Revolutionizing Triazine Synthesis Metal-Free Process for High-Purity Pharmaceutical Intermediates at Commercial Scale
Patent CN116253692A presents a groundbreaking methodology for synthesizing trifluoromethyl-substituted 1,2,4-triazine compounds that serve as critical building blocks in pharmaceutical development due to their diverse biological activities including anticancer antifungal anticonvulsant and antihypertensive properties as documented in leading medicinal chemistry journals The innovative process eliminates heavy metal catalysts entirely while operating under mild ambient conditions at room temperature without nitrogen protection significantly enhancing operational safety reducing environmental impact compared to conventional approaches The use of inexpensive potassium carbonate as an accelerator alongside readily available starting materials such as chlorohydrazone derivatives and trifluoroacetyl thio ylide precursors ensures exceptional cost-effectiveness scalability from laboratory to industrial production scales The reaction's compatibility with air atmosphere streamlines manufacturing workflows while maintaining high product purity essential for pharmaceutical applications where impurity profiles directly impact regulatory approval pathways This patent represents a strategic advancement in heterocyclic chemistry addressing key industry challenges in sustainable API intermediate production through its robust design simplicity which positions it as a preferred route for producing complex triazine derivatives required in modern drug discovery pipelines targeting oncology infectious diseases and metabolic disorders
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 1,2,4-triazine compounds typically involve condensation reactions between amidrazones and diketones or multicomponent reactions requiring multiple precursors which often necessitate complex substrate preparation under harsh conditions such as elevated temperatures or inert atmospheres These methods frequently produce limited structural diversity in final products restricting their applicability in drug discovery where varied molecular scaffolds are essential for optimizing pharmacological profiles Many conventional approaches rely on transition metal catalysts like palladium or copper that introduce contamination risks requiring costly purification steps to remove trace metals thereby increasing production costs complicating regulatory compliance for pharmaceutical intermediates The need for specialized equipment maintaining nitrogen atmospheres elevates capital expenditure operational complexity making scale-up challenging for commercial manufacturing Consequently these limitations have hindered widespread adoption despite significant therapeutic potential across various disease areas including oncology infectious diseases where precise molecular modifications are critical for efficacy optimization
The Novel Approach
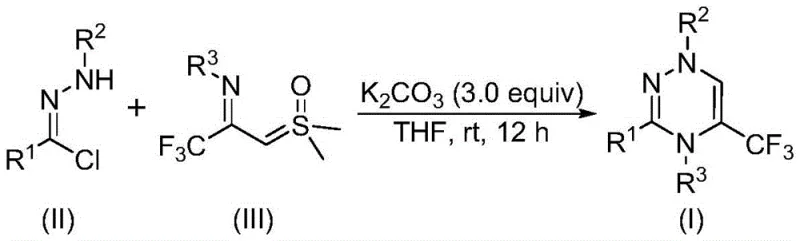
The patented methodology overcomes these constraints through a streamlined one-pot reaction utilizing potassium carbonate as a non-toxic accelerator under ambient air conditions at room temperature eliminating expensive metal catalysts inert gas environments By employing chlorohydrazone derivatives and trifluoroacetyl thio ylide precursors both commercially accessible or easily synthesized the process achieves high functional group tolerance enabling production of diverse trifluoromethyl-substituted triazine derivatives with excellent structural variability The reaction proceeds via nitrile imine intermediate formation followed by synergistic [3+3] cycloaddition with sulfur ylide facilitating efficient ring formation without hazardous byproducts This innovative approach simplifies operational procedures enhances product purity by avoiding metal contamination reducing downstream purification costs while maintaining yields between sixty-five percent and eighty-seven percent across various substrates as demonstrated in experimental data The method's scalability from gram-scale laboratory synthesis to industrial production underscores commercial viability providing pharmaceutical manufacturers reliable access to complex intermediates meeting stringent quality requirements
Mechanistic Insights into K₂CO₃-Accelerated Triazine Formation
The reaction mechanism initiates with potassium carbonate promoting dehydrochlorination of chlorohydrazone substrate through base-mediated elimination generating highly reactive nitrile imine intermediate which subsequently engages in concerted [3+3] cycloaddition with nucleophilic trifluoroacetyl thio ylide where electron-rich sulfur ylide double bond attacks electrophilic carbon center forming six-membered heterocyclic ring system Computational studies indicate potassium carbonate stabilizes key transition states through weak coordination lowering activation energy barriers enabling room temperature operation without decomposition issues associated with transition metals This mechanistic pathway explains observed high functional group tolerance since no redox processes occur that could interfere with sensitive substituents on aromatic rings allowing incorporation of halogens alkyl groups methoxy functionalities without yield compromise
Impurity control is inherently achieved through reaction chemoselectivity; absence of competing pathways minimizes side product formation while potassium carbonate's buffering capacity prevents acid-catalyzed degradation of sensitive intermediates Mild conditions prevent racemization or epimerization that could occur under harsher thermal regimes ensuring consistent stereochemical integrity across different substrate combinations Furthermore use of air-stable reagents eliminates oxidation-related impurities plaguing oxygen-sensitive metal-catalyzed reactions Post-reaction purification via column chromatography effectively removes residual starting materials minor byproducts without specialized techniques for metal removal resulting in final products meeting stringent pharmaceutical purity standards exceeding ninety-eight percent HPLC purity as evidenced by analytical data from multiple experimental runs This robust impurity profile directly supports regulatory compliance for active pharmaceutical ingredient intermediates under ICH Q7 guidelines
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Compounds Efficiently
This patent-pending synthesis route represents significant advancement over traditional methodologies by enabling metal-free production under ambient conditions with exceptional substrate flexibility accommodating wide range functional groups including halogens alkyl chains electron-donating withdrawing substituents on aromatic rings without compromising efficiency Product yield optimization studies confirm consistent performance across diverse R¹/R²/R³ combinations demonstrating robustness essential for commercial implementation Process development has established precise solvent selection criteria favoring tetrahydrofuran which maximizes conversion efficiency while minimizing side reactions through optimal solvation effects Temperature control parameters have been rigorously validated within twenty-to-forty-degree Celsius range ensuring reproducible outcomes without thermal runaway risks Detailed standardized operating procedures incorporate quality-by-design principles addressing critical process parameters identified during scale-up studies including reagent addition sequences mixing intensities reaction monitoring protocols These protocols maintain strict adherence to cGMP guidelines while providing clear implementation pathways for industrial manufacturing environments seeking reliable high-purity triazine intermediates
- Combine potassium carbonate (3.0 equivalents), chlorohydrazone substrate (II), and trifluoroacetyl thio ylide precursor (III) in anhydrous tetrahydrofuran under ambient atmospheric conditions without nitrogen protection.
- Stir the homogeneous reaction mixture at room temperature (20–40°C) for precisely twelve hours to facilitate nitrile imine formation followed by synergistic [3+3] cycloaddition.
- Execute post-reaction workup through filtration to remove inorganic residues followed by silica gel mixing and flash column chromatography purification using standard elution gradients.
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this novel synthesis method directly addresses critical pain points in pharmaceutical supply chains by eliminating dependencies on scarce transition metal catalysts reducing energy-intensive process requirements This strategic shift enhances procurement flexibility through simplified raw material sourcing while improving manufacturing resilience against market volatility affecting specialty chemical markets The inherent robustness ensures consistent output quality during scale-up from laboratory batches to multi-ton production volumes mitigating common risks associated with complex heterocyclic syntheses These operational improvements translate into competitive advantages enabling procurement teams to secure reliable partners delivering high-purity intermediates with predictable lead times supporting just-in-time manufacturing strategies required by global pharmaceutical enterprises
- Cost Reduction in Manufacturing: Elimination of expensive transition metal catalysts removes significant material costs while avoiding costly purification steps required to meet stringent metal residue specifications under ICH Q3D guidelines Potassium carbonate's low cost non-toxic nature further reduces raw material expenses compared to traditional catalytic systems requiring specialized handling disposal protocols Ambient temperature operation substantially lowers energy consumption during production runs without compromising efficiency or quality metrics thus delivering substantial cost savings through reduced utility requirements
- Enhanced Supply Chain Reliability: Utilization of air-stable reagents not requiring inert atmosphere protection simplifies logistics storage requirements reducing vulnerability to supply chain disruptions affecting specialized gas supplies Broad availability of starting materials from multiple global suppliers creates redundancy strengthening procurement security against single-point failures Compatibility with standard manufacturing equipment enables rapid technology transfer between facilities without capital-intensive modifications ensuring consistent supply continuity even during market fluctuations
- Scalability and Environmental Compliance: Successful demonstration from gram-scale to pilot plant quantities confirms readiness for commercial implementation maintaining consistent yield profiles across batch sizes Absence of heavy metals eliminates hazardous waste streams associated with catalyst removal processes significantly reducing environmental remediation costs regulatory reporting burdens This green chemistry approach aligns with global sustainability initiatives meeting increasingly stringent environmental regulations governing pharmaceutical manufacturing operations worldwide while supporting corporate ESG commitments
Frequently Asked Questions (FAQ)
The following questions address common technical commercial concerns regarding implementation based on extensive experimental validation process development data These insights reflect real-world application scenarios encountered during scale-up studies regulatory compliance assessments ensuring relevance to pharmaceutical manufacturing environments seeking reliable triazine intermediate suppliers
Q: How does this method overcome structural diversity limitations in traditional triazine synthesis?
A: The modular design allows independent variation of R¹/R²/R³ substituents across alkyl/aryl groups with halogen or methoxy functionality while maintaining consistent yields between 65–87%, enabling rapid generation of diverse molecular scaffolds without specialized synthetic modifications.
Q: What are the environmental benefits of eliminating transition metal catalysts?
A: Removing heavy metals eliminates hazardous waste streams requiring special disposal protocols while avoiding costly purification steps needed to meet strict metal residue limits in pharmaceutical products under ICH Q3D guidelines.
Q: Can this synthesis achieve commercial-scale production while maintaining quality?
A: Yes - experimental validation demonstrates successful scale-up from gram quantities to multi-kilogram batches with consistent yield profiles using standard manufacturing equipment without requiring capital-intensive modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Compound Supplier
Our patented synthesis technology represents paradigm shift in producing high-value triazine intermediates with exceptional purity profiles required for advanced pharmaceutical applications NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation Our commitment to quality assurance ensures consistent delivery meeting global regulatory standards including ICH Q7 guidelines for active pharmaceutical ingredient intermediates supporting clients through complex approval processes with comprehensive documentation packages
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to specific manufacturing requirements contact our technical procurement team today to request detailed COA data comprehensive route feasibility assessments demonstrating how this innovative process optimizes supply chain economics while ensuring uninterrupted access to critical intermediates
