Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112480015B introduces a transformative multi-component one-pot methodology for synthesizing 2-trifluoromethyl substituted quinazolinones, a privileged structure found in numerous bioactive molecules ranging from antifungals to anticancer agents. This innovation addresses critical bottlenecks in traditional heterocycle synthesis by leveraging a palladium-catalyzed carbonylation cascade that operates under relatively mild thermal conditions without the need for hazardous high-pressure carbon monoxide gas. For R&D directors and process chemists, this represents a significant leap forward in constructing complex nitrogen-containing heterocycles with high atom economy and operational simplicity.
The strategic incorporation of the trifluoromethyl group is particularly noteworthy, as fluorine atoms dramatically enhance the metabolic stability, lipophilicity, and bioavailability of drug candidates. By streamlining the introduction of this moiety directly during the ring-closing step, the disclosed method offers a direct pathway to high-value pharmaceutical intermediates. The protocol utilizes commercially available nitro compounds and trifluoroethylimidoyl chlorides, bypassing the need for expensive pre-functionalized starting materials. This approach not only accelerates the discovery phase by enabling rapid analog synthesis but also lays a solid foundation for scalable manufacturing processes that meet the rigorous demands of modern API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazolinone cores has relied on methodologies that are fraught with logistical and safety challenges, severely impacting their viability for commercial scale-up. Traditional routes often necessitate the use of high-pressure carbon monoxide atmospheres, requiring specialized autoclaves and stringent safety protocols that inflate capital expenditure and operational complexity. Furthermore, many established protocols depend on ruthenium or platinum catalysts, which are not only prohibitively expensive but also pose significant challenges regarding residual metal removal in final drug substances. Other common strategies involve the condensation of nitrobenzamides with benzylamines or the cyclization of 2-bromoformylaniline derivatives, which frequently suffer from narrow substrate scope, low yields, and the requirement for harsh reaction conditions that can degrade sensitive functional groups.
Additionally, the reliance on pre-activated substrates, such as acid anhydrides or specific halogenated precursors, adds multiple synthetic steps prior to the core ring formation, thereby reducing overall process efficiency and increasing waste generation. These conventional methods often struggle with functional group tolerance, limiting the chemical diversity accessible to medicinal chemists. The cumulative effect of these limitations is a synthesis route that is costly, dangerous, and difficult to optimize for industrial throughput, creating a pressing need for a more streamlined and versatile alternative that can accommodate the diverse structural requirements of modern drug discovery pipelines.
The Novel Approach
The methodology disclosed in CN112480015B fundamentally reimagines the quinazolinone synthesis landscape by employing a tandem carbonylation strategy that merges reduction, coupling, and cyclization into a single operational sequence. By utilizing molybdenum hexacarbonyl [Mo(CO)6] as a solid carbon monoxide surrogate, the process elegantly circumvents the dangers associated with handling gaseous CO, releasing the necessary carbonyl species in situ at a controlled temperature of 120 °C. This shift from high-pressure gas to a solid reagent drastically simplifies the reactor setup, allowing the reaction to proceed in standard glassware or standard stainless steel reactors without the need for specialized high-pressure equipment. The use of a palladium chloride and dppp ligand system ensures high catalytic turnover and excellent selectivity, driving the reaction to completion with impressive yields often exceeding 90% across a wide range of substrates.
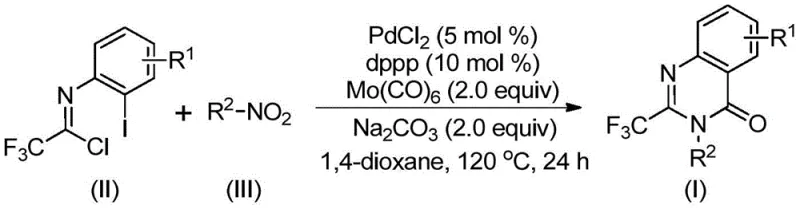
Moreover, this novel approach exhibits exceptional substrate compatibility, tolerating a broad spectrum of electronic and steric environments on both the nitro compound and the imidoyl chloride components. Whether the substrate bears electron-withdrawing halogens, electron-donating alkyl groups, or bulky polycyclic aromatic systems, the catalytic system maintains high efficiency. This versatility is crucial for generating diverse libraries of compounds for structure-activity relationship (SAR) studies. The one-pot nature of the reaction minimizes intermediate isolation steps, reducing solvent consumption and processing time, which translates directly into lower manufacturing costs and a reduced environmental footprint. This represents a paradigm shift towards greener, more efficient chemical manufacturing for complex heterocyclic intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated orchestration of reduction and organometallic cycles that ensures high fidelity in product formation. The process initiates with the reduction of the nitro group on the aromatic substrate to the corresponding amine by Mo(CO)6, which acts as both the CO source and the reducing agent. Once the amine is generated in situ, it undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride, forming a trifluoroacetamidine intermediate. This step is critical as it sets up the nitrogen framework required for the subsequent ring closure. Simultaneously, the palladium catalyst, activated by the dppp ligand, undergoes oxidative addition into the carbon-iodine bond of the imidoyl chloride derivative, generating a reactive organopalladium(II) species that is poised for carbonyl insertion.
As the temperature reaches 120 °C, Mo(CO)6 decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This acyl species is highly electrophilic and susceptible to intramolecular nucleophilic attack by the adjacent nitrogen atom of the amidine moiety. Under the influence of the base (sodium carbonate), a seven-membered palladacycle is formed, which subsequently undergoes reductive elimination to forge the final C-N bond, releasing the 2-trifluoromethyl quinazolinone product and regenerating the active Pd(0) catalyst. This intricate mechanism ensures that the carbonyl group is incorporated precisely at the desired position while maintaining the integrity of the trifluoromethyl group, resulting in a clean impurity profile that simplifies downstream purification efforts significantly.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to maximize the efficiency of the carbonylation cascade. The standard protocol involves charging a reaction vessel with palladium chloride (5 mol %), the bidentate phosphine ligand dppp (10 mol %), and molybdenum hexacarbonyl (2.0 equivalents) alongside the nitro substrate and trifluoroethylimidoyl chloride. Sodium carbonate serves as the base to neutralize the HCl byproduct and facilitate the cyclization step. The reaction is typically conducted in 1,4-dioxane, which provides optimal solubility for all components and stabilizes the catalytic intermediates. Heating the mixture to 120 °C for a period ranging from 16 to 30 hours allows the multi-step cascade to reach full conversion, after which simple filtration and chromatographic purification yield the high-purity target compound.
- Combine palladium chloride (5 mol %), dppp ligand (10 mol %), Mo(CO)6 (2.0 equiv), sodium carbonate (2.0 equiv), trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane.
- Heat the reaction mixture to 120 °C and stir for 16 to 30 hours under inert atmosphere to facilitate the carbonylation cascade.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The primary driver of cost reduction lies in the substitution of expensive, specialized starting materials with commodity chemicals; nitro compounds are among the most abundant and affordable organic building blocks available globally, ensuring a stable and low-cost supply chain. Furthermore, the elimination of high-pressure carbon monoxide gas removes the need for costly safety infrastructure and specialized transport logistics, significantly lowering the barrier to entry for manufacturing facilities. The robustness of the catalyst system also implies longer catalyst life and potentially lower loading rates in optimized processes, contributing to substantial cost savings in precious metal consumption.
- Cost Reduction in Manufacturing: The transition to a one-pot process inherently reduces the number of unit operations, which directly correlates to lower labor costs, reduced energy consumption, and minimized solvent usage. By avoiding the isolation of unstable intermediates and eliminating the need for cryogenic conditions or high-pressure reactors, the overall capital expenditure (CAPEX) and operational expenditure (OPEX) are drastically simplified. The use of earth-abundant iron or inexpensive palladium systems compared to platinum or rhodium alternatives further drives down the raw material bill of materials, making the final API intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Reliance on widely available nitro compounds and standard organic solvents like dioxane mitigates the risk of supply disruptions that often plague specialized reagent markets. Since the starting materials are commodity chemicals produced at massive scales by multiple vendors globally, procurement teams can easily qualify secondary suppliers to ensure business continuity. The simplicity of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, providing flexibility in production planning and inventory management to meet fluctuating market demands without delay.
- Scalability and Environmental Compliance: The avoidance of toxic carbon monoxide gas aligns perfectly with increasingly stringent environmental, health, and safety (EHS) regulations, reducing the regulatory burden and permitting timelines for new production lines. The high atom economy of the reaction minimizes waste generation, supporting sustainability goals and reducing waste disposal costs. The demonstrated scalability from milligram to gram levels in the patent suggests a smooth path to kilogram and ton-scale production, as the reaction does not rely on mixing-sensitive parameters that typically fail upon scale-up, ensuring consistent quality and yield regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this synthesis route for industrial applications. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this chemistry into existing production portfolios.
Q: What are the advantages of using nitro compounds over pre-activated amines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-activated amines or acid anhydrides. This method eliminates the need for harsh pre-activation steps, reducing overall raw material costs and simplifying the supply chain for large-scale production.
Q: How does this method improve safety compared to traditional carbonylation?
A: Traditional methods often require high-pressure carbon monoxide gas, which poses significant safety risks. This patent utilizes Mo(CO)6 as a solid CO surrogate, releasing CO in situ at 120 °C, thereby eliminating the need for high-pressure gas cylinders and specialized autoclaves.
Q: What is the substrate scope for the R2 group in the quinazolinone structure?
A: The method demonstrates excellent compatibility with various R2 groups, including substituted phenyl rings (with methyl, methoxy, halogen, or trifluoromethyl groups), naphthyl groups, cyclohexyl, and alkyl chains, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates like 2-trifluoromethyl quinazolinones. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from pilot plant to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of this Pd-mediated carbonylation, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for clinical and commercial API synthesis.
We invite you to leverage our technical expertise to optimize this route for your specific needs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient one-pot method can improve your margins. Please contact our technical procurement team today to request specific COA data for related analogues and comprehensive route feasibility assessments, and let us partner with you to accelerate your drug development timeline with confidence and precision.
