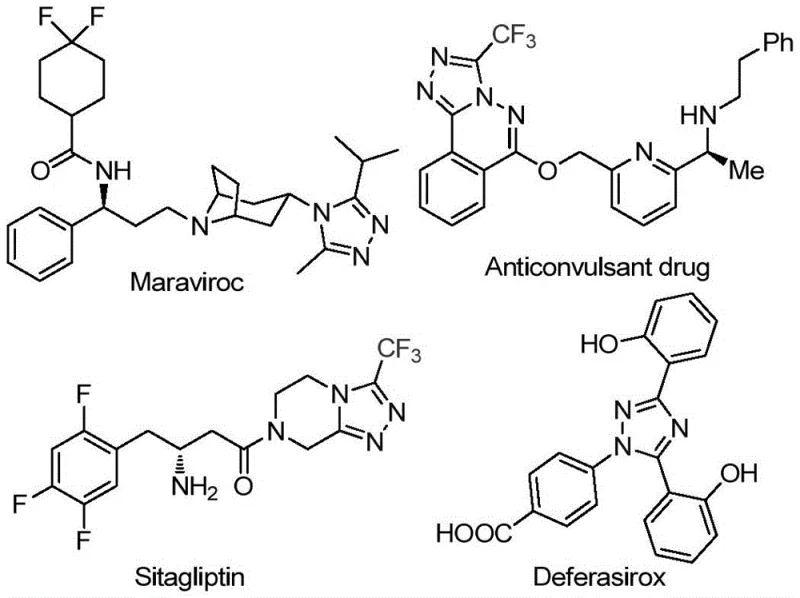
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value drugs such as Maraviroc, Sitagliptin, and Deferasirox. This innovation addresses the longstanding challenge of introducing both trifluoromethyl and acyl groups into the triazole core simultaneously, a modification known to drastically enhance metabolic stability and lipophilicity. By leveraging a non-metal promoted cyclization strategy, this technology offers a streamlined pathway that bypasses the complexities of traditional transition metal catalysis. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the synthesis of complex nitrogen-containing heterocycles while maintaining stringent purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, particularly those bearing electron-withdrawing trifluoromethyl groups, has relied heavily on transition metal catalysts or harsh reaction conditions. Conventional methodologies often necessitate the use of expensive palladium or copper catalysts, which introduce significant cost burdens and complicate downstream processing due to the rigorous requirement for heavy metal residue removal. Furthermore, many existing protocols demand strictly anhydrous and oxygen-free environments, requiring specialized equipment and inert gas manifolds that increase capital expenditure and operational complexity. The sensitivity of intermediates in these traditional routes often leads to variable yields and inconsistent impurity profiles, posing substantial risks for supply chain reliability and regulatory compliance in GMP manufacturing settings.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113105402B utilizes a metal-free system driven by elemental iodine and dimethyl sulfoxide (DMSO). This approach capitalizes on the dual role of DMSO as both a solvent and an oxidant in the Kornblum oxidation of aryl ketones, generating reactive dicarbonyl intermediates in situ. The subsequent condensation with trifluoroethylimide hydrazide proceeds smoothly under relatively mild thermal conditions without the need for exotic reagents. This novel route not only simplifies the operational workflow by eliminating the need for inert atmospheres but also utilizes commercially abundant starting materials like aryl ethyl ketones. The result is a highly practical synthesis that maintains high functional group tolerance, allowing for the diverse substitution patterns seen in modern drug design while ensuring a cleaner reaction profile.
Mechanistic Insights into Iodine-Promoted Cyclization
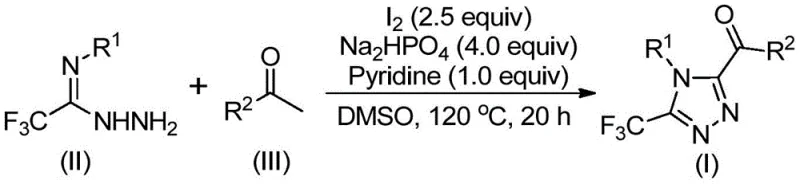
The core of this synthetic breakthrough lies in the elegant cascade of reactions initiated by the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes an iodination followed by a Kornblum oxidation to form an aryl 1,2-diketone intermediate. This oxidative transformation is critical as it activates the carbon backbone for nucleophilic attack. Upon the addition of trifluoroethylimide hydrazide, a dehydration condensation occurs, forming a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine acts as a buffer system, facilitating the subsequent intramolecular cyclization. The iodine species likely promotes the final ring closure by activating the imine bond or stabilizing the transition state, leading to the formation of the stable 1,2,4-triazole ring. This mechanistic pathway avoids the formation of toxic byproducts associated with metal catalysts and ensures that the trifluoromethyl group remains intact throughout the process.
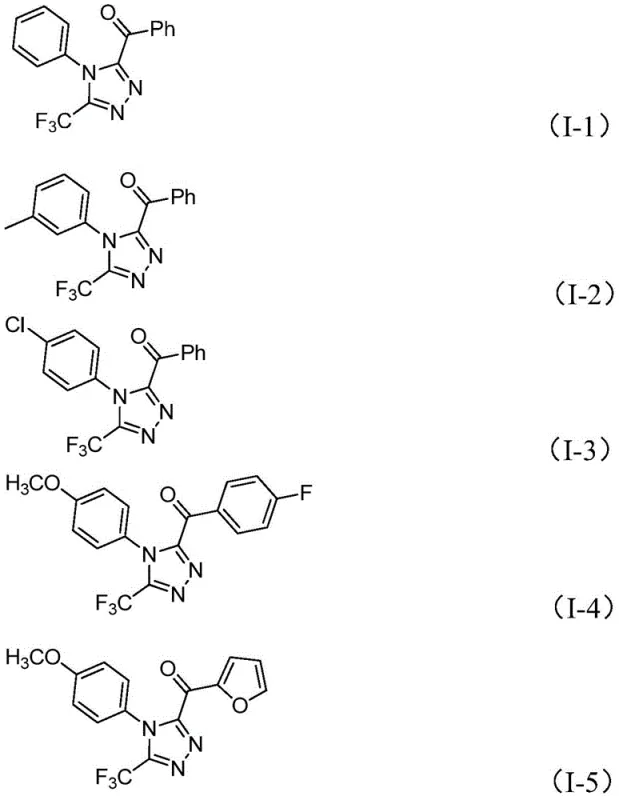
From an impurity control perspective, this mechanism offers distinct advantages. The absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or over-oxidation, which are common pitfalls in traditional cross-coupling strategies. The reaction conditions are sufficiently robust to tolerate various substituents on the aryl rings, including halogens, alkoxy groups, and alkyl chains, as demonstrated by the successful synthesis of derivatives I-1 through I-5. The use of column chromatography for purification, as described in the patent examples, allows for the effective separation of any unreacted starting materials or minor side products, ensuring that the final API intermediate meets the high-purity specifications demanded by regulatory bodies. This level of control is essential for minimizing genotoxic impurities and ensuring batch-to-batch consistency.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemistry teams looking to transfer reactions from the lab to pilot plants. The procedure involves a sequential addition strategy where the oxidation step is separated from the cyclization step, allowing for better control over exotherms and reaction kinetics. By optimizing the molar ratios of iodine, base, and hydrazide, manufacturers can maximize conversion rates while minimizing reagent waste. The detailed standardized synthesis steps below outline the precise conditions required to achieve optimal yields, serving as a foundational guide for scaling this technology to commercial production levels.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis offers tangible strategic benefits beyond mere chemical efficiency. The shift away from precious metal catalysts directly translates to significant cost reduction in pharmaceutical intermediate manufacturing, as the price volatility of palladium and other noble metals is completely circumvented. Moreover, the reliance on commodity chemicals like iodine, DMSO, and simple aryl ketones ensures a stable and diversified supply base, reducing the risk of raw material shortages that can disrupt production schedules. The simplified workup procedure, which avoids complex extraction protocols needed for metal removal, further accelerates the manufacturing cycle time, enhancing overall throughput capacity.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost driver from the bill of materials. Additionally, the avoidance of specialized anhydrous solvents and inert gas setups reduces utility and infrastructure costs. The use of stoichiometric iodine, which is relatively inexpensive compared to organometallic complexes, combined with the high atom economy of the cyclization step, results in a substantially lower cost of goods sold (COGS) for the final triazole intermediate.
- Enhanced Supply Chain Reliability: The starting materials, specifically aryl ethyl ketones and trifluoroethylimide hydrazides, are widely available from multiple global suppliers, preventing single-source dependency. The robustness of the reaction against moisture and oxygen means that storage and handling requirements for raw materials are less stringent, reducing logistics costs and the risk of material degradation during transport. This resilience ensures consistent delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The absence of heavy metals simplifies wastewater treatment and waste disposal, aligning with increasingly strict environmental regulations. The use of DMSO, a solvent with a favorable safety profile and recyclability potential, further supports sustainable manufacturing practices and reduces the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this technology for your specific production needs.
Q: Does this synthesis method require expensive heavy metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metal promoter, significantly reducing raw material costs and eliminating the need for complex heavy metal removal steps typically required in traditional catalytic cycles.
Q: What are the key advantages regarding reaction conditions?
A: The method operates effectively without strict anhydrous or oxygen-free conditions, simplifying the operational requirements and allowing for easier scale-up in standard industrial reactors compared to sensitive organometallic approaches.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the protocol has been demonstrated to be easily expandable from gram levels to larger scales, utilizing cheap and readily available starting materials like aryl ethyl ketones, which supports robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. Our capability to implement metal-free synthetic routes like the one described in CN113105402B underscores our dedication to providing cost-effective and environmentally responsible solutions.
We invite you to collaborate with us to leverage this innovative technology for your pipeline projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall project costs.
