Scalable Synthesis of Bempedoic Acid Intermediates via Palladium-Catalyzed Cross-Coupling
The pharmaceutical landscape for cholesterol management has shifted significantly with the approval of Bempedoic acid, an ATP Citrate Lyase (ACL) inhibitor that offers a crucial alternative for patients intolerant to statins. Patent CN111285760B introduces a transformative synthesis method that addresses the longstanding manufacturing bottlenecks associated with this high-value active pharmaceutical ingredient. Unlike traditional routes that rely on cumbersome purification techniques, this novel approach leverages palladium-catalyzed cross-coupling reactions to construct the carbon skeleton with precision. The technical breakthrough lies in the strategic use of a boronic ester intermediate, which facilitates a robust Suzuki-Miyaura coupling under mild conditions. This innovation not only streamlines the synthetic pathway but also fundamentally alters the economic and operational feasibility of producing this complex dicarboxylic acid derivative at an industrial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in patent WO2004067489, presented severe challenges for commercial manufacturing, primarily due to their reliance on physically demanding purification steps. The conventional process necessitated reduced pressure distillation for the initial purification, requiring the handling of products with extremely high boiling points exceeding 230°C, which poses significant energy and safety risks in a plant environment. Furthermore, the subsequent purification steps depended heavily on column chromatography, a technique that is notoriously difficult to scale, consumes vast quantities of solvents, and generates substantial hazardous waste. These inefficiencies resulted in a final product purity of merely 83.8%, which is often insufficient for stringent pharmaceutical applications without further costly reprocessing. The combination of high thermal stress, solvent intensity, and low yield made the traditional route economically unviable for reliable high-purity API intermediate supply chains.
The Novel Approach
The methodology outlined in CN111285760B circumvents these thermal and chromatographic hurdles by introducing a convergent synthesis strategy centered on organoboron chemistry. By converting a bromo-ester precursor into a pinacol boronic ester (Compound I), the process enables a highly selective carbon-carbon bond formation with a dibromo-ketone chain. This Suzuki coupling step proceeds efficiently at moderate temperatures around 60°C, avoiding the thermal degradation risks associated with high-temperature distillation. Crucially, the intermediates and the final product can be isolated through simple crystallization using common solvent systems like ethyl acetate, petroleum ether, or methyl tert-butyl ether. This shift from chromatography to crystallization represents a paradigm shift in process chemistry, drastically simplifying the workflow and enabling the achievement of purities exceeding 98% directly from the reactor, thereby enhancing cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Pd-Catalyzed Suzuki-Miyaura Coupling
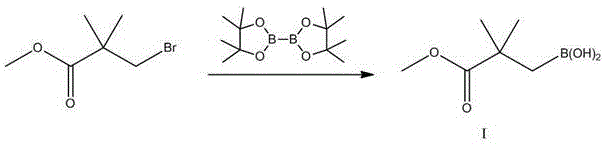
The core of this synthetic advancement is the two-stage palladium-catalyzed sequence that constructs the central carbon framework of Bempedoic acid. The first stage involves the borylation of methyl 3-bromo-2,2-dimethylpropionate using bis(pinacolato)diboron in the presence of a palladium catalyst such as Pd(OAc)2 and a base like potassium acetate. This transformation generates the key nucleophilic species, Compound I, which is stable and易于 handle. The second stage is the cross-coupling of this boronic ester with 5-keto-1,9-dibromononane. In this step, the palladium catalyst, often Pd(dppf)Cl2, facilitates the oxidative addition into the carbon-bromine bond of the ketone chain, followed by transmetallation with the boron species and reductive elimination to forge the new C-C bond. This mechanism is highly tolerant of the ketone functionality present in the chain, showcasing the chemoselectivity required for complex molecule synthesis.
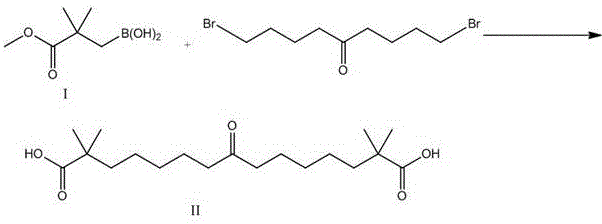
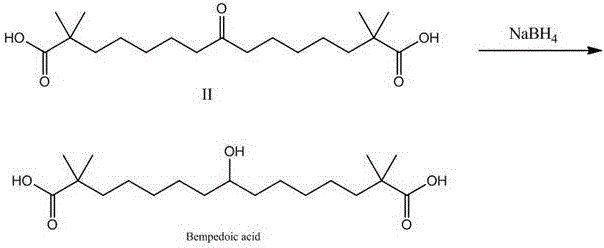
Following the coupling reaction, the process incorporates a hydrolysis step to reveal the carboxylic acid functionalities, yielding Compound II. The final transformation involves the stereoselective reduction of the central ketone group to a secondary alcohol using sodium borohydride in methanol at 0°C. This mild reducing agent ensures that the ester groups (which are subsequently hydrolyzed or managed) and the sensitive carbon backbone remain intact while selectively targeting the carbonyl. The entire mechanistic pathway is designed to minimize side reactions and impurity formation, which is critical for meeting the rigorous impurity profiles demanded by regulatory bodies for cardiovascular drugs. The ability to control the reaction environment through nitrogen protection and precise temperature control further underscores the robustness of this catalytic cycle.
How to Synthesize Bempedoic Acid Efficiently
The synthesis of Bempedoic acid via this patented route involves a logical sequence of borylation, coupling, and reduction that can be adapted for pilot and commercial scales. The process begins with the preparation of the boronic ester building block, followed by its coupling with the central ketone chain, and concludes with a straightforward reduction and crystallization. Detailed standard operating procedures regarding reagent stoichiometry, solvent volumes, and specific work-up protocols are essential for reproducibility. For a comprehensive guide on the exact molar ratios and temperature profiles required to maximize yield and purity, please refer to the standardized synthesis steps provided below.
- Prepare Compound I by reacting methyl 3-bromo-2,2-dimethylpropionate with bis-pinacol borate using a palladium catalyst.
- Synthesize Compound II by coupling Compound I with 5-keto-1,9-dibromononane under basic conditions followed by hydrolysis.
- Reduce Compound II using sodium borohydride in methanol to obtain the final Bempedoic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this crystallization-based synthesis route offers profound strategic advantages over legacy methods. The elimination of column chromatography is not merely a technical improvement; it is a major economic driver that removes one of the most expensive and time-consuming unit operations in fine chemical manufacturing. By relying on crystallization, the process significantly reduces solvent consumption and waste disposal costs, leading to substantial cost savings in the overall production budget. Furthermore, the avoidance of high-temperature vacuum distillation reduces energy consumption and equipment maintenance requirements, contributing to a lower total cost of ownership for the manufacturing facility. These efficiencies translate directly into a more competitive pricing structure for the final API intermediate.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps eliminates the need for large columns of silica gel and the associated massive volumes of elution solvents, which are major cost centers in traditional synthesis. Additionally, the use of standard palladium catalysts and common bases like potassium acetate ensures that raw material costs remain predictable and manageable. The high yield and purity achieved in each step mean that less starting material is wasted, optimizing the atom economy of the process. Consequently, manufacturers can achieve a leaner production model with significantly reduced operational expenditures compared to distillation-heavy routes.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as bis-pinacol borate and various palladium catalysts, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate moderate temperatures and standard atmospheric pressures (under nitrogen), allows for production in a wider range of facilities without needing specialized high-vacuum or high-heat infrastructure. This flexibility ensures consistent production schedules and reduces the likelihood of delays caused by equipment failure or complex process upsets, thereby securing a steady flow of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling a process that relies on crystallization is inherently safer and more predictable than scaling one dependent on distillation or chromatography, as heat transfer and mass transfer issues are easier to manage in crystallizers. The reduction in solvent waste and the absence of silica gel disposal align with modern green chemistry principles and environmental regulations, simplifying the permitting and compliance process for new manufacturing lines. This environmental compatibility future-proofs the supply chain against tightening ecological standards, ensuring long-term viability for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the practical aspects of adopting this technology. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing production capabilities.
Q: How does this new synthesis method improve upon prior art like WO2004067489?
A: The new method eliminates the need for high-temperature vacuum distillation and column chromatography, which were bottlenecks in previous processes, resulting in significantly higher purity (up to 98.6%) and better industrial feasibility.
Q: What are the key intermediates in this Bempedoic acid synthesis route?
A: The process relies on two critical intermediates: Compound I (a boronic ester derivative) and Compound II (a dicarboxylic acid precursor formed via Suzuki coupling), both of which can be purified via crystallization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reliance on crystallization rather than chromatography for purification makes the process highly scalable and cost-effective for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bempedoic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN111285760B into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Suzuki coupling route are fully realized in practice. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the utmost safety and precision, supported by rigorous QC labs that enforce stringent purity specifications to meet global pharmacopoeia standards. We understand that consistency is key in the pharmaceutical supply chain, and our commitment to quality assurance guarantees that every batch of Bempedoic acid intermediate meets the highest benchmarks for potency and impurity control.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this efficient synthesis technology for their cholesterol-lowering drug portfolios. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can accelerate your time-to-market while maximizing value.
