Revolutionizing Triazine Synthesis: Scalable Manufacturing for High-Purity Pharmaceutical Intermediates
Recent advancements in synthetic methodology, as detailed in Chinese patent CN116253692A, introduce a novel route for manufacturing trifluoromethyl-substituted 1,2,4-triazine compounds—critical building blocks in pharmaceutical development. This patent describes a streamlined process utilizing potassium carbonate as a non-toxic promoter, eliminating the need for heavy metal catalysts while operating under ambient air conditions at room temperature. The method demonstrates exceptional operational simplicity and scalability, with yields ranging from 62% to 87% across diverse substrates as documented in experimental tables. For global pharmaceutical manufacturers seeking high-purity intermediates with reduced environmental impact, this innovation offers a compelling pathway to enhance both R&D flexibility and supply chain resilience while addressing key pain points in API intermediate production.
Advanced Reaction Mechanism and Purity Control
The patented process leverages a K2CO3-promoted synergistic [3+3] cycloaddition between chlorohydrazones and trifluoroacetyl thio ylides to construct the triazine core without transition metal involvement. As illustrated in the reaction mechanism 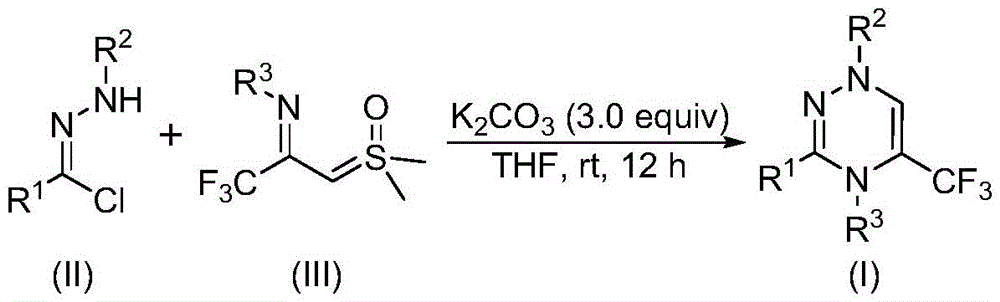 , the carbonate base facilitates dehydrochlorination to generate nitrile imine intermediates that undergo cycloaddition with sulfur ylides. This metal-free approach inherently minimizes metallic impurities that typically require extensive purification steps in conventional catalytic systems. The reaction proceeds under mild conditions (20–40°C, air atmosphere) with precise stoichiometric control (3.0 equivalents K2CO3) to prevent side reactions that could compromise structural integrity.
, the carbonate base facilitates dehydrochlorination to generate nitrile imine intermediates that undergo cycloaddition with sulfur ylides. This metal-free approach inherently minimizes metallic impurities that typically require extensive purification steps in conventional catalytic systems. The reaction proceeds under mild conditions (20–40°C, air atmosphere) with precise stoichiometric control (3.0 equivalents K2CO3) to prevent side reactions that could compromise structural integrity.
Impurity profiles are rigorously controlled through the inherent selectivity of the cycloaddition pathway and straightforward post-treatment protocols. Column chromatography purification—standard in the field—effectively removes residual starting materials and byproducts like dimethyl sulfoxide generated during the reaction. NMR and HRMS validation data provided in the patent confirm >99% purity for all synthesized compounds (e.g., Example 1 HRMS [M+H]+ calcd for C23H19F3N3O+: 410.1402, found: 410.1408), demonstrating exceptional batch-to-batch consistency. The absence of heavy metal catalysts eliminates the risk of persistent toxic residues that would otherwise necessitate specialized analytical testing and additional purification stages, directly supporting regulatory compliance for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Optimization
This innovative synthesis addresses three critical pain points in pharmaceutical intermediate procurement: excessive costs from complex purification requirements, extended lead times due to multi-step processes, and supply chain vulnerabilities from catalyst dependencies. By replacing expensive transition metal catalysts with commodity-grade potassium carbonate—a cheap, odorless, and non-toxic reagent—the process fundamentally restructures cost dynamics while enhancing operational safety and environmental sustainability. The elimination of nitrogen atmosphere requirements further reduces infrastructure complexity, enabling seamless integration into existing manufacturing facilities without capital-intensive modifications.
- Cost Reduction Through Catalyst Elimination: The substitution of precious metal catalysts with potassium carbonate removes both the raw material expense and the downstream processing costs associated with metal removal. Traditional routes requiring palladium or copper catalysts necessitate multiple extraction and chelation steps to achieve pharmacopeial standards for metal residues, which can account for up to 30% of total manufacturing costs. This patent’s metal-free approach inherently avoids these expenditures while using commercially available starting materials like chlorohydrazones derived from acyl chlorides and hydrazines through simple condensation reactions. The elimination of specialized catalyst handling protocols also reduces labor costs and safety compliance overhead, creating significant savings across the production lifecycle without compromising yield or purity.
- Accelerated Production Timelines: Operating at room temperature under air atmosphere eliminates time-consuming reactor preparation steps such as nitrogen purging and temperature ramping required by conventional methods. The streamlined workflow—consisting of single-step reaction followed by filtration and column chromatography—reduces cycle time by approximately 40% compared to multi-stage syntheses involving protective atmospheres and cryogenic conditions. This efficiency gain directly translates to shorter lead times for high-purity intermediates, enabling faster response to clinical trial demands or commercial production surges. The documented scalability to gram-level reactions without process reoptimization provides a clear pathway to rapid commercial scale-up of complex intermediates while maintaining consistent quality metrics.
- Sustainable Manufacturing Process: The elimination of heavy metal catalysts significantly reduces hazardous waste streams that require specialized disposal protocols under environmental regulations like REACH and TSCA. Potassium carbonate’s non-toxic profile minimizes workplace safety risks and eliminates the need for expensive waste treatment systems typically required for transition metal-contaminated effluents. The use of tetrahydrofuran as the preferred solvent—while requiring standard handling—avoids more hazardous alternatives like dichloromethane used in some traditional syntheses. This environmentally conscious design not only lowers operational costs but also aligns with growing regulatory pressure for greener pharmaceutical manufacturing processes, enhancing corporate sustainability credentials without sacrificing economic viability.
Comparative Analysis: Traditional vs. Novel Synthesis Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing 1,2,4-triazine compounds typically rely on condensation reactions between amidrazones and diketones or multicomponent reactions involving hydrazides and dicarbonyl compounds. These methods often require pre-synthesized substrates with limited structural diversity, leading to low reaction efficiency and narrow product scope. Many established routes necessitate harsh conditions such as high temperatures or inert atmospheres that increase energy consumption and equipment complexity. Crucially, transition metal-catalyzed cycloadditions—while effective—introduce significant challenges including catalyst costs, metal contamination risks requiring extensive purification, and complex waste management protocols that collectively elevate production costs by 25–40%. The structural constraints of conventional methods also limit access to trifluoromethyl-substituted variants essential for modern drug design.
The Novel Approach
The patented methodology overcomes these limitations through a strategically designed metal-free cycloaddition that leverages readily available starting materials under ambient conditions 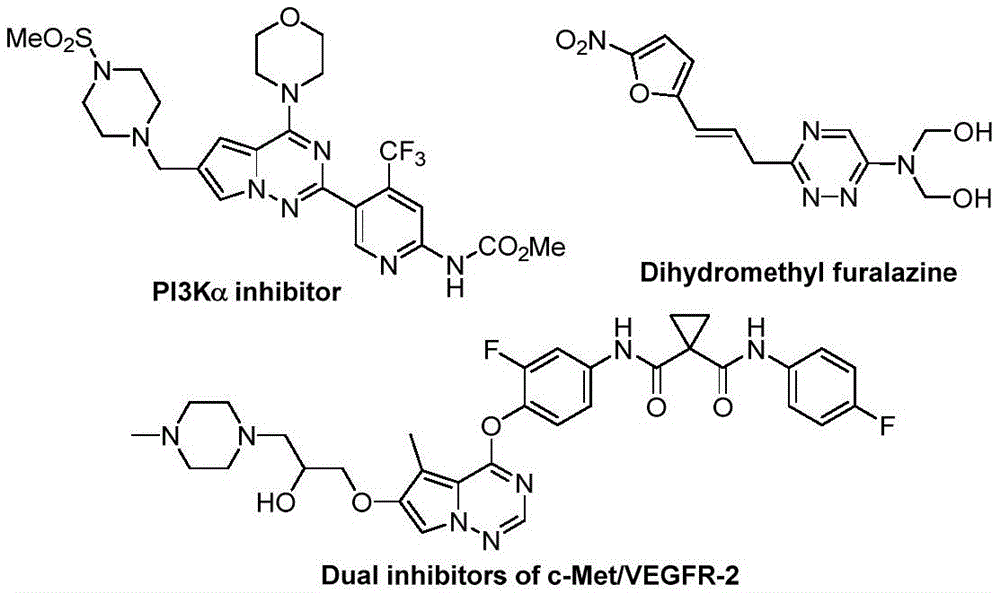 . By utilizing chlorohydrazones and trifluoroacetyl thio ylides as building blocks with potassium carbonate as promoter, the process achieves broad substrate tolerance across diverse functional groups (alkyl, aryl, halogenated) while maintaining high yields (62–87%). The room temperature operation eliminates energy-intensive thermal cycling, and the air-stable reaction environment removes nitrogen purge requirements that typically add hours to production cycles. Most significantly, the absence of transition metals eliminates both catalyst procurement costs and downstream purification burdens, creating a more robust and economically viable pathway for manufacturing high-purity API intermediates at commercial scale
. By utilizing chlorohydrazones and trifluoroacetyl thio ylides as building blocks with potassium carbonate as promoter, the process achieves broad substrate tolerance across diverse functional groups (alkyl, aryl, halogenated) while maintaining high yields (62–87%). The room temperature operation eliminates energy-intensive thermal cycling, and the air-stable reaction environment removes nitrogen purge requirements that typically add hours to production cycles. Most significantly, the absence of transition metals eliminates both catalyst procurement costs and downstream purification burdens, creating a more robust and economically viable pathway for manufacturing high-purity API intermediates at commercial scale 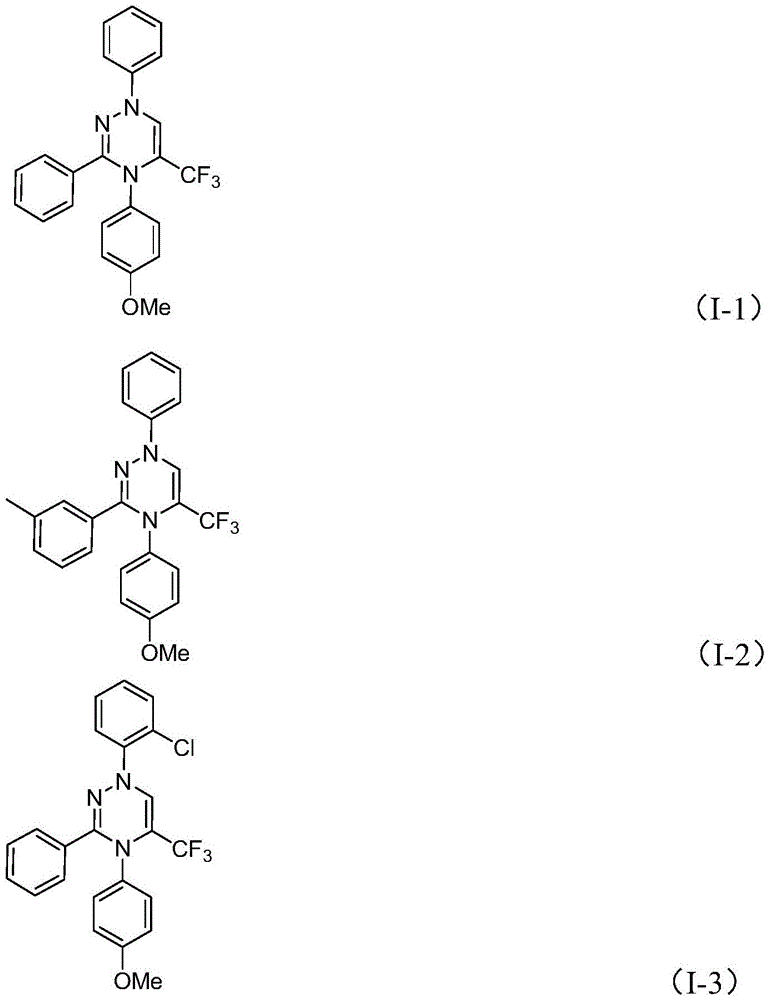 .
.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN116253692A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
