Advanced Rhodium-Catalyzed C-H Activation for Scalable Indole Derivative Manufacturing
Introduction to Next-Generation Indole Synthesis
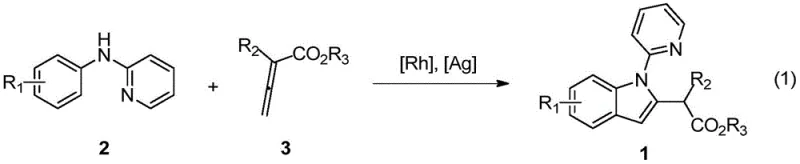
The pharmaceutical industry continuously seeks more efficient pathways to construct privileged scaffolds like the indole core, which is ubiquitous in bioactive molecules ranging from cholesterol-lowering agents to anticancer drugs. Patent CN111285846B introduces a transformative synthetic methodology that leverages transition metal catalysis to streamline the production of 2-(2-indolyl)-acetate derivatives. This technology represents a significant departure from classical stoichiometric approaches, utilizing a rhodium-catalyzed C-H activation strategy that couples readily available N-phenyl-2-aminopyridines with allenoic acid esters. By bypassing the need for pre-functionalized halides or harsh cyclization conditions, this process offers a streamlined route that aligns perfectly with modern green chemistry principles and the demands of high-throughput medicinal chemistry.
For R&D directors and process chemists, the implications of this patent are profound, as it solves long-standing issues regarding regioselectivity and substrate tolerance in indole formation. The method employs a robust catalytic system involving a pentamethylcyclopentadienyl rhodium complex, which facilitates the ortho-C-H bond activation of the aromatic ring followed by insertion of the allenoate. This results in a highly convergent synthesis where complex molecular architectures are assembled in a single operational step. The ability to tolerate diverse functional groups on both the amine and the allenoate components ensures that this platform technology can be adapted for the rapid generation of analog libraries, accelerating the lead optimization phase in drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole ring has relied heavily on classical named reactions such as the Fischer, Madelung, or Bischler syntheses, which, while effective for simple substrates, suffer from significant drawbacks in modern process chemistry. These traditional methods often necessitate the use of corrosive acids, high temperatures, and hydrazine derivatives that pose safety hazards and generate substantial amounts of toxic waste. Furthermore, conventional routes frequently lack the flexibility to introduce substituents at specific positions without extensive protecting group manipulations, leading to longer synthetic sequences and reduced overall yields. The requirement for pre-functionalized starting materials, such as ortho-haloanilines, adds unnecessary steps and cost, creating bottlenecks in the supply chain for complex intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN111285846B utilizes a direct C-H functionalization strategy that dramatically simplifies the synthetic landscape for indole derivatives. By employing a rhodium catalyst to activate the inert C-H bond directly, the process eliminates the need for pre-installed leaving groups, thereby improving atom economy and reducing raw material costs. The reaction proceeds under relatively mild thermal conditions, typically around 60°C in 1,2-dichloroethane, which minimizes energy consumption and reduces the risk of thermal degradation for sensitive functional groups. This approach not only enhances the sustainability profile of the manufacturing process but also opens up new chemical space by allowing the coupling of diverse allenoates that were previously difficult to incorporate using standard cyclization protocols.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The core of this technological advancement lies in the sophisticated catalytic cycle driven by the cationic Rh(III) species generated in situ. The mechanism initiates with the coordination of the pyridine nitrogen to the rhodium center, directing the metal to the ortho-position of the phenyl ring for C-H bond cleavage via a concerted metalation-deprotonation (CMD) pathway. This step is facilitated by the acetate base present in the reaction mixture, which acts as an internal proton shuttle. Following the formation of the five-membered rhodacycle intermediate, the allenoic acid ester coordinates to the metal center and undergoes migratory insertion into the Rh-C bond. This insertion step is highly regioselective, ensuring that the new carbon-carbon bond is formed at the correct position to enable subsequent cyclization.
Once the insertion is complete, the intermediate undergoes a reductive elimination or a similar cyclization event to forge the indole core, releasing the product and regenerating the active Rh(III) catalyst through oxidation by the silver salt. The presence of silver hexafluoroantimonate is critical for generating the cationic rhodium species, while silver acetate serves as the terminal oxidant to close the catalytic loop. This precise control over the mechanistic steps ensures high purity of the final product, as side reactions such as polymerization of the allenoate are minimized by the rapid consumption of the reactive intermediate within the coordination sphere of the metal. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for maximum efficiency and minimal impurity formation.
How to Synthesize 2-(2-indolyl)-acetate Efficiently
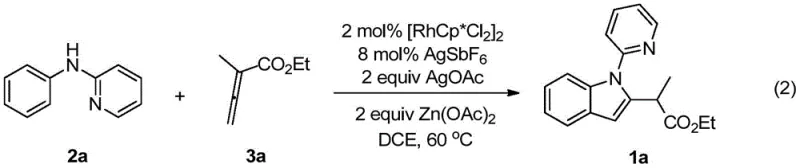
The practical implementation of this synthesis is designed for reproducibility and scalability, utilizing standard laboratory equipment and commercially available reagents. The protocol involves charging a reaction vessel with the N-phenyl-2-aminopyridine substrate, the rhodium dimer catalyst, and the necessary silver additives under an inert atmosphere to prevent catalyst deactivation. Upon addition of the solvent and the allenoate coupling partner, the mixture is heated to facilitate the C-H activation and cyclization sequence. While the general procedure is robust, optimizing the stoichiometry of the oxidant and the choice of solvent can further enhance yields for specific substrate combinations. For detailed standardized operating procedures and specific molar ratios tailored to your target molecule, please refer to the comprehensive guide below.
- Charge a reaction vessel with N-phenyl-2-aminopyridine, [RhCp*Cl2]2 catalyst, AgSbF6 additive, AgOAc oxidant, and Zn(OAc)2 under nitrogen atmosphere.
- Add 1,2-dichloroethane (DCE) as the solvent and introduce the allenoic acid ester coupling component.
- Heat the mixture to 60°C for 24 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this rhodium-catalyzed route offers compelling economic and logistical benefits over legacy manufacturing processes. The primary advantage stems from the simplification of the raw material portfolio; by utilizing simple N-phenyl-2-aminopyridines and allenoates, manufacturers can source starting materials from a broader supplier base, reducing dependency on specialized, high-cost intermediates. This diversification of the supply chain mitigates risks associated with raw material shortages and price volatility, ensuring a more stable production schedule for downstream API manufacturing. Additionally, the high atom economy of the reaction means that a greater proportion of the input mass is converted into the desired product, inherently lowering the cost of goods sold by minimizing waste disposal fees and maximizing material utilization.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps, such as halogenation or lithiation, significantly reduces the number of unit operations required to produce the target indole scaffold. This reduction in step count translates directly into lower labor costs, decreased solvent consumption, and reduced utility usage, as fewer heating and cooling cycles are needed. Furthermore, the use of a catalytic amount of rhodium, despite being a precious metal, is offset by the high turnover number and the ability to recover and recycle the metal from the reaction residue, leading to substantial long-term cost savings compared to stoichiometric reagents.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens, esters, and ethers, allows for the use of commodity chemicals that are readily available in bulk quantities. This compatibility with diverse substrates means that supply chain managers do not need to secure exotic or custom-synthesized building blocks, which often have long lead times and limited availability. The reliability of the process ensures consistent output quality, reducing the need for extensive rework or rejection of batches due to impurity profiles that often plague older synthetic methods.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram scale is facilitated by the mild reaction temperatures and the use of common organic solvents like dichloroethane, which are well-understood in industrial settings. The process generates less hazardous waste compared to traditional indole syntheses that rely on strong acids or heavy metal stoichiometric oxidants, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This environmental friendliness not only reduces regulatory burden but also enhances the corporate sustainability profile, which is a key metric for modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the experimental data and scope outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for project managers evaluating the integration of this route into existing production lines or new product development pipelines.
Q: What are the advantages of this Rh-catalyzed method over traditional Fischer indole synthesis?
A: Unlike traditional methods that often require harsh acidic conditions and pre-functionalized hydrazines, this Rh-catalyzed C-H activation operates under mild conditions (60°C) with high atom economy and eliminates the need for pre-functionalization of the substrate.
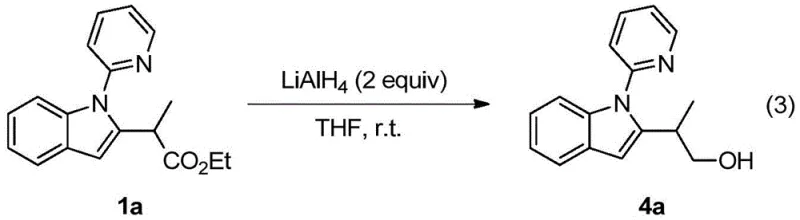
Q: Can the resulting indole derivatives be further functionalized?
A: Yes, the patent demonstrates that the ester group on the product can be successfully reduced to primary alcohols using LiAlH4, proving the scaffold's utility for generating diverse functionalized libraries for drug discovery.
Q: What is the optimal catalyst system for this transformation?
A: The patent identifies [RhCp*Cl2]2 as the optimal catalyst precursor, used in conjunction with AgSbF6 as an activator and AgOAc as the terminal oxidant to achieve high yields and regioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-indolyl)-acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced C-H activation technologies in accelerating the delivery of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-(2-indolyl)-acetate derivative meets the exacting standards required for global regulatory filings.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and competitiveness in your drug development programs.
