Innovative Metal-Free Process Delivers High-Purity Trifluoromethyl Triazine Intermediates with Enhanced Scalability for Pharma Supply Chains
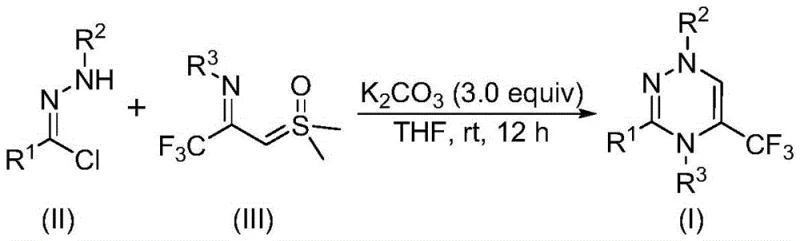
The recently granted Chinese patent CN116253692A introduces a groundbreaking synthetic route for trifluoromethyl-substituted 1,2,4-triazine compounds, which serve as critical building blocks in modern pharmaceutical development due to their diverse biological activities including anticancer, antifungal, and antihypertensive properties as documented in leading journals such as Journal of Medicinal Chemistry (2022). This innovative methodology operates under ambient conditions without requiring inert atmosphere or expensive transition metal catalysts, representing a significant advancement over conventional approaches that often suffer from harsh reaction parameters and complex purification requirements. The process utilizes readily available starting materials such as chlorohydrazones and trifluoroacetyl thio ylides combined with potassium carbonate as a non-toxic accelerator, enabling straightforward scalability from laboratory to industrial production scales while maintaining stringent purity specifications required for pharmaceutical applications. By eliminating the need for heavy metal catalysts and high-energy input conditions, this patent offers a sustainable pathway that aligns with green chemistry principles while providing consistent product quality across diverse substrate combinations as demonstrated through multiple experimental examples in the disclosure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 1,2,4-triazine compounds typically rely on condensation reactions between amidrazones and diketones or multicomponent reactions involving hydrazides and dicarbonyl compounds, which often require pre-synthesized specialized substrates that increase both complexity and cost. These conventional methods frequently operate under elevated temperatures or inert atmospheres with transition metal catalysts that introduce significant purification challenges due to residual metal contamination risks that compromise final product purity standards required by regulatory agencies. Furthermore, the limited structural diversity achievable through existing methodologies restricts medicinal chemistry optimization efforts while low reaction efficiencies necessitate multiple processing steps that extend production timelines and increase waste generation. The inherent instability of some intermediates in traditional routes also creates scalability barriers when transitioning from laboratory to commercial manufacturing environments where consistent yield and purity are paramount for pharmaceutical supply chains.
The Novel Approach
The patented methodology overcomes these limitations through a streamlined one-pot process that operates efficiently at room temperature under ambient air conditions without nitrogen protection or expensive catalysts. By utilizing potassium carbonate as a non-toxic accelerator alongside commercially available chlorohydrazones and trifluoroacetyl thio ylides in standard organic solvents like THF, this approach eliminates the need for specialized equipment while maintaining high functional group tolerance across diverse substrates. The reaction proceeds through a synergistic [3+3] cycloaddition mechanism that avoids hazardous intermediates while delivering consistent yields between 62% to 87% across fifteen experimental examples with various substituents. Crucially, the absence of transition metals removes both purification complexities and regulatory concerns associated with heavy metal residues, while the room temperature operation significantly reduces energy consumption compared to conventional thermal processes requiring elevated temperatures.
Mechanistic Insights into K2CO3-Promoted Triazine Formation
The reaction mechanism begins with potassium carbonate facilitating dehydrochlorination of the chlorohydrazone substrate to generate a nitrile imine intermediate through base-mediated elimination of hydrogen chloride. This highly reactive species then undergoes a concerted [3+3] cycloaddition with the trifluoroacetyl thio ylide component, where the sulfur ylide acts as a three-atom synthon to form the six-membered triazine ring system. The process proceeds through a stepwise nucleophilic addition followed by intramolecular substitution that eliminates dimethyl sulfoxide as a byproduct while establishing the characteristic triazine scaffold with precise regioselectivity. The mild basicity of potassium carbonate optimizes this transformation by maintaining ideal proton transfer conditions without promoting unwanted side reactions that could compromise product purity or yield.
Impurity control is achieved through the inherent selectivity of the cycloaddition pathway which minimizes competing reaction channels while potassium carbonate's buffering capacity prevents acid-catalyzed decomposition pathways common in alternative methodologies. The absence of transition metals eliminates potential coordination complexes that could lead to persistent impurities requiring extensive purification steps. Furthermore, the room temperature operation suppresses thermal degradation pathways that typically generate byproducts in conventional high-energy processes. This combination of factors results in cleaner reaction profiles where post-treatment requires only simple filtration through silica gel followed by standard column chromatography to achieve pharmaceutical-grade purity levels without additional specialized purification techniques.
How to Synthesize Trifluoromethyl Triazine Efficiently
This innovative synthesis route represents a significant advancement in manufacturing trifluoromethyl triazine intermediates by eliminating traditional barriers to commercial production. The patented process leverages readily available starting materials and standard laboratory equipment to deliver high-purity products through a streamlined workflow that maintains exceptional reproducibility across different scales. Detailed standardized synthesis steps are provided below to enable seamless implementation by R&D teams seeking reliable manufacturing solutions for these critical pharmaceutical building blocks.
- Combine potassium carbonate (3.0 equivalents), chlorohydrazone substrate, and trifluoroacetyl thio ylide in anhydrous THF under ambient air conditions
- Stir the reaction mixture at room temperature (20-40°C) for 10-14 hours without nitrogen protection or specialized equipment
- Perform post-treatment by filtration through silica gel followed by column chromatography purification to isolate the target triazine compound
Commercial Advantages for Procurement and Supply Chain Teams
This novel manufacturing approach directly addresses critical pain points in pharmaceutical intermediate procurement by transforming complex multi-step syntheses into a single streamlined process that enhances both cost efficiency and supply chain resilience. The elimination of specialized equipment requirements and hazardous reagents reduces capital expenditure while improving operational safety profiles across manufacturing facilities. By utilizing commodity chemicals instead of expensive catalysts or protected atmospheres, this methodology creates significant flexibility in sourcing strategies that mitigates vulnerability to supply chain disruptions common in traditional pharmaceutical intermediate production.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts eliminates associated procurement costs and complex purification steps required to remove metal residues, resulting in substantial operational savings through reduced raw material expenses and simplified waste management protocols without compromising product quality standards.
- Enhanced Supply Chain Reliability: Utilizing commercially available starting materials with stable global supply chains ensures consistent raw material availability while eliminating dependence on scarce or geopolitically sensitive catalysts that create procurement vulnerabilities in traditional manufacturing approaches.
- Scalability and Environmental Compliance: The room temperature air-stable process enables straightforward scale-up from laboratory to commercial production without reoptimization requirements while generating minimal hazardous waste streams that align with increasingly stringent environmental regulations across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN116253692A's experimental data and implementation protocols. These insights have been derived directly from the disclosed methodology to provide accurate guidance for procurement and R&D decision-making processes regarding this innovative manufacturing solution.
Q: How does this metal-free synthesis improve impurity profiles compared to conventional methods?
A: The elimination of transition metal catalysts prevents heavy metal contamination while potassium carbonate's mild basicity minimizes side reactions. This results in cleaner reaction profiles with reduced byproduct formation, directly enhancing final product purity without requiring additional purification steps that complicate traditional routes.
Q: What scalability advantages does the room temperature air-stable process offer?
A: Operating at ambient conditions eliminates energy-intensive temperature control systems and inert atmosphere requirements. This simplifies reactor design and allows seamless scale-up from laboratory to commercial production without reoptimization, significantly reducing capital expenditure and accelerating time-to-market for new pharmaceutical intermediates.
Q: How does avoiding heavy metal catalysts impact supply chain reliability?
A: Removing dependence on scarce transition metals eliminates volatile pricing risks and geopolitical supply constraints. The use of commodity chemicals like potassium carbonate ensures consistent raw material availability while simplifying regulatory compliance for global pharmaceutical manufacturers requiring uninterrupted intermediate supply.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazine Compound Supplier
Our patented methodology represents a paradigm shift in manufacturing high-value triazine intermediates through an operationally simple yet scientifically sophisticated approach that delivers exceptional purity profiles essential for pharmaceutical applications. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities for comprehensive quality assurance.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative process can optimize your specific supply chain requirements. Contact us today to obtain detailed COA data and route feasibility assessments tailored to your pharmaceutical intermediate needs.
