Advanced Rhodium-Catalyzed C-H Activation for Scalable Indole Derivative Manufacturing
Introduction to Next-Generation Indole Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and versatile synthetic methodologies. A significant breakthrough in this domain is detailed in Chinese Patent CN111285846B, which discloses a novel synthetic route for 2-(2-indolyl)-acetate derivatives. This technology leverages advanced transition metal catalysis to construct the indole core, a privileged scaffold found in countless bioactive molecules ranging from cholesterol-lowering agents like Fluvastatin to anticancer compounds such as Dictyodendrin B. By utilizing easily prepared N-phenyl-2-aminopyridine as a starting material, this method achieves ortho-position C-H bond activation under the influence of a rhodium salt, followed by coupling with allenoic acid esters. 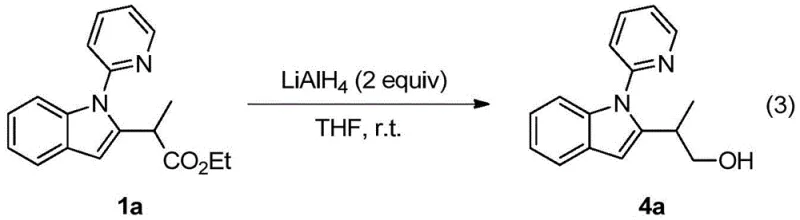 This approach not only streamlines the synthesis of complex indole structures but also offers exceptional functional group tolerance, allowing for the creation of diverse molecular architectures without the need for tedious pre-functionalization steps. For R&D directors and procurement specialists alike, this represents a pivotal shift towards atom-economical processes that reduce both chemical waste and overall production costs.
This approach not only streamlines the synthesis of complex indole structures but also offers exceptional functional group tolerance, allowing for the creation of diverse molecular architectures without the need for tedious pre-functionalization steps. For R&D directors and procurement specialists alike, this represents a pivotal shift towards atom-economical processes that reduce both chemical waste and overall production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole ring has relied heavily on classical named reactions such as the Fischer, Madelung, and Bischler indole syntheses. While these methods have served the industry well for decades, they are increasingly viewed as suboptimal for modern drug discovery and process chemistry. The Fischer indole synthesis, for instance, typically requires strongly acidic conditions and specific phenylhydrazine substrates, which can be hazardous to handle and limit the scope of compatible functional groups. Furthermore, these traditional routes often suffer from poor regioselectivity and low atom economy, generating substantial amounts of stoichiometric byproducts that complicate purification and increase environmental burden. In many cases, the substrates required for these cyclizations are not readily available or require multi-step preparation themselves, thereby elongating the supply chain and increasing the lead time for critical API intermediates. The rigidity of these older methods often forces chemists to design synthetic routes around the limitations of the cyclization step rather than optimizing for the final target molecule.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes a direct C-H activation strategy that fundamentally changes the retrosynthetic logic for indole derivatives. By employing a rhodium catalyst to activate the inert C-H bond at the ortho-position of N-phenyl-2-aminopyridine, the process bypasses the need for pre-halogenated or pre-functionalized precursors. 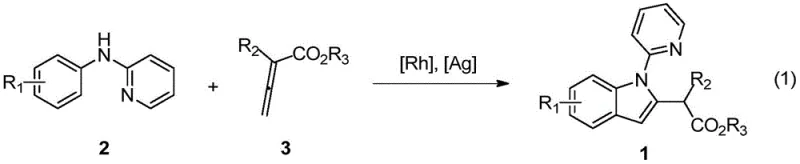 This direct coupling with allenoic acid esters proceeds under remarkably mild conditions, typically around 60°C, which preserves sensitive functional groups that would otherwise decompose under the harsh thermal or acidic conditions of classical methods. The reaction exhibits excellent regioselectivity, ensuring that the indole ring forms precisely at the desired position without generating difficult-to-separate isomers. This level of precision is crucial for maintaining high purity standards in pharmaceutical manufacturing. Moreover, the use of allenoates as coupling partners introduces a unique structural diversity, allowing for the rapid generation of libraries of 2-substituted indoles that are valuable for structure-activity relationship (SAR) studies.
This direct coupling with allenoic acid esters proceeds under remarkably mild conditions, typically around 60°C, which preserves sensitive functional groups that would otherwise decompose under the harsh thermal or acidic conditions of classical methods. The reaction exhibits excellent regioselectivity, ensuring that the indole ring forms precisely at the desired position without generating difficult-to-separate isomers. This level of precision is crucial for maintaining high purity standards in pharmaceutical manufacturing. Moreover, the use of allenoates as coupling partners introduces a unique structural diversity, allowing for the rapid generation of libraries of 2-substituted indoles that are valuable for structure-activity relationship (SAR) studies.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the rhodium complex, specifically the dimer [RhCp*Cl2]2. The mechanism initiates with the coordination of the pyridine nitrogen atom to the rhodium center, acting as a directing group to facilitate the activation of the adjacent aromatic C-H bond. This step forms a stable five-membered rhodacycle intermediate, a critical species that lowers the activation energy for the subsequent bond-forming events. The presence of silver hexafluoroantimonate (AgSbF6) serves as a halide scavenger, generating the cationic rhodium species necessary for high catalytic activity. Once the C-H bond is activated, the allenoate substrate coordinates to the metal center and undergoes migratory insertion into the Rh-C bond. This insertion step is highly regioselective, dictated by the electronic and steric properties of the allenoate, ensuring the formation of the correct carbon-carbon bond connectivity. Following insertion, reductive elimination or protonolysis releases the cyclized indole product and regenerates the active rhodium catalyst, completing the cycle. The addition of zinc acetate as an additive further enhances the reaction efficiency, likely by assisting in the proton transfer steps or stabilizing key intermediates within the catalytic manifold.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The concerted nature of the organometallic steps minimizes the formation of random polymeric byproducts or decomposition fragments often seen in free-radical processes. The mild reaction temperature of 60°C significantly reduces the risk of thermal degradation of the starting materials or the product, leading to cleaner reaction profiles. Furthermore, the high specificity of the directing group strategy ensures that C-H activation occurs exclusively at the ortho-position relative to the amino-pyridine moiety, preventing side reactions on other parts of the molecule. This inherent selectivity simplifies the downstream purification process, often allowing for straightforward silica gel chromatography to achieve high-purity isolates. For process chemists, understanding this mechanistic pathway is vital for troubleshooting and optimizing the reaction on a larger scale, ensuring consistent quality and yield.
How to Synthesize 2-(2-indolyl)-acetate Efficiently
The practical implementation of this synthesis is designed to be robust and accessible for laboratory and pilot-scale operations. The protocol involves combining the N-phenyl-2-aminopyridine substrate with the rhodium catalyst system in a standard organic solvent, followed by the addition of the allenoate coupling partner. The reaction is heated to a moderate temperature, allowing the catalytic cycle to proceed to completion over a defined period. Detailed operational parameters, including precise molar ratios of the silver oxidant and zinc additive, are critical for maximizing yield. 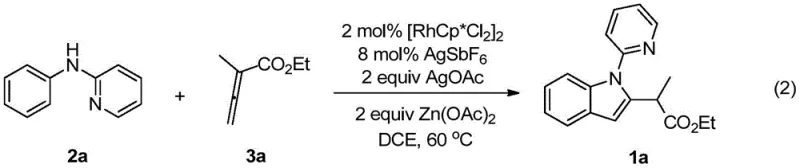 For those seeking to replicate or adapt this chemistry, the standardized synthetic steps provided in the technical documentation below offer a clear roadmap for achieving optimal results.
For those seeking to replicate or adapt this chemistry, the standardized synthetic steps provided in the technical documentation below offer a clear roadmap for achieving optimal results.
- Combine N-phenyl-2-aminopyridine, rhodium catalyst [RhCp*Cl2]2, silver hexafluoroantimonate, silver acetate, and zinc acetate in a reaction vessel under nitrogen.
- Add 1,2-dichloroethane solvent and the allenoate coupling partner, then heat the mixture to 60°C for 24 hours to facilitate C-H activation and cyclization.
- Upon completion, remove solvent via rotary evaporation and purify the crude residue using silica gel column chromatography to isolate the target indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rh-catalyzed C-H activation technology presents compelling economic and logistical benefits. The primary driver for cost reduction lies in the simplification of the raw material supply chain. Since the method does not require pre-functionalized substrates such as halogenated anilines or specialized hydrazines, manufacturers can source cheaper, commodity-grade starting materials like N-phenyl-2-aminopyridine. This elimination of upstream synthetic steps directly translates to a lower cost of goods sold (COGS). Additionally, the high atom economy of the reaction means that a greater proportion of the input mass is converted into the final product, reducing the volume of chemical waste that requires expensive disposal. The mild reaction conditions also imply lower energy consumption compared to high-temperature reflux processes, contributing to a smaller carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the streamlined synthetic sequence. By removing the need for pre-functionalization, the total number of unit operations is reduced, which decreases labor costs, solvent usage, and equipment occupancy time. The use of a highly active rhodium catalyst, while potentially expensive per gram, is employed in low loading (typically around 2 mol%), and its efficiency in driving the reaction to high conversion offsets the initial catalyst cost. Furthermore, the ability to use simple allenoates instead of complex alkynes or halides opens up a wider market of affordable reagents. The overall process intensity is lower, meaning less capital expenditure is required for reactors capable of withstanding extreme pressures or temperatures, making it an attractive option for contract manufacturing organizations looking to optimize their asset utilization.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the reliance on broadly available chemical building blocks. N-phenyl-2-aminopyridines are stable, easy-to-handle solids that can be sourced from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions—tolerating a range of functional groups and operating at moderate temperatures—ensures consistent batch-to-batch reproducibility, which is critical for maintaining steady production schedules. Unlike sensitive organometallic reagents that require cryogenic storage or inert atmosphere handling throughout the entire process, this method only requires standard nitrogen protection during the reaction, simplifying logistics and storage requirements. This reliability allows for better inventory planning and reduces the likelihood of production delays caused by raw material shortages or quality deviations.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram or ton scale is facilitated by the homogeneous nature of the catalytic system and the use of common industrial solvents like 1,2-dichloroethane. The reaction does not generate hazardous gases or require exotic reagents that pose significant safety risks upon scale-up. From an environmental compliance standpoint, the atom-economic nature of the transformation aligns well with green chemistry principles, reducing the E-factor (mass of waste per mass of product). The simplified workup procedure, involving standard solvent removal and chromatography, minimizes the generation of aqueous waste streams contaminated with heavy metals or strong acids. This makes the process easier to permit and operate within strict regulatory frameworks, ensuring long-term sustainability for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indole synthesis technology. These insights are derived directly from the experimental data and scope outlined in the patent literature, providing a realistic view of the method's capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing production pipelines.
Q: What are the primary advantages of this Rh-catalyzed method over traditional indole synthesis?
A: Unlike traditional methods like Fischer indole synthesis which often require harsh acidic conditions and specific hydrazine substrates, this Rh-catalyzed C-H activation operates under mild conditions (60°C) with high atom economy. It eliminates the need for pre-functionalized substrates, significantly simplifying the synthetic route and reducing waste generation.
Q: Can the resulting 2-(2-indolyl)-acetate derivatives be further functionalized?
A: Yes, the patent explicitly demonstrates that the ester group in the product can be reduced to alcohols using LiAlH4, indicating high versatility for downstream derivatization. This allows medicinal chemists to access a wide library of functionalized indole scaffolds essential for drug discovery programs.
Q: Is this process suitable for large-scale commercial production?
A: The process utilizes commercially available starting materials like N-phenyl-2-aminopyridine and allenoates, and employs standard solvents like 1,2-dichloroethane. The mild reaction temperature and robust catalyst system suggest good potential for scale-up, provided appropriate safety measures for silver salts and rhodium recovery are implemented.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-indolyl)-acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this rhodium-catalyzed cyclization can be successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-(2-indolyl)-acetate derivative meets the highest international standards. Our commitment to quality assurance means that we can deliver high-purity pharmaceutical intermediates that are ready for immediate use in downstream API synthesis.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage potential clients to contact us directly to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. Let us help you optimize your supply chain and reduce time-to-market with our reliable, scalable, and cost-effective manufacturing solutions.
