Scalable Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives via Novel Palladium Catalysis
Scalable Synthesis of 3-Alkenyl Quinolin-2(1H)one Derivatives via Novel Palladium Catalysis
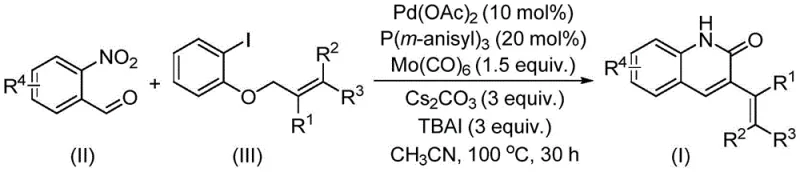
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly quinolin-2(1H)-one derivatives, due to their profound biological activities ranging from antibiotic to antitumor properties. A significant breakthrough in this domain is detailed in patent CN114478375A, which discloses a highly efficient preparation method for 3-alkenyl quinolin-2(1H) ketone derivatives. This technology leverages a sophisticated palladium-catalyzed reductive aminocarbonylation strategy, utilizing o-nitrobenzaldehyde as a dual nitrogen and formyl source. The significance of this development cannot be overstated for R&D teams focused on complex API intermediates, as it offers a streamlined pathway to access privileged scaffolds found in drugs like nybomycin. By integrating this methodology, manufacturers can achieve high-purity pharmaceutical intermediates with improved operational simplicity.
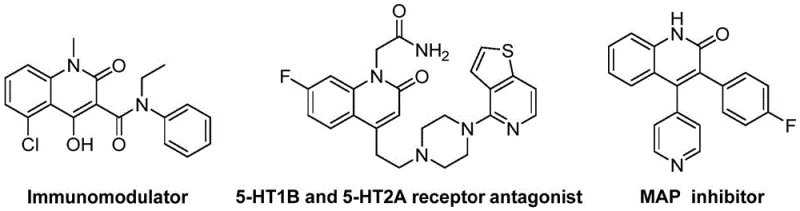
The structural diversity achievable through this method is exemplified by the various bioactive motifs shown in the reference data, highlighting the versatility of the quinolin-2(1H)one core in medicinal chemistry. These compounds serve as critical building blocks for endothelin receptor antagonists and angiotensin II receptor antagonists, underlining the commercial value of mastering their synthesis. For procurement managers and supply chain heads, understanding the underlying chemistry of such patents is essential for securing reliable pharmaceutical intermediate suppliers who can deliver consistent quality at scale. The ability to synthesize these derivatives efficiently directly impacts the cost reduction in API manufacturing and ensures a stable supply chain for downstream drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of quinolin-2(1H)one derivatives has relied heavily on transition metal-catalyzed carbonylation reactions involving aryl or vinyl halides. While effective, these conventional methods often suffer from significant drawbacks that hinder large-scale industrial application. Primary among these issues is the reliance on allyl chlorides, acetates, carbonates, or phosphates as electrophiles, which can be toxic, corrosive, or difficult to handle safely in a commercial plant environment. Furthermore, many existing protocols require harsh reaction conditions, expensive ligands, or generate substantial amounts of hazardous waste, complicating the environmental compliance and waste treatment processes. The limited substrate scope of older methods often restricts the introduction of diverse functional groups, forcing chemists to employ lengthy protection-deprotection sequences that drastically reduce overall yield and increase production lead times.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes allyl aryl ethers as electrophiles, representing a paradigm shift towards greener and more sustainable chemistry. Allyl aryl ethers are naturally occurring, low-toxicity compounds that are significantly easier to operate with compared to their halide counterparts. This method employs o-nitrobenzaldehyde not just as a reactant but as a multifunctional reagent providing both nitrogen and carbonyl components, thereby simplifying the reaction stoichiometry. The process demonstrates exceptional functional group tolerance, allowing for the incorporation of methyl, methoxy, trifluoromethyl, and halogen substituents without compromising reaction efficiency. As illustrated by the specific examples generated, this route consistently delivers high yields, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
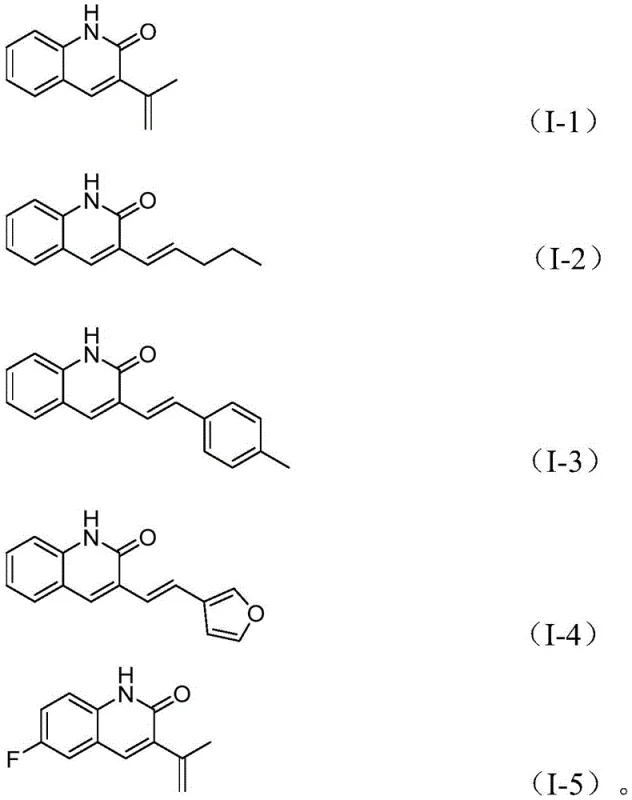
Mechanistic Insights into Palladium-Catalyzed Reductive Aminocarbonylation
The core of this innovative synthesis lies in the intricate interplay between the palladium catalyst and the molybdenum carbonyl source. The reaction mechanism initiates with the oxidative addition of the palladium species to the allyl aryl ether, facilitated by the specific ligand environment provided by tris(3-methoxyphenyl)phosphine. This step is critical for activating the relatively inert ether bond, a transformation that is historically challenging in organic synthesis. Subsequently, the insertion of carbon monoxide, generated in situ from molybdenum hexacarbonyl, leads to the formation of an acyl-palladium intermediate. The presence of o-nitrobenzaldehyde introduces a unique reductive aminocarbonylation pathway where the nitro group is reduced while simultaneously participating in the cyclization event to form the quinolinone ring. This dual functionality minimizes the need for external reducing agents and streamlines the catalytic cycle.
Understanding the impurity profile is paramount for R&D directors ensuring product purity. The use of cesium carbonate as a base and tetrabutylammonium iodide as an additive plays a crucial role in suppressing side reactions such as homocoupling or premature hydrolysis of the intermediate species. The specific choice of acetonitrile as the solvent ensures optimal solubility of the polar intermediates while maintaining a reaction temperature of 100°C that is high enough to drive the kinetics but low enough to prevent thermal degradation of sensitive functional groups. The result is a clean reaction profile where the desired 3-alkenyl quinolin-2(1H)one is the major product, significantly reducing the burden on downstream purification processes like column chromatography. This mechanistic robustness translates directly into higher batch consistency and reliability for industrial partners.
How to Synthesize 3-Alkenyl Quinolin-2(1H)one Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurities. The protocol involves charging a sealed tube with the catalytic system comprising palladium acetate and the specialized phosphine ligand, alongside the carbonyl source and base. The substrates, o-nitrobenzaldehyde and the specific allyl aryl ether derivative, are then introduced in a defined molar ratio to ensure complete conversion. The reaction mixture is heated under controlled conditions to facilitate the cascade transformation. For detailed operational specifics regarding stoichiometry, workup procedures, and purification techniques, please refer to the standardized guide below which outlines the exact experimental steps validated by the patent data.
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum carbonyl, cesium carbonate, and tetrabutylammonium iodide in a sealed tube.
- Add o-nitrobenzaldehyde and allyl aryl ether substrates along with acetonitrile solvent to the reaction mixture.
- Heat the mixture at 100°C for 30 hours, then filter and purify via column chromatography to isolate the derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that address key pain points in the global supply chain for fine chemicals. The shift towards using readily available and inexpensive starting materials like o-nitrobenzaldehyde and allyl aryl ethers directly impacts the raw material cost structure. Unlike proprietary or scarce reagents, these inputs are commodity chemicals with stable market pricing, shielding manufacturers from volatile cost fluctuations. Furthermore, the operational simplicity of the process—requiring standard heating and stirring equipment without the need for high-pressure autoclaves typically associated with gas-phase carbonylation—lowers the barrier to entry for contract manufacturing organizations. This accessibility ensures that multiple qualified suppliers can potentially adopt the technology, enhancing supply chain resilience and reducing the risk of single-source dependency for critical API intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic allyl halides in favor of benign allyl aryl ethers results in significant savings on raw material procurement and hazardous waste disposal. Additionally, the high atom economy achieved by using o-nitrobenzaldehyde as a dual-purpose reagent reduces the overall mass intensity of the process. By minimizing the number of synthetic steps and avoiding complex protection strategies, the total processing time is shortened, leading to lower utility costs and increased throughput capacity. These factors collectively contribute to a more competitive cost position for the final pharmaceutical intermediate, allowing for better margin management in downstream drug pricing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture inherent in the catalyst system design, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining uninterrupted production schedules and meeting strict delivery deadlines for global pharmaceutical clients. The wide substrate scope means that a single manufacturing platform can produce a diverse library of derivatives, allowing for agile responses to changing market demands or clinical trial requirements. Suppliers utilizing this technology can offer greater flexibility and shorter lead times for high-purity pharmaceutical intermediates, strengthening partnerships with innovator companies.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the use of common solvents like acetonitrile and standard heating protocols. The absence of highly toxic gases or extreme pressures simplifies the engineering controls required for large-scale reactors, making it safer and more cost-effective to expand production capacity. Moreover, the generation of less hazardous byproducts aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with effluent treatment. This sustainability profile is becoming a key differentiator for suppliers seeking to partner with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of using allyl aryl ethers in this synthesis?
A: Unlike traditional allyl halides, allyl aryl ethers are less toxic, naturally occurring, and easier to handle, while still serving as effective electrophiles in this Pd-catalyzed system.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating substituents such as methoxy, fluoro, chloro, and trifluoromethyl groups on the aromatic rings without significant yield loss.
Q: What is the role of o-nitrobenzaldehyde in this reaction?
A: o-Nitrobenzaldehyde serves a dual purpose as both the nitrogen source and the formyl (carbonyl) source, simplifying the reagent list and improving atom economy compared to multi-component alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN114478375A for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-alkenyl quinolin-2(1H)one derivative meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of palladium catalysis and deliver products with the consistency required for regulatory filings.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your supply chain objectives. Let us be your trusted partner in bringing high-quality pharmaceutical intermediates to the global market efficiently and sustainably.
