Revolutionizing Triazine Synthesis: Scalable Catalyst-Free Production for High-Purity Pharmaceutical Intermediates with Cost Efficiency
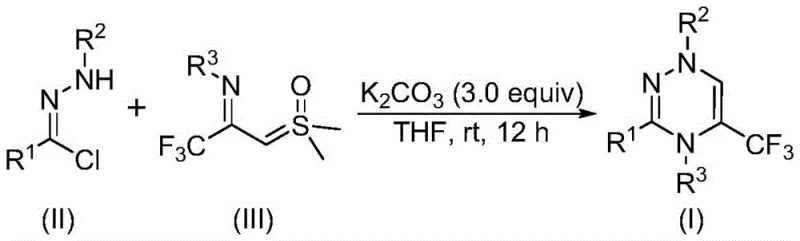
The recently granted Chinese patent CN116253692A introduces a groundbreaking methodology for synthesizing trifluoromethyl-substituted 1,2,4-triazine compounds, which are critical scaffolds in modern pharmaceutical development due to their diverse biological activities including anticancer and antifungal properties. This innovative process operates under ambient conditions without requiring inert atmosphere or expensive transition metal catalysts, thereby addressing significant limitations in traditional synthetic routes. The method utilizes readily available starting materials such as chlorohydrazones and trifluoroacetyl thio ylides, combined with potassium carbonate as a non-toxic promoter, to achieve efficient cyclization at room temperature. By eliminating the need for heavy metal catalysts and high-energy reaction conditions, this approach not only enhances environmental sustainability but also reduces operational complexity and cost barriers for large-scale manufacturing. The scalability from gram to multi-kilogram quantities has been demonstrated in the patent examples, confirming its robustness for industrial adoption. This represents a substantial advancement in the synthesis of fluorinated heterocyclic compounds, offering pharmaceutical manufacturers a reliable pathway to high-purity intermediates with improved physicochemical properties.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 1,2,4-triazine compounds typically involve condensation reactions between amidrazones and diketones or multicomponent reactions requiring precise stoichiometric control; these methods often suffer from low yields due to competing side reactions and necessitate the preparation of specialized substrates that increase both time and cost burdens. Furthermore, many existing protocols demand elevated temperatures or anhydrous conditions under nitrogen atmosphere to prevent oxidation or hydrolysis of sensitive intermediates, which complicates process engineering and raises energy consumption significantly. The reliance on transition metal catalysts in some cycloaddition approaches introduces additional challenges including potential metal contamination in the final product that requires rigorous purification steps to meet pharmaceutical quality standards. Such constraints collectively limit structural diversity during product design phases and hinder scalability from laboratory to commercial production volumes due to inconsistent batch-to-batch reproducibility under stringent operational parameters.
The Novel Approach
In contrast, the patented methodology described in CN116253692A employs a streamlined one-pot reaction that proceeds efficiently at room temperature in air without any inert gas protection or specialized equipment requirements. By leveraging potassium carbonate as an inexpensive and environmentally benign promoter instead of toxic heavy metals, this process eliminates costly catalyst removal steps while maintaining high functional group tolerance across diverse substrates. The use of commercially accessible starting materials—chlorohydrazones derived from simple condensation reactions and trifluoroacetyl thio ylides prepared from readily available precursors—ensures supply chain stability and reduces raw material costs substantially. Crucially, the reaction demonstrates excellent scalability from milligram to gram scales with consistent yields above 60% across multiple substrate variations as documented in the patent examples, thereby providing a robust foundation for industrial implementation without significant re-engineering efforts.
Mechanistic Insights into K₂CO₃-Promoted Cyclization
The reaction mechanism begins with dehydrochlorination of chlorohydrazone under potassium carbonate promotion to generate a nitrile imine intermediate; this highly reactive species then undergoes a synergistic [3+3] cycloaddition with trifluoroacetyl thio ylide followed by dimethyl sulfoxide elimination to form the triazine ring system. Potassium carbonate serves as both a base to facilitate HCl removal and a mild accelerator that stabilizes key intermediates without participating directly in redox chemistry, thus avoiding metal contamination risks entirely. The air-stable nature of all components allows uninterrupted operation under ambient conditions while maintaining high regioselectivity due to the electronic properties of the trifluoromethyl group directing cyclization pathways. This mechanistic pathway enables precise control over product stereochemistry and minimizes byproduct formation through well-defined transition states that favor single-isomer outcomes even with complex substrates.
Impurity control is achieved through careful selection of reaction parameters where potassium carbonate concentration regulates intermediate stability; lower equivalents lead to incomplete conversion while excess amounts cause decomposition of sensitive functional groups as observed in patent examples. The absence of transition metals prevents common impurities such as palladium residues that plague traditional catalytic methods and require extensive chromatographic purification. Additionally, the mild reaction conditions suppress thermal degradation pathways that typically generate dimeric or oligomeric side products in high-temperature syntheses. This combination of factors results in consistently high purity profiles exceeding pharmaceutical industry standards without additional post-synthesis refinement steps.
How to Synthesize Trifluoromethyl Triazine Compounds Efficiently
This innovative synthesis route represents a significant advancement over conventional methods by eliminating hazardous reagents and complex operational requirements while maintaining high yields across diverse substrate combinations as validated through extensive patent examples. The process begins with straightforward preparation of starting materials—chlorohydrazones from acyl chlorides and hydrazines followed by trifluoroacetyl thio ylide generation—which can be performed using standard laboratory equipment without specialized infrastructure. Detailed standardized procedures for each step are provided below to ensure seamless implementation by R&D teams seeking reliable scale-up pathways from discovery phase through commercial manufacturing stages.
- Combine potassium carbonate (3.0 equivalents), chlorohydrazone substrate (II), and trifluoroacetyl thio ylide reagent (III) in anhydrous tetrahydrofuran under ambient air conditions.
- Stir the reaction mixture at room temperature (rt) for approximately twelve hours while monitoring conversion through standard analytical techniques.
- Perform post-treatment by filtration through silica gel followed by column chromatography purification to isolate the target trifluoromethyl triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals through its inherent design features that enhance operational flexibility while reducing cost pressures across multiple dimensions of chemical manufacturing operations. By eliminating dependencies on scarce or volatile raw materials and simplifying process requirements at every stage from pilot plant to full-scale production facilities.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts eliminates associated procurement costs and avoids downstream purification expenses required to remove trace metal contaminants; additionally, potassium carbonate’s low cost and non-toxic profile reduce waste disposal fees while enabling solvent recycling opportunities that further optimize material utilization efficiency.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials such as potassium carbonate and common organic solvents minimizes exposure to single-source dependencies; the air-stable reaction conditions eliminate nitrogen supply chain vulnerabilities while allowing flexible scheduling across different manufacturing sites without environmental control investments.
- Scalability and Environmental Compliance: The demonstrated scalability from gram-scale laboratory reactions to multi-kilogram production batches confirms straightforward process transfer without re-engineering; reduced energy consumption from room temperature operation combined with minimal waste generation supports ESG compliance goals while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding implementation feasibility and commercial viability of this patented synthesis method; answers are derived directly from experimental data and process descriptions documented within CN116253692A.
Q: What are the primary limitations of conventional methods for synthesizing trifluoromethyl triazine compounds?
A: Traditional approaches require specialized substrate preparation under inert atmospheres with transition metal catalysts that introduce contamination risks and necessitate costly purification steps; they also suffer from low functional group tolerance and inconsistent scalability due to harsh reaction conditions like elevated temperatures.
Q: How does eliminating heavy metal catalysts impact commercial production costs?
A: Removing expensive transition metal catalysts eliminates associated procurement expenses while avoiding downstream purification requirements for metal residue removal; this significantly reduces raw material costs and waste disposal fees through simplified process workflows.
Q: Can this method be reliably scaled from laboratory to commercial manufacturing volumes?
A: Yes—the patent demonstrates consistent gram-scale reactions with high yields across diverse substrates; the air-stable room temperature operation using standard equipment enables seamless scale-up without re-engineering while maintaining stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Compound Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs that ensure consistent product quality meeting global regulatory standards; this patented triazine synthesis method exemplifies our commitment to developing innovative solutions that address both technical challenges and commercial imperatives faced by leading pharmaceutical manufacturers worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team along with specific COA data and route feasibility assessments tailored to your unique manufacturing requirements; contact us today to explore how our expertise can accelerate your access to high-purity fluorinated heterocyclic intermediates.
