Advanced Rhodium-Catalyzed C-H Activation for Scalable Indole Intermediate Manufacturing
Advanced Rhodium-Catalyzed C-H Activation for Scalable Indole Intermediate Manufacturing
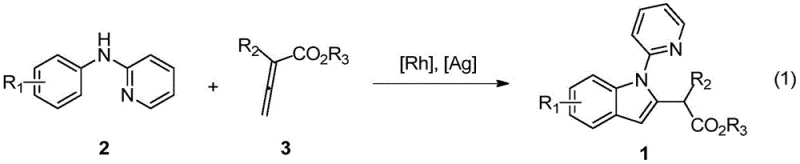
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly indole derivatives which serve as critical building blocks for numerous bioactive molecules. Patent CN111285846B introduces a groundbreaking synthetic methodology that leverages rhodium-catalyzed C-H bond activation to synthesize 2-(2-indolyl)-acetate derivatives with exceptional precision and efficiency. This technology represents a significant paradigm shift from classical stoichiometric approaches, utilizing easily prepared N-phenyl-2-aminopyridine as a divergent synthon. By employing a robust catalytic system involving rhodium salts and silver additives, the process achieves direct ortho-C-H functionalization followed by cyclization with allenoic acid esters. This approach not only streamlines the synthetic route but also enhances the overall atom economy, addressing key sustainability goals in modern chemical manufacturing while providing access to a wide array of structurally diverse indole cores essential for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for constructing the indole nucleus, such as the Fischer, Madelung, or Bischler indole syntheses, have long served as the backbone of heterocyclic chemistry but suffer from inherent inefficiencies that hinder modern process development. These classical methods often necessitate harsh reaction conditions, including strong acids or bases and elevated temperatures, which can lead to poor functional group tolerance and significant safety hazards during scale-up. Furthermore, these routes typically require pre-functionalized starting materials, such as hydrazines or nitro-compounds, introducing additional synthetic steps that increase material costs and generate substantial chemical waste. The requirement for specific substrate activation limits the structural diversity accessible through these pathways, often forcing chemists to design lengthy linear syntheses to install necessary substituents. Consequently, the reliance on these legacy technologies results in lower overall yields, higher E-factors, and increased operational complexity, creating bottlenecks for the rapid production of high-purity pharmaceutical intermediates required by global supply chains.
The Novel Approach
In stark contrast to these legacy limitations, the technology disclosed in CN111285846B utilizes a sophisticated transition metal-catalyzed strategy that directly activates inert C-H bonds, bypassing the need for pre-functionalization entirely. This novel approach employs a cationic rhodium(III) catalyst system to facilitate the oxidative coupling of N-phenyl-2-aminopyridine with allenoates, proceeding through a highly selective cyclization mechanism. The reaction operates under remarkably mild conditions, typically at 60°C in 1,2-dichloroethane, which preserves sensitive functional groups and minimizes energy consumption. By leveraging the directing group ability of the pyridyl moiety, the system achieves excellent regioselectivity, ensuring the formation of the desired 2-substituted indole scaffold without significant isomeric byproducts. This streamlined protocol not only reduces the step count but also significantly improves the atom economy of the transformation, aligning perfectly with the principles of green chemistry and offering a superior alternative for the cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The core of this transformative synthesis lies in the intricate catalytic cycle driven by the pentamethylcyclopentadienyl rhodium(III) complex, which acts as a potent electrophile to activate the aromatic C-H bond. The mechanism initiates with the coordination of the pyridine nitrogen of the N-phenyl-2-aminopyridine substrate to the rhodium center, facilitating a concerted metalation-deprotonation (CMD) process that cleaves the ortho-C-H bond to form a stable five-membered rhodacycle intermediate. This organometallic species then undergoes migratory insertion with the electron-deficient allenoate, a unique coupling partner that offers multiple reaction centers for diversification. Following the insertion step, the intermediate undergoes a series of proton transfers and reductive elimination events, ultimately releasing the cyclized indole product and regenerating the active Rh(III) catalyst species in the presence of the silver oxidant. Understanding this mechanistic pathway is crucial for R&D directors as it highlights the precise control over bond formation, ensuring high purity and minimizing the formation of difficult-to-remove impurities that often plague traditional indole syntheses.
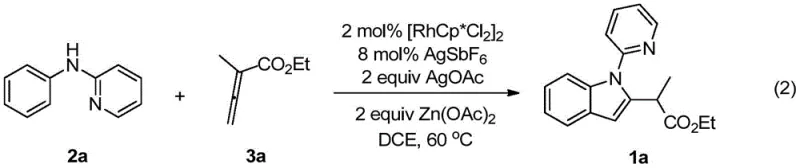
Furthermore, the versatility of this catalytic system is evidenced by its broad substrate scope, accommodating various electronic and steric environments on both the aniline and allenoate components. The use of silver hexafluoroantimonate as an additive plays a critical role in generating the cationic rhodium species necessary for efficient C-H cleavage, while silver acetate serves as the terminal oxidant to close the catalytic cycle. The inclusion of zinc acetate as an additive further enhances the reaction efficiency, likely by assisting in the deprotonation step or stabilizing transition states. This robust mechanistic framework allows for the synthesis of a wide library of 2-(2-indolyl)-acetate derivatives with diverse R-groups, including alkyl, benzyl, and allyl substituents, without compromising yield or selectivity. Such mechanistic reliability ensures that the process can be reliably transferred from laboratory discovery to commercial scale-up of complex pharmaceutical intermediates, providing a solid foundation for process chemistry teams.
How to Synthesize 2-(2-indolyl)-acetate Derivatives Efficiently
The practical implementation of this rhodium-catalyzed protocol is designed for operational simplicity, making it highly attractive for process development teams aiming to establish robust manufacturing workflows. The general procedure involves charging a reaction vessel with the N-phenyl-2-aminopyridine substrate, the dimeric rhodium catalyst precursor, and the requisite silver and zinc additives under an inert nitrogen atmosphere to prevent catalyst deactivation. Once the solid reagents are suspended in 1,2-dichloroethane, the allenoate coupling partner is introduced, and the mixture is heated to a moderate temperature of 60°C for approximately 24 hours to ensure complete conversion. Following the reaction period, the workup procedure is straightforward, involving the removal of volatiles under reduced pressure and purification via standard silica gel column chromatography to isolate the target indole derivative in high purity.
- Charge a reaction vessel with N-phenyl-2-aminopyridine, [RhCp*Cl2]2 catalyst, AgSbF6 additive, AgOAc oxidant, and Zn(OAc)2 additive under nitrogen.
- Add 1,2-dichloroethane (DCE) solvent and the allenoate coupling partner, then heat the mixture to 60°C for 24 hours.
- Upon completion, remove solvent via rotary evaporation and purify the crude residue using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this C-H activation technology offers compelling strategic advantages that extend beyond mere technical novelty, directly impacting the bottom line and operational resilience. The elimination of pre-functionalization steps translates to a significant reduction in raw material costs and processing time, as expensive halogenated precursors or specialized hydrazines are no longer required. This streamlined synthesis reduces the overall inventory burden and simplifies the sourcing strategy, as the starting materials like N-phenyl-2-aminopyridine and allenoates are commercially available and easy to prepare in large quantities. Moreover, the mild reaction conditions mitigate the need for specialized high-pressure or high-temperature equipment, lowering capital expenditure requirements for production facilities and enhancing workplace safety profiles. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market demands for complex heterocyclic building blocks.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in atom economy achieved by directly utilizing C-H bonds rather than relying on leaving groups. By avoiding the generation of stoichiometric salt waste associated with traditional substitution reactions, the process significantly lowers waste disposal costs and environmental compliance burdens. Additionally, the high catalytic efficiency of the rhodium system means that precious metal loading can be minimized while maintaining high turnover numbers, further reducing the cost of goods sold. The ability to produce diverse derivatives from a common set of starting materials also allows for economies of scale in purchasing, driving down unit costs across the entire product portfolio.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials ensures a consistent and reliable supply of critical intermediates, reducing the risk of production delays caused by raw material shortages. The robustness of the reaction conditions means that the process is less susceptible to variations in utility quality or minor operational deviations, leading to more predictable batch cycles and on-time delivery performance. Furthermore, the simplified purification workflow reduces the dependency on complex separation technologies, minimizing potential bottlenecks in the downstream processing stages. This reliability is paramount for maintaining continuous manufacturing operations and meeting the stringent delivery schedules demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with increasingly stringent regulatory standards by minimizing the use of hazardous reagents and reducing the overall carbon footprint of the synthesis. The absence of harsh acidic or basic conditions simplifies effluent treatment processes, allowing for easier compliance with local environmental regulations and reducing the cost of wastewater management. The scalability of the process is supported by the use of common organic solvents and standard heating equipment, facilitating a smooth transition from gram-scale laboratory experiments to multi-kilogram pilot runs and eventual commercial production. This seamless scalability ensures that supply can be ramped up quickly to meet surging demand without compromising product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed indole synthesis technology, providing clarity for stakeholders evaluating its potential integration into their supply chains. These insights are derived directly from the experimental data and scope defined within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this advanced methodology for specific project requirements and long-term strategic planning.
Q: What are the primary advantages of this Rh-catalyzed method over traditional Fischer indole synthesis?
A: Unlike traditional methods requiring harsh acidic conditions and pre-functionalized hydrazines, this Rh-catalyzed C-H activation operates under mild conditions (60°C) with high atom economy, eliminating the need for substrate pre-functionalization and reducing waste generation.
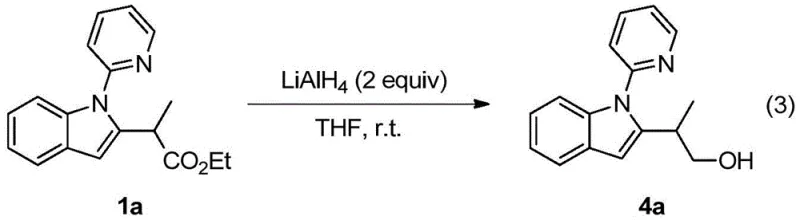
Q: Can the resulting 2-(2-indolyl)-acetate derivatives be further functionalized?
A: Yes, the ester group on the side chain allows for versatile downstream transformations, such as reduction to primary alcohols using LiAlH4, enabling the synthesis of diverse bioactive scaffolds.
Q: Is this process suitable for large-scale commercial production?
A: The process utilizes readily available starting materials and standard separation techniques like column chromatography, making it highly amenable to scale-up for industrial pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-indolyl)-acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the landscape of pharmaceutical intermediate production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 2-(2-indolyl)-acetate derivatives we produce. We are committed to delivering high-quality chemical solutions that empower our clients to accelerate their drug development timelines while maintaining the highest standards of safety and efficacy.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge synthetic route for their specific pipeline needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating how this efficient methodology can optimize your manufacturing budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to drive innovation and efficiency in your chemical supply chain.
