Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, particularly quinolin-2(1H)-one scaffolds which serve as critical cores in numerous bioactive agents ranging from antibiotics to antitumor drugs. A significant breakthrough in this domain is detailed in patent CN114478375A, which discloses a highly efficient preparation method for 3-alkenyl quinolin-2(1H) ketone derivatives. This innovation leverages a palladium-catalyzed reductive aminocarbonylation strategy that fundamentally shifts the paradigm from traditional, hazardous carbonylation protocols to a safer, more operationally simple process. By utilizing o-nitrobenzaldehyde as a dual nitrogen and formyl source alongside allyl aryl ethers, this method achieves excellent reaction efficiency while maintaining a broad tolerance for diverse functional groups. For R&D directors and procurement specialists, this represents a pivotal opportunity to streamline the supply chain for high-value pharmaceutical intermediates, ensuring both cost-effectiveness and regulatory compliance in the synthesis of complex heterocyclic systems.
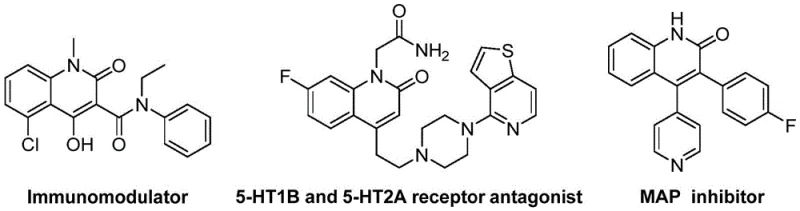
The structural versatility of the resulting 3-alkenyl quinolin-2(1H)one derivatives is paramount for drug discovery programs. As illustrated in the provided data, these scaffolds are not merely theoretical constructs but are directly applicable to the synthesis of potent immunomodulators, receptor antagonists, and MAP inhibitors. The ability to access these structures through a unified synthetic platform allows medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies. Furthermore, the use of readily available starting materials ensures that the transition from bench-scale discovery to kilogram-scale production is seamless, addressing a common bottleneck in the commercialization of new chemical entities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles has relied heavily on transition metal-catalyzed carbonylation reactions using gaseous carbon monoxide or unstable acyl chlorides. Traditional approaches often employ allyl chlorides, acetates, carbonates, or phosphates as electrophilic partners, which present significant logistical and safety challenges. Allyl chlorides, for instance, are lachrymatory and toxic, requiring specialized containment systems and rigorous waste neutralization protocols that inflate operational expenditures. Moreover, the reliance on high-pressure CO gas introduces severe safety risks, necessitating expensive autoclaves and continuous monitoring systems to prevent catastrophic leaks. These conventional methods also frequently suffer from limited substrate scope, where sensitive functional groups on the aromatic ring may degrade under the harsh conditions required to activate less reactive electrophiles. Consequently, the overall yield and purity of the final API intermediate are often compromised, leading to costly downstream purification steps and extended lead times for project milestones.
The Novel Approach
In stark contrast, the methodology described in patent CN114478375A introduces a transformative approach by utilizing allyl aryl ethers as the electrophilic coupling partner. This strategic substitution replaces hazardous halogenated reagents with stable, low-toxicity ethers that are naturally abundant and commercially inexpensive. The reaction proceeds via a palladium-catalyzed reductive aminocarbonylation mechanism where o-nitrobenzaldehyde serves a dual role, providing both the nitrogen atom for the heterocycle and the carbonyl carbon, thereby eliminating the need for external amine sources or separate formylation steps. This atom-economical design significantly simplifies the reaction setup, allowing it to be conducted in standard sealed tubes at moderate temperatures of 100°C. The integration of molybdenum hexacarbonyl as a solid CO surrogate further enhances the safety profile, making this process ideally suited for facilities that lack high-pressure gas infrastructure. This novel route not only improves the environmental footprint of the synthesis but also delivers superior yields across a wide array of substituted substrates.
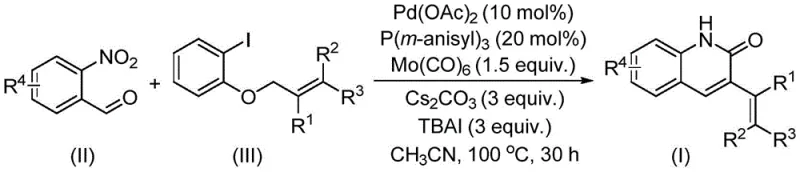
Mechanistic Insights into Pd-Catalyzed Reductive Aminocarbonylation
The success of this synthetic transformation hinges on a sophisticated catalytic cycle involving palladium and molybdenum species. The reaction initiates with the oxidative addition of the palladium catalyst to the allyl aryl ether, facilitated by the electron-rich tris(3-methoxyphenyl)phosphine ligand. Simultaneously, molybdenum hexacarbonyl decomposes under the reaction conditions to release carbon monoxide in situ, which coordinates to the palladium center to form an acyl-palladium complex. The presence of tetrabutylammonium iodide acts as a crucial additive, likely assisting in the activation of the ether bond and stabilizing the cationic palladium intermediates. Subsequently, the o-nitrobenzaldehyde undergoes a reductive cyclization process, where the nitro group is reduced to an amine functionality that immediately attacks the activated acyl species. This intramolecular condensation closes the quinolinone ring, followed by beta-hydride elimination to establish the characteristic 3-alkenyl double bond. The precise tuning of the ligand environment and the use of cesium carbonate as a mild base ensure that the catalytic turnover is high, minimizing the formation of side products such as homocoupled dimers or hydrodehalogenated byproducts.
From an impurity control perspective, this mechanism offers distinct advantages over traditional Friedel-Crafts or condensation routes. The specificity of the palladium insertion into the allyl ether bond prevents random alkylation on the aromatic ring, which is a common issue in acid-catalyzed cyclizations. Furthermore, the use of o-nitrobenzaldehyde as a self-contained nitrogen and carbon source reduces the complexity of the reaction mixture, as there are fewer exogenous reagents that could participate in competing side reactions. The mild basicity of cesium carbonate ensures that acid-sensitive protecting groups or labile functional motifs on the substrate remain intact throughout the 30-hour reaction period. This high level of chemoselectivity translates directly to a cleaner crude reaction profile, reducing the burden on purification teams and ensuring that the final product meets stringent pharmaceutical purity specifications with minimal effort. The robustness of this catalytic system against various electronic environments on the aromatic ring further guarantees consistent quality across different batches.
How to Synthesize 3-Alkenyl Quinolin-2(1H)one Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as outlined in the patent examples. The process begins by charging a reaction vessel with the palladium catalyst, phosphine ligand, molybdenum source, and base, followed by the addition of the specific o-nitrobenzaldehyde and allyl aryl ether substrates in acetonitrile. The mixture is then heated to 100°C for approximately 30 hours to ensure complete conversion. Post-reaction workup involves simple filtration to remove inorganic salts, followed by silica gel chromatography to isolate the pure quinolinone derivative. This standardized protocol has been validated across numerous substrates, demonstrating its reliability for producing diverse analogues.
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum hexacarbonyl, cesium carbonate, and tetrabutylammonium iodide in a sealed tube.
- Add o-nitrobenzaldehyde and the specific allyl aryl ether substrate to the reaction mixture along with acetonitrile solvent.
- Heat the mixture to 100°C for 30 hours, then filter and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical efficiency. The shift from hazardous allyl halides to stable allyl aryl ethers fundamentally alters the raw material sourcing strategy, allowing companies to tap into a broader, more competitive market of commodity chemicals. Since allyl aryl ethers are non-volatile and non-corrosive, they can be transported and stored using standard logistics infrastructure, eliminating the need for specialized hazmat containers and reducing freight costs significantly. Furthermore, the elimination of high-pressure carbon monoxide gas removes a major regulatory hurdle, simplifying facility permitting and insurance requirements. This streamlined operational model translates into substantial cost savings and a more resilient supply chain that is less susceptible to disruptions caused by safety incidents or regulatory audits.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the replacement of expensive and dangerous reagents with cheap, commercially available alternatives. By utilizing o-nitrobenzaldehyde as a dual-purpose reagent, the process reduces the total number of raw materials required, lowering the bill of materials for each batch. Additionally, the use of molybdenum hexacarbonyl as a solid CO source eliminates the capital expenditure associated with high-pressure gas handling equipment and the recurring costs of gas cylinder rentals. The high reaction efficiency reported in the patent implies that less solvent and energy are wasted on processing unreacted starting materials, further driving down the cost per kilogram of the final API intermediate. These cumulative savings allow for a more competitive pricing structure in the global market for pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability and stability of key starting materials. Allyl aryl ethers are robust compounds with long shelf lives, unlike allyl halides which can degrade or hydrolyze upon exposure to moisture. This stability ensures that inventory can be held safely for longer periods without quality degradation, providing a buffer against market fluctuations. Moreover, the wide substrate tolerance of the reaction means that if a specific substituted aldehyde becomes unavailable, alternative analogues can often be synthesized using the same core protocol without extensive re-optimization. This flexibility empowers supply chain planners to maintain production schedules even when facing raw material shortages, ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology is inherently designed for scalability. The absence of toxic gas evolution and the use of mild temperatures make the transition from laboratory glassware to industrial reactors straightforward and safe. From an environmental standpoint, the process generates significantly less hazardous waste compared to traditional methods that produce stoichiometric amounts of halide salts. The simplified workup procedure, which avoids complex extractions or neutralizations of corrosive acids, reduces the volume of wastewater generated. This alignment with green chemistry principles not only lowers waste disposal costs but also enhances the company's sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific pipeline projects. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of using allyl aryl ethers over allyl chlorides in this synthesis?
A: Allyl aryl ethers are significantly less toxic and more stable than allyl chlorides or carbonates. They eliminate the need for handling hazardous halogenated electrophiles, thereby simplifying waste treatment and improving operator safety during large-scale manufacturing.
Q: How does this method address the safety concerns of traditional carbonylation reactions?
A: This protocol utilizes molybdenum hexacarbonyl (Mo(CO)6) as a solid carbon monoxide surrogate. This avoids the use of high-pressure CO gas cylinders, drastically reducing the risk of leakage and explosion in the production facility.
Q: Is this synthetic route suitable for substrates with sensitive functional groups?
A: Yes, the patent data demonstrates wide substrate tolerance. The mild reaction conditions (100°C) and the specific ligand system allow for the compatibility of various substituents including halogens, methoxy groups, and heterocycles without significant degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in patent CN114478375A for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-alkenyl quinolinone derivatives meets global regulatory requirements. We are committed to delivering high-purity pharmaceutical intermediates that empower your research and commercial success.
We invite you to leverage our technical expertise to optimize your supply chain for these critical heterocyclic building blocks. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your long-term growth objectives in the competitive pharmaceutical landscape.
