Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives for Commercial Scale-Up
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting broad pharmacological potential. A significant breakthrough in this domain is detailed in patent CN114478375A, which discloses a robust preparation method for 3-alkenyl quinolin-2(1H)ketone derivatives. This technology leverages a sophisticated palladium-catalyzed reductive aminocarbonylation strategy, utilizing o-nitrobenzaldehyde as a dual nitrogen and formyl source. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this methodology represents a paradigm shift towards safer, more atom-economical processes that bypass the limitations of traditional high-pressure carbonylation techniques.
The strategic importance of the quinolin-2(1H)-one core cannot be overstated, as it serves as a privileged structure in the design of antibiotics, antitumor agents, and receptor antagonists. The ability to access diverse 3-alkenyl substituted variants efficiently opens new avenues for drug discovery campaigns targeting endothelin receptors and angiotensin II pathways. By integrating this patented approach into our manufacturing portfolio, we address the critical industry demand for high-purity pharmaceutical intermediates that are scalable and compliant with stringent regulatory standards. The following analysis dissects the technical merits and commercial viability of this innovative synthetic pathway.
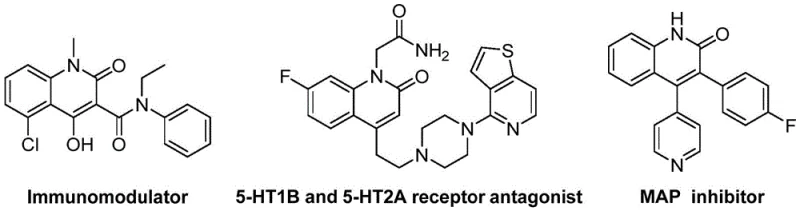
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles has relied heavily on transition metal-catalyzed carbonylation reactions involving gaseous carbon monoxide. While effective, these conventional methods present substantial logistical and safety hurdles for large-scale API manufacturing. The requirement for high-pressure CO gas cylinders necessitates specialized infrastructure, rigorous safety protocols, and often limits the reaction to dedicated facilities equipped for handling toxic gases. Furthermore, traditional electrophiles such as allyl chlorides, acetates, or carbonates, while reactive, often generate stoichiometric amounts of salt waste or require harsh activation conditions that can compromise sensitive functional groups on the substrate.
Additionally, the reliance on pre-functionalized allyl derivatives increases the step count and overall cost of goods sold (COGS). In many legacy processes, the installation of the allyl group requires separate synthetic steps prior to the cyclization event, leading to lower overall yields and increased solvent consumption. For supply chain managers, these inefficiencies translate into longer lead times and higher vulnerability to raw material price fluctuations. The inability to directly utilize simpler, more abundant precursors like allyl ethers has long been a bottleneck in the streamlined production of complex quinolinone architectures.
The Novel Approach
The methodology outlined in patent CN114478375A elegantly circumvents these challenges by employing a palladium-catalyzed system that utilizes solid molybdenum carbonyl [Mo(CO)6] as the carbon monoxide source. This innovation effectively decouples the synthesis from the need for high-pressure gas infrastructure, allowing the reaction to proceed in standard sealed vessels at moderate temperatures (100 °C). The reaction couples o-nitrobenzaldehyde directly with allyl aryl ethers, a class of electrophiles that are naturally abundant, low in toxicity, and operationally simple to handle compared to their halide counterparts.
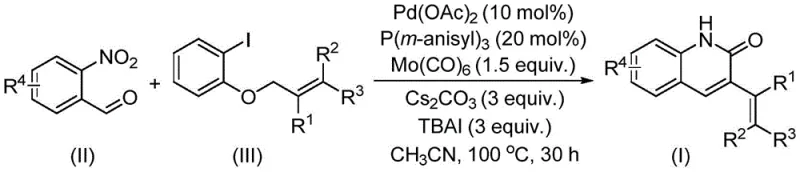
As illustrated in the reaction scheme above, the process integrates the reduction of the nitro group, the carbonylation, and the cyclization into a single pot. This telescoped approach significantly reduces the number of isolation steps, thereby minimizing material loss and solvent waste. The use of o-nitrobenzaldehyde as both the nitrogen and formyl source is a masterstroke of atom economy, ensuring that every atom of the starting material contributes to the final molecular architecture. This novel approach not only enhances the safety profile of the manufacturing process but also drastically simplifies the operational workflow, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Reductive Aminocarbonylation
The catalytic cycle driving this transformation is a complex interplay of organometallic steps orchestrated by the palladium catalyst, specifically palladium acetate [Pd(OAc)2] ligated with tris(3-methoxyphenyl)phosphine. The mechanism initiates with the oxidative addition of the allyl aryl ether to the active Pd(0) species, generating a pi-allyl palladium intermediate. Concurrently, the Mo(CO)6 decomposes under the reaction conditions to release CO, which coordinates to the metal center. The o-nitrobenzaldehyde then undergoes a reductive process, likely facilitated by the phosphine ligand or the metal hydride species generated in situ, converting the nitro group into a reactive amine or imine equivalent capable of nucleophilic attack.
Subsequent migratory insertion of the CO ligand into the Pd-C bond forms an acyl-palladium species. This highly reactive intermediate then undergoes intramolecular nucleophilic attack by the reduced nitrogen species, closing the quinolinone ring and releasing the final product upon reductive elimination. The presence of cesium carbonate (Cs2CO3) acts as a base to neutralize acidic byproducts and facilitate the deprotonation steps essential for the cyclization. Tetrabutylammonium iodide (TBAI) serves as a crucial additive, likely enhancing the solubility of the inorganic base in the organic phase and stabilizing the cationic palladium intermediates, thus accelerating the turnover frequency of the catalyst.
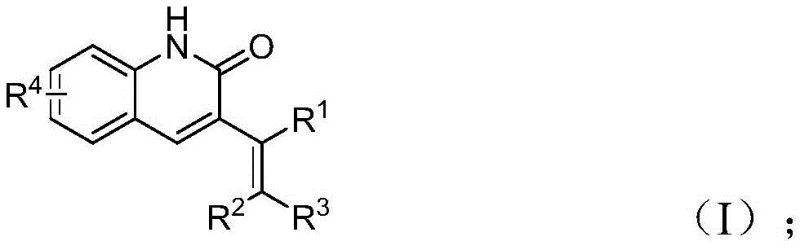
From an impurity control perspective, this mechanism offers distinct advantages. The high chemoselectivity of the palladium catalyst ensures that side reactions, such as homocoupling of the allyl species or over-reduction of the aldehyde, are minimized. The mild reaction temperature of 100 °C prevents thermal degradation of sensitive functional groups, resulting in a cleaner crude reaction profile. This inherent selectivity reduces the burden on downstream purification, allowing for the attainment of high-purity quinolinone derivatives with fewer chromatographic passes. The tolerance for diverse substituents (R1, R2, R3, R4) as shown in the general structure indicates a robust catalytic system capable of accommodating steric and electronic variations without significant loss in efficiency.
How to Synthesize 3-Alkenyl Quinolin-2(1H)one Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, aligning with the needs of process chemistry teams aiming for rapid technology transfer. The protocol involves charging a sealed tube with the requisite molar ratios of palladium acetate (10 mol%), ligand (20 mol%), Mo(CO)6 (1.5 equiv.), base, and additives, followed by the substrates in acetonitrile. The detailed standardized synthesis steps, including precise workup procedures and purification parameters, are provided in the guide below to ensure consistent batch-to-batch quality.
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum carbonyl, cesium carbonate, tetrabutylammonium iodide, o-nitrobenzaldehyde, and allyl aryl ether in acetonitrile.
- Heat the reaction mixture in a sealed tube at 100 °C for approximately 30 hours under stirring conditions.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits beyond mere chemical yield. The shift from gaseous CO to solid Mo(CO)6 fundamentally alters the risk profile of the manufacturing site, eliminating the need for specialized high-pressure gas handling certifications and reducing insurance liabilities. This operational simplification allows for production in a wider range of facilities, thereby enhancing supply chain resilience and reducing the risk of bottlenecks associated with specialized infrastructure.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the utilization of commodity chemicals. O-nitrobenzaldehyde and allyl aryl ethers are widely available bulk chemicals with stable pricing, unlike specialized allyl halides which may command premium prices due to lower production volumes. Furthermore, the telescoped nature of the reaction—combining reduction, carbonylation, and cyclization—eliminates the need for intermediate isolation and purification steps. This consolidation significantly lowers solvent consumption, energy usage, and labor costs, leading to substantial cost savings in the overall production budget without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system and the wide substrate scope ensure that supply continuity is maintained even if specific raw material grades fluctuate. The ability to tolerate various functional groups means that alternative starting materials can be sourced if primary suppliers face disruptions. Additionally, the use of non-hazardous solid reagents simplifies logistics and storage requirements, reducing the complexity of inbound supply chain management and ensuring that raw materials can be stocked safely for extended periods without degradation.
- Scalability and Environmental Compliance: From an environmental standpoint, this process aligns with green chemistry principles by maximizing atom economy and minimizing waste generation. The absence of stoichiometric halide waste (common in allyl chloride routes) reduces the load on wastewater treatment systems. The scalability is further supported by the use of standard heating equipment rather than high-pressure autoclaves, facilitating a smoother transition from kilogram-scale pilot runs to multi-ton commercial production. This ease of scale-up ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable, meeting the aggressive timelines of modern drug development programs.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction parameters and scope. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, ensuring accuracy and relevance for process development planning.
Q: What are the key advantages of using allyl aryl ethers in this synthesis?
A: Unlike traditional allyl chlorides or acetates, allyl aryl ethers are naturally occurring, low-toxicity, and easy to handle. This patent demonstrates the first successful use of allyl aryl ethers in this specific aminocarbonylation, expanding substrate availability and safety.
Q: What is the role of Molybdenum Carbonyl in this reaction?
A: Molybdenum carbonyl [Mo(CO)6] serves as a solid carbon monoxide source. This eliminates the need for handling hazardous high-pressure CO gas cylinders, making the process significantly safer and more amenable to standard laboratory or pilot plant equipment.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method exhibits excellent functional group tolerance. It successfully accommodates substituents such as halogens (F, Cl), methoxy groups, and various alkyl chains on both the aldehyde and the ether components, yielding products with high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from bench-scale discovery to commercial manufacturing requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in patent CN114478375A can be seamlessly integrated into your supply chain. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 3-alkenyl quinolin-2(1H)one derivatives meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and value for your organization.
