Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
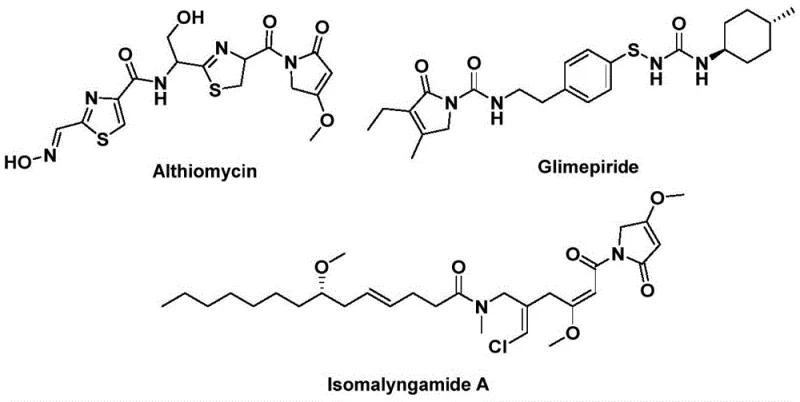
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for a diverse array of potent bioactive natural products and pharmaceutical agents. As illustrated in the biological context, prominent molecules such as Althiomycin, known for its significant antibacterial properties, Glimepiride, a widely prescribed hypoglycemic agent, and Isomalyngamide A, which exhibits potential anticancer activity, all share this critical heterocyclic framework. Recognizing the immense therapeutic value of this chemical space, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This innovative protocol leverages a palladium-catalyzed bis-carbonylation strategy to construct the pyrrol-2-one ring system in a single operational step, marking a significant departure from conventional multi-step sequences. By utilizing inexpensive and commercially accessible starting materials like propargylamines and benzyl chlorides, this technology not only streamlines the synthetic route but also enhances the overall atom economy and process safety, making it an attractive candidate for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied upon classical cyclization strategies that often suffer from significant operational drawbacks and limited substrate scope. Traditional pathways frequently necessitate the use of pre-functionalized cyclic precursors or require harsh reaction conditions involving strong acids or bases, which can lead to the degradation of sensitive functional groups present on the molecule. Furthermore, many existing methods rely on the direct use of carbon monoxide gas, a highly toxic and hazardous reagent that imposes stringent safety requirements and specialized equipment needs on the manufacturing facility, thereby increasing capital expenditure and operational complexity. These conventional approaches often result in lower overall yields due to the accumulation of side products during multi-step sequences, and the purification of the final intermediates can be arduous, requiring extensive chromatographic separation to remove closely related impurities. Consequently, the cost of goods for these valuable intermediates remains prohibitively high, limiting their accessibility for broader drug discovery programs and commercial scale-up efforts.
The Novel Approach
In stark contrast to these legacy methods, the novel palladium-catalyzed bis-carbonylation process described in the patent data offers a streamlined, one-pot solution that dramatically simplifies the synthesis of these complex heterocycles. This methodology ingeniously employs a solid carbon monoxide substitute, specifically identified as TFBen (1,3,5-tricarboxylic acid phenol ester derivative), which safely releases CO in situ under thermal conditions, effectively eliminating the hazards associated with handling gaseous carbon monoxide. The reaction proceeds through a sophisticated cascade involving the insertion of palladium into the benzyl chloride bond, followed by sequential carbonyl insertions and cyclization with the propargylamine moiety. This tandem process allows for the rapid assembly of the five-membered lactam ring with high regioselectivity and efficiency. Moreover, the reaction conditions are remarkably mild, typically operating between 100°C and 120°C in acetonitrile, which ensures compatibility with a wide range of sensitive functional groups. The simplicity of the work-up procedure, involving basic filtration and standard column chromatography, further underscores the practical utility of this approach for both laboratory research and industrial production.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, proceeding through a well-defined cycle of oxidative addition, migratory insertion, and reductive elimination. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, liberated from the solid surrogate TFBen, inserts into the palladium-carbon bond to form an acyl-palladium species. This electrophilic acyl intermediate then undergoes nucleophilic attack by the nitrogen atom of the propargylamine, facilitating the formation of a five-membered ring palladium complex. A second molecule of carbon monoxide is then inserted into this cyclic intermediate, expanding the coordination sphere and setting the stage for the final ring closure. The catalytic cycle concludes with a reductive elimination step that releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium(0) catalyst, allowing the cycle to continue. This intricate dance of bond-breaking and bond-forming events is highly efficient, minimizing the formation of byproducts and ensuring high turnover numbers for the catalyst system.
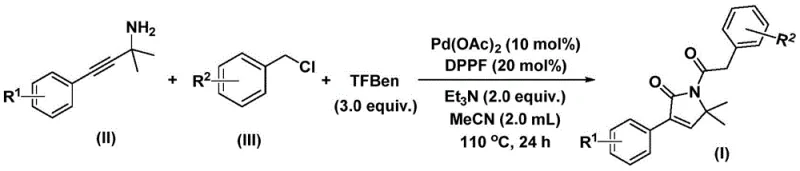
From an impurity control perspective, the mechanism inherently favors the formation of the target lactam structure due to the thermodynamic stability of the five-membered ring and the specific geometry imposed by the ligand environment. The use of DPPP (1,1'-bis(diphenylphosphino)ferrocene) as a bidentate ligand plays a crucial role in stabilizing the palladium center and directing the regioselectivity of the carbonyl insertions. This ligand choice helps to suppress competing pathways such as homocoupling of the benzyl chloride or polymerization of the alkyne moiety, which are common side reactions in palladium-catalyzed processes involving alkynes. Furthermore, the use of triethylamine as a base serves to neutralize the hydrochloric acid generated during the oxidative addition step, preventing acid-catalyzed decomposition of the product or the starting materials. The robustness of this catalytic system is evidenced by its ability to tolerate various substituents on the aromatic rings, including electron-withdrawing halogens and electron-donating alkoxy groups, without significant erosion of yield or selectivity, thereby providing a reliable platform for synthesizing diverse libraries of analogs.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The execution of this synthesis is designed for operational simplicity, requiring standard laboratory equipment and readily available reagents to achieve high-purity outputs. The process begins by charging a reaction vessel with the palladium catalyst, the ferrocene-based ligand, the solid CO source, and the base in an organic solvent, typically acetonitrile, which provides optimal solubility for all components. To this mixture, the specific propargylamine and benzyl chloride derivatives are added, and the system is heated to promote the catalytic cycle. The reaction progress is monitored to ensure complete consumption of the starting materials, typically achieved within a 24 to 48-hour window depending on the electronic nature of the substrates. Following the reaction, the mixture is cooled, filtered to remove insoluble palladium black and salts, and the filtrate is concentrated. The resulting crude material is then subjected to purification via silica gel column chromatography to afford the pure 1,5-dihydro-2H-pyrrol-2-one compound. For a comprehensive, step-by-step guide on the exact molar ratios and specific work-up details, please refer to the standardized protocol below.
- Combine palladium acetate catalyst, DPPP ligand, solid CO source (TFBen), triethylamine base, propargylamine substrate, and benzyl chloride derivative in acetonitrile solvent.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify the crude product via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route presents a compelling value proposition centered around cost optimization and supply security. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, reducing the need for specialized gas handling infrastructure and lowering insurance and compliance costs associated with toxic materials. Furthermore, the reliance on commodity chemicals such as benzyl chlorides and simple propargylamines ensures a robust and diversified supply base, mitigating the risk of raw material shortages that often plague more exotic synthetic routes. The high efficiency of the one-pot process translates directly into reduced processing time and lower energy consumption per kilogram of product, contributing to a leaner and more sustainable manufacturing footprint. By consolidating multiple synthetic steps into a single operation, the overall throughput of the facility can be significantly increased without the need for additional reactor volume, effectively debottlenecking production capacity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive pre-functionalized cyclic starting materials and the reduction in unit operations. By avoiding the isolation of unstable intermediates and utilizing a catalytic amount of palladium rather than stoichiometric reagents, the direct material costs are substantially lowered. Additionally, the simplified work-up procedure reduces the consumption of solvents and silica gel during purification, leading to significant savings in waste disposal and consumables. The high yields reported across a broad substrate scope mean that less raw material is wasted on failed batches or low-yielding steps, maximizing the return on investment for every kilogram of input material purchased.
- Enhanced Supply Chain Reliability: The use of bench-stable reagents and standard organic solvents like acetonitrile ensures that the supply chain is resilient to logistical disruptions. Unlike processes requiring cryogenic conditions or high-pressure gas cylinders, this method can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The tolerance for various functional groups allows for the flexible sourcing of raw materials; for instance, if a specific substituted benzyl chloride is unavailable, alternative derivatives can often be substituted with minimal process re-optimization. This flexibility provides procurement teams with greater leverage in vendor negotiations and ensures continuity of supply even when specific upstream feedstocks face market volatility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with green chemistry principles by replacing toxic carbon monoxide gas with a solid surrogate, thereby eliminating the risk of accidental release. The reaction operates at atmospheric pressure and moderate temperatures, reducing the energy intensity of the process and the associated carbon footprint. The waste stream is primarily composed of benign organic salts and spent catalyst, which can be managed through standard waste treatment protocols, simplifying regulatory compliance. The scalability of the process is proven by its robustness in batch reactions, making the transition from gram-scale laboratory synthesis to ton-scale commercial production straightforward and predictable, ensuring that supply can meet growing market demand without technical barriers.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the scope and limitations of this palladium-catalyzed method. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, offering a transparent view of what can be expected during process development. Understanding these nuances is critical for projecting timelines and resource allocation for new drug candidates or agrochemical intermediates that rely on this pyrrol-2-one core. We encourage our partners to review these details closely to identify opportunities for integrating this efficient chemistry into their existing pipelines.
Q: What are the primary advantages of this carbonylation method over traditional cyclization routes?
A: This novel approach utilizes a one-step palladium-catalyzed bis-carbonylation strategy that eliminates the need for pre-functionalized cyclic precursors. It operates under relatively mild thermal conditions (100-120°C) using readily available benzyl chlorides and propargylamines, significantly simplifying the synthetic workflow compared to multi-step traditional methods.
Q: What is the substrate scope regarding electronic effects on the aromatic rings?
A: The methodology demonstrates excellent functional group tolerance, accommodating both electron-donating groups (such as methoxy and methyl) and electron-withdrawing groups (including fluoro, chloro, bromo, and trifluoromethyl) on both the propargylamine and benzyl chloride components without significant loss in reaction efficiency.
Q: Is this process suitable for large-scale manufacturing of bioactive intermediates?
A: Yes, the process is highly amenable to scale-up due to the use of stable solid carbon monoxide substitutes instead of toxic gas, simple post-treatment procedures involving standard filtration and chromatography, and the commercial availability of all key starting materials and catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the fast-paced pharmaceutical industry. Our team of expert process chemists has thoroughly analyzed this patented technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to your supply chain. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-one derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel analogs for SAR studies or reliable tonnage supply for clinical trials, our CDMO capabilities are tailored to support your project from milligram to metric ton scale with unwavering quality and consistency.
We invite you to engage with our technical procurement team to discuss how this advanced carbonylation technology can drive value in your specific projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined route for your target molecules. Please contact us today to request specific COA data for our catalog compounds or to initiate a confidential discussion regarding route feasibility assessments for your proprietary candidates. Let us help you accelerate your development timeline and reduce your cost of goods with our superior manufacturing expertise.
