Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for Pharmaceutical Applications
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for safer, more efficient, and scalable methodologies. A significant breakthrough in this domain is documented in patent CN112694430B, which details a novel preparation method for 1,5-dihydro-2H-pyrrole-2-one compounds. This specific structural backbone is of paramount importance in medicinal chemistry, serving as the core framework for potent natural products and pharmaceutical agents such as althiomycin, glimepiride, and isomalyngamide A. The patented approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargyl amines and benzyl chlorides as readily accessible starting materials. By replacing traditional gaseous carbon monoxide sources with solid phenol esters, this innovation addresses critical safety and operational challenges inherent in carbonylation chemistry. For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, this technology represents a robust pathway to accessing complex nitrogen-containing heterocycles with high purity and structural diversity.
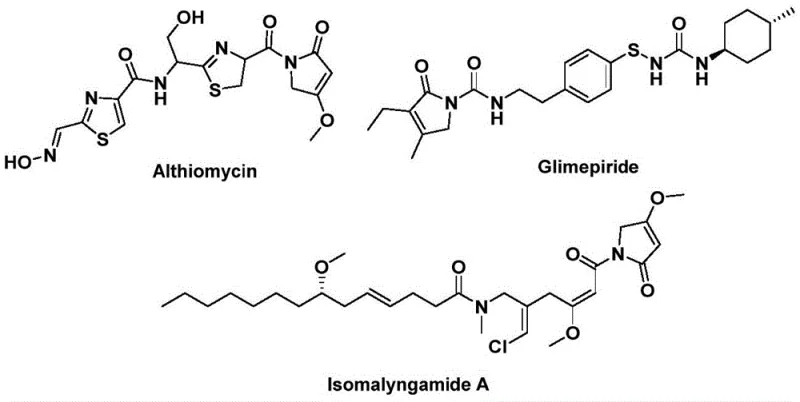
The significance of this synthetic advancement cannot be overstated when considering the broader context of drug discovery and development. The 1,5-dihydro-2H-pyrrol-2-one motif is ubiquitous in molecules exhibiting antibacterial, hypoglycemic, and anticancer activities. However, traditional synthetic routes to access these scaffolds often suffer from limitations such as harsh reaction conditions, poor atom economy, or the requirement for specialized equipment to handle toxic gases. The methodology outlined in the patent offers a streamlined alternative that not only simplifies the operational workflow but also expands the chemical space available to medicinal chemists. By enabling the efficient construction of this core structure in a single step from simple precursors, the process facilitates the rapid generation of analog libraries for structure-activity relationship (SAR) studies. This capability is essential for accelerating the timeline from hit identification to lead optimization in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles has relied heavily on direct carbonylation using carbon monoxide gas. While effective, this approach presents substantial logistical and safety hurdles for both laboratory and industrial settings. The use of high-pressure CO gas requires specialized autoclaves and rigorous safety protocols to prevent leakage and exposure, which significantly increases capital expenditure and operational complexity. Furthermore, traditional methods often struggle with substrate compatibility, particularly when dealing with sensitive functional groups that may degrade under the requisite high temperatures or pressures. Many existing protocols also suffer from low regioselectivity or require stoichiometric amounts of expensive transition metal reagents, leading to increased waste generation and higher purification costs. These factors collectively contribute to extended lead times and reduced overall process efficiency, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
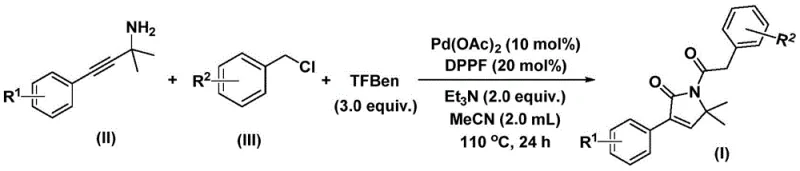
In stark contrast, the novel palladium-catalyzed bis-carbonylation method described in patent CN112694430B offers a transformative solution to these longstanding challenges. By employing 1,3,5-triacetoxybenzene as a solid carbon monoxide substitute, the process eliminates the need for handling hazardous gaseous CO, thereby enhancing workplace safety and simplifying reactor design. The reaction proceeds under relatively mild conditions, typically at 110°C in acetonitrile, using a catalytic system composed of palladium acetate and a diphosphine ligand. This homogeneous catalytic system demonstrates exceptional versatility, accommodating a wide range of substituents on both the propargyl amine and benzyl chloride substrates without compromising yield or selectivity. The one-pot nature of the reaction allows for the direct formation of the five-membered lactam ring through a cascade sequence involving oxidative addition, CO insertion, and cyclization. This streamlined approach not only reduces the number of synthetic steps but also minimizes solvent usage and waste generation, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and predicting substrate scope. The catalytic cycle initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generating a benzyl-palladium(II) intermediate. Concurrently, the thermal decomposition of 1,3,5-triacetoxybenzene releases carbon monoxide in situ, which subsequently inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargyl amine, facilitating the formation of a five-membered ring palladium complex. A second molecule of carbon monoxide is inserted into this cyclic intermediate, expanding the coordination sphere and setting the stage for the final ring closure. The cycle concludes with a reductive elimination step that releases the desired 1,5-dihydro-2H-pyrrole-2-one product and regenerates the active palladium catalyst. This intricate dance of organometallic steps ensures high atom efficiency and precise control over the formation of the carbonyl functionalities within the heterocyclic core.
Beyond the primary catalytic cycle, the robustness of this method is further evidenced by its tolerance to diverse electronic and steric environments. The use of triethylamine as a base plays a dual role, neutralizing the hydrochloric acid byproduct generated during the oxidative addition and potentially assisting in the deprotonation steps required for cyclization. The choice of acetonitrile as the solvent is also critical, as it provides the necessary polarity to dissolve the ionic intermediates while remaining stable under the reaction conditions. Impurity control is inherently managed by the high selectivity of the palladium catalyst, which favors the desired bis-carbonylation pathway over competing side reactions such as homocoupling or simple amidation. For process chemists, this mechanistic clarity allows for fine-tuning of catalyst loading and reaction time to maximize throughput while minimizing the formation of difficult-to-remove impurities, ensuring that the final product meets the stringent purity specifications required for downstream pharmaceutical applications.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical implementation of this synthesis is designed to be straightforward and adaptable to various scales of production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in a defined molar ratio, followed by heating to the specified temperature. The simplicity of the workup procedure, involving filtration and standard column chromatography, makes it particularly attractive for both small-scale discovery and larger-scale manufacturing. Detailed standardized synthetic steps for this transformation are provided in the guide below, ensuring reproducibility and consistency across different batches.
- Combine palladium acetate, DPPP ligand, triethylamine, 1,3,5-triacetoxybenzene, propargyl amine, and benzyl chloride in acetonitrile solvent within a reaction vessel.
- Heat the reaction mixture to 110°C and maintain stirring for 24 hours to facilitate the bis-carbonylation and cyclization process.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages for procurement managers and supply chain leaders focused on cost reduction in pharmaceutical intermediate manufacturing. The reliance on commodity chemicals such as benzyl chlorides and propargyl amines as starting materials ensures a stable and cost-effective supply chain, mitigating the risks associated with sourcing exotic or proprietary reagents. Furthermore, the elimination of high-pressure gas infrastructure reduces capital investment requirements for production facilities, allowing for more flexible and decentralized manufacturing strategies. The high reaction efficiency and yield reported in the patent data suggest that material throughput can be maximized, reducing the cost per kilogram of the final active ingredient. These factors collectively contribute to a more resilient and economically viable supply chain for critical drug substances.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of hazardous gaseous carbon monoxide with a benign solid surrogate. This change drastically simplifies the engineering controls required for the reaction, removing the need for expensive high-pressure reactors and specialized gas handling systems. Additionally, the use of a catalytic amount of palladium, rather than stoichiometric reagents, significantly lowers the raw material cost per batch. The high conversion rates observed mean that less starting material is wasted, and the simplified purification process reduces solvent consumption and labor hours associated with downstream processing. These cumulative efficiencies translate into substantial cost savings that can be passed down through the supply chain, enhancing the competitiveness of the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized reagents or the logistical challenges of transporting hazardous materials. This synthesis method mitigates those risks by utilizing widely available, off-the-shelf starting materials that are produced in large volumes by the global chemical industry. The stability of the solid CO source allows for easier storage and transportation compared to compressed gas cylinders, reducing the likelihood of production delays due to supply shortages. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from multiple vendors. This reliability is crucial for maintaining uninterrupted production schedules and meeting the demanding delivery timelines of major pharmaceutical clients.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, the ability to scale processes while minimizing waste becomes a key differentiator. This palladium-catalyzed method aligns with green chemistry principles by operating in a common organic solvent and generating minimal hazardous byproducts. The absence of toxic gas emissions simplifies compliance with environmental regulations and reduces the burden on waste treatment facilities. The scalability of the reaction is supported by its homogeneous nature and moderate temperature requirements, which facilitate heat transfer and mixing in larger reactors. This ease of scale-up ensures that the transition from laboratory gram-scale to multi-ton commercial production can be achieved smoothly, supporting the long-term growth and sustainability of the manufacturing operation without incurring prohibitive environmental costs.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial aspects of this technology, we have compiled a set of frequently asked questions based on the patent data. These answers provide clarity on the operational parameters, substrate scope, and potential applications of the synthesized compounds. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new drug development projects.
Q: What is the primary advantage of using 1,3,5-triacetoxybenzene in this synthesis?
A: 1,3,5-Triacetoxybenzene serves as a solid, easy-to-handle carbon monoxide substitute, eliminating the need for hazardous high-pressure CO gas cylinders while maintaining high reaction efficiency.
Q: What types of functional groups are tolerated in this palladium-catalyzed protocol?
A: The method demonstrates excellent substrate compatibility, tolerating various substituents including alkyl, alkoxy, halogens (F, Cl, Br), trifluoromethyl, and cyano groups on both the amine and benzyl chloride components.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available starting materials, operates at moderate temperatures (110°C), and avoids toxic gas handling, making it highly scalable and safe for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
The technological potential of this palladium-catalyzed bis-carbonylation route is immense, offering a pathway to high-value heterocyclic intermediates that are essential for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative academic and patent discoveries into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot plant to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our CDMO services are tailored to meet the exacting standards of the global pharmaceutical industry.
We invite you to explore the possibilities of this advanced synthesis method for your upcoming projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic viability of this route for your specific needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to optimize your supply chain and accelerate the delivery of life-saving medicines to patients worldwide.
