Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharma Supply Chains
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharma Supply Chains
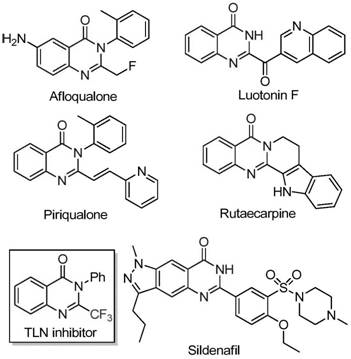
The pharmaceutical industry continuously seeks robust and cost-effective pathways to access privileged heterocyclic scaffolds, particularly those incorporating fluorine atoms which often enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology leverages an inexpensive iron-catalyzed cyclization strategy that transforms readily available trifluoroethylimidoyl chloride and isatin derivatives into high-value intermediates. For R&D directors and procurement specialists, this represents a pivotal shift away from expensive precious metal catalysts towards earth-abundant iron chemistry, promising substantial reductions in production costs without compromising purity or yield. The method's ability to tolerate diverse functional groups ensures its applicability across a wide range of drug discovery programs targeting oncology and infectious diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl groups has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. These traditional routes are frequently plagued by severe reaction conditions that necessitate rigorous exclusion of moisture and oxygen, alongside the use of expensive and sometimes hazardous reagents. Furthermore, the substrate scope in these legacy methods is often narrow, limiting the structural diversity accessible to medicinal chemists during lead optimization phases. Low yields and the generation of difficult-to-remove impurities further exacerbate the economic burden, making large-scale manufacturing of these critical intermediates inefficient and cost-prohibitive for many supply chain managers. The reliance on specialized fluorinating agents also introduces volatility in raw material pricing and availability, creating bottlenecks in the global supply of these essential pharmaceutical building blocks.
The Novel Approach
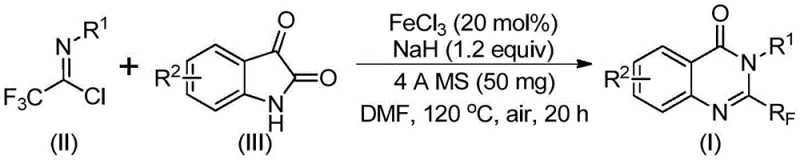
In stark contrast, the methodology described in the patent utilizes a synergistic combination of ferric chloride and sodium hydride to drive the cyclization of trifluoroethylimidoyl chloride with isatins under relatively mild conditions. This innovative route eliminates the need for precious metal catalysts, substituting them with abundant and non-toxic iron salts that drastically lower the catalyst cost profile. The reaction proceeds efficiently in polar aprotic solvents like DMF, demonstrating exceptional functional group tolerance that allows for the incorporation of halogens, alkyl groups, and electron-withdrawing nitro groups without significant yield degradation. By employing a two-stage temperature protocol, initially at 40°C followed by heating to 120°C, the process ensures complete conversion while minimizing side reactions. This approach not only simplifies the operational workflow but also enhances the overall atom economy, making it an attractive candidate for green chemistry initiatives within modern pharmaceutical manufacturing facilities.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base-promoted nucleophilic attack and transition metal-catalyzed decarbonylation. Initially, the strong base sodium hydride facilitates the formation of a carbon-nitrogen bond between the nitrogen of the isatin and the electrophilic carbon of the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. Subsequently, the ferric chloride catalyst activates the system for a decarbonylation event, which is crucial for the formation of the quinazolinone core structure. This iron-mediated step likely involves the coordination of the carbonyl oxygen to the metal center, weakening the carbon-carbon bond and facilitating the extrusion of carbon monoxide. The resulting species then undergoes intramolecular cyclization and aromatization to yield the stable 2-trifluoromethyl quinazolinone product. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as the balance between base strength and catalyst loading directly influences the rate of decarbonylation versus potential hydrolysis side reactions.
Impurity control in this system is inherently managed by the specificity of the iron catalyst and the use of 4A molecular sieves, which play a critical role in scavenging trace water that could otherwise hydrolyze the sensitive imidoyl chloride starting material. The presence of molecular sieves ensures that the reaction environment remains anhydrous, thereby preventing the formation of carboxylic acid byproducts that are common in similar acylation reactions. Additionally, the choice of DMF as a solvent provides a high dielectric constant that stabilizes the charged intermediates formed during the base-promoted steps, further driving the reaction towards the desired product. The robustness of this catalytic cycle allows for the use of air as the oxidant or atmosphere, removing the need for inert gas purging and simplifying the engineering controls required for scale-up. This level of mechanistic clarity provides R&D teams with the confidence to adapt the protocol for continuous flow processing or kilogram-scale batch production.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed to be straightforward, requiring standard laboratory equipment and avoiding exotic reagents that complicate sourcing. The process begins with the precise weighing of ferric chloride and sodium hydride, which are added to a reaction vessel containing the substrate mixture in DMF. The inclusion of 4A molecular sieves at the outset is a critical procedural step that safeguards the integrity of the electrophile throughout the extended reaction time. Operators should monitor the temperature profile carefully, maintaining the initial lower temperature to allow for the formation of the intermediate before ramping up to the higher temperature required for the cyclization and decarbonylation steps. Detailed standardized synthetic steps and specific stoichiometric ratios for various substrates are provided in the technical guide below to ensure reproducibility and high purity outcomes.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed protocol offers transformative benefits regarding cost structure and logistical reliability. The substitution of expensive palladium or rhodium catalysts with commodity-grade ferric chloride results in a direct and significant reduction in raw material expenditure, which cascades down to lower overall manufacturing costs for the final API intermediate. Moreover, the starting materials, specifically isatins and aromatic amines used to generate the imidoyl chlorides, are widely produced commodities with stable global supply chains, mitigating the risk of shortages that often plague specialty fluorinated reagents. The simplicity of the workup procedure, involving filtration and standard column chromatography, reduces the demand for complex purification infrastructure and minimizes solvent consumption, aligning with sustainability goals. This streamlined process enhances the agility of the supply chain, allowing for faster response times to market demands and more flexible production scheduling without the burden of extensive catalyst recovery systems.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the necessity for expensive metal scavenging resins and rigorous residual metal testing, which are significant cost drivers in pharmaceutical production. By utilizing iron, a metal with negligible toxicity concerns compared to heavy metals, the downstream purification burden is drastically simplified, leading to substantial operational savings. The high atom economy of the reaction ensures that a greater proportion of the input mass is converted into valuable product, reducing waste disposal costs associated with unreacted starting materials. Furthermore, the ability to run the reaction under air rather than strict inert atmosphere conditions lowers the capital expenditure required for specialized reactor setups and gas handling systems.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals like isatin and ferric chloride ensures a stable and diversified supplier base, reducing dependency on single-source vendors for niche fluorinated synthons. This diversification strengthens the resilience of the supply chain against geopolitical disruptions or regional production halts, ensuring consistent availability of critical intermediates. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites with minimal re-validation, providing flexibility in capacity planning. Additionally, the long shelf-life and stability of the reagents allow for strategic stockpiling, further buffering the supply chain against short-term market fluctuations.
- Scalability and Environmental Compliance: The protocol's demonstrated success at the gram level with potential for industrial scale-up indicates a low technical risk for technology transfer to pilot and commercial plants. The use of DMF, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols, minimizing environmental impact. The absence of toxic heavy metals in the catalyst system simplifies wastewater treatment and solid waste disposal, ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces regulatory hurdles but also enhances the corporate sustainability metrics of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this route for their own production pipelines. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this iron-catalyzed method over traditional synthesis?
A: This method utilizes inexpensive ferric chloride instead of costly precious metals or harsh reagents like trifluoroacetic anhydride, significantly reducing raw material costs while maintaining high functional group tolerance and yield.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability from gram-level laboratory synthesis to potential industrial application, utilizing robust conditions and commercially available starting materials like isatin.
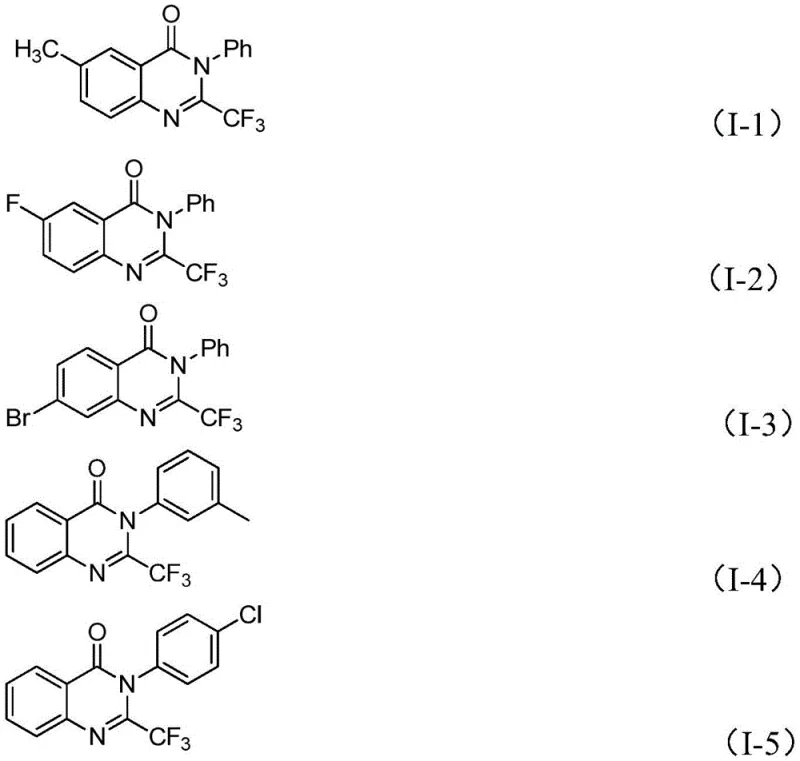
Q: What represents the key structural diversity achievable with this protocol?
A: The protocol accommodates various substituents on both the aryl ring of the imidoyl chloride and the isatin core, allowing for the synthesis of diverse derivatives including methyl, halogen, and methoxy-substituted quinazolinones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the iron-catalyzed cyclization technology and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are supported by rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cost-effective technology for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your journey from bench to bedside.
