Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Value Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral building blocks essential for modern drug discovery. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in the availability of these complex scaffolds. Historically, the synthesis of axially chiral styrenes has been constrained by limited methodologies, often relying on harsh conditions or scarce starting materials. This innovation leverages a chiral phosphoric acid catalytic system to achieve efficient kinetic resolution, delivering products with exceptional optical purity. For R&D teams focused on developing novel ligands or organocatalysts, this technology offers a robust pathway to access structural diversity that was previously difficult to obtain. The ability to generate these compounds with high enantiomeric excess opens new avenues for creating next-generation catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for constructing axially chiral styrene skeletons have predominantly relied on transition metal catalysis, such as palladium-catalyzed allylation or titanium-mediated additions. These conventional routes often suffer from significant drawbacks, including the requirement for strict anhydrous conditions, sensitivity to air and moisture, and the inherent toxicity associated with heavy metal residues. Furthermore, the substrate scope in many metal-catalyzed processes is frequently narrow, limiting the structural variety of the resulting chiral molecules. The reliance on expensive metal catalysts also introduces substantial cost burdens and complicates the purification process, as removing trace metal contaminants to meet pharmaceutical standards can be technically challenging and resource-intensive. Consequently, there has been a persistent industry need for a more sustainable and versatile synthetic approach.
The Novel Approach
The methodology disclosed in CN111848322B represents a paradigm shift by utilizing organocatalysis to resolve racemic mixtures efficiently. By employing a chiral phosphoric acid catalyst in dichloromethane at mild temperatures ranging from 0°C to 10°C, this process achieves high levels of stereocontrol without the need for transition metals. The reaction utilizes readily available racemic precursors and operates under ambient pressure, significantly simplifying the operational complexity. This approach not only enhances the safety profile of the synthesis but also broadens the applicability to a wide range of substrates with varying electronic and steric properties. The result is a streamlined process that delivers axially chiral oxindole-substituted styrenes with yields reaching up to 54% and enantiomeric excess values as high as 96%, demonstrating superior efficiency compared to legacy methods.
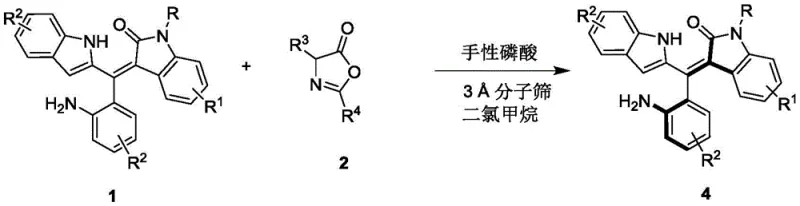
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
The success of this resolution strategy lies in the precise activation mode of the chiral phosphoric acid catalyst. The catalyst functions through a dual hydrogen-bonding network, simultaneously activating the electrophilic and nucleophilic components of the reaction mixture. This bifunctional activation creates a highly organized chiral environment that differentiates between the enantiomers of the racemic starting material. The bulky substituents on the binaphthyl backbone of the catalyst, such as the 2-naphthyl groups, provide the necessary steric hindrance to enforce strict facial selectivity during the bond-forming event. This mechanistic feature ensures that one enantiomer reacts significantly faster than the other, leading to the accumulation of the desired axially chiral product with high optical purity. Understanding this interaction is crucial for scaling the process, as it highlights the importance of maintaining precise stoichiometric ratios and temperature control to preserve the integrity of the chiral pocket.
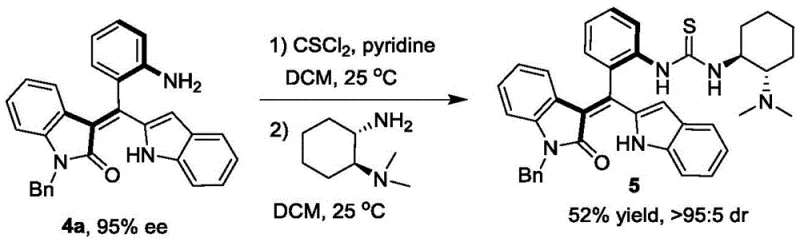
Beyond the primary resolution, the utility of the resulting compounds is further amplified through derivatization. The resolved axially chiral styrenes can be transformed into chiral tertiary amine catalysts, which exhibit remarkable performance in downstream asymmetric transformations. For instance, the derived catalysts have been shown to facilitate the asymmetric [4+2] cyclization of o-methylenebenzoquinones with malononitrile, achieving yields of 90% and 91% ee. This cascade of functionality demonstrates that the initial resolution step is not merely an endpoint but a gateway to generating high-value chiral tools. The robustness of the chiral axis in these molecules ensures that the stereochemical information is retained throughout subsequent synthetic manipulations, making them ideal candidates for complex multi-step syntheses in medicinal chemistry.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for producing these valuable intermediates. The process begins with the preparation of the reaction mixture containing the racemic substrate and the specific chiral phosphoric acid catalyst in an appropriate solvent system. Strict adherence to the specified molar ratios and reaction temperatures is critical to maximizing the enantiomeric excess of the final product. Following the reaction period, a straightforward workup procedure involving filtration and concentration allows for the isolation of the crude material. Detailed standard operating procedures for the purification and characterization steps are essential for ensuring consistent quality across different batches. For comprehensive technical details regarding reagent grades and specific equipment requirements, please refer to the standardized synthesis guide below.
- Combine racemic Formula 1 compound and Formula 2 compound in dichloromethane solvent with 3A molecular sieves.
- Add 10 mol% chiral phosphoric acid catalyst (Formula 3) and stir at 0°C to 10°C until TLC indicates completion.
- Filter off molecular sieves, concentrate the filtrate, and purify via silica gel column chromatography to isolate the axially chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this organocatalytic resolution technology offers distinct strategic advantages over traditional metal-dependent syntheses. The elimination of precious metal catalysts such as palladium or titanium removes a major variable from the cost structure, mitigating the risks associated with volatile metal prices and supply shortages. Additionally, the mild reaction conditions reduce energy consumption and minimize the need for specialized high-pressure or cryogenic equipment, leading to significant operational expenditure savings. The simplicity of the workup procedure, which avoids complex metal scavenging steps, further accelerates the production timeline and reduces waste generation. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with organocatalysts drastically lowers the raw material costs associated with the synthesis. Furthermore, the removal of heavy metal clearance steps simplifies the downstream processing, reducing the consumption of specialized resins and solvents required for purification. This streamlined workflow translates to substantial cost savings in large-scale manufacturing environments, making the production of these complex chiral intermediates economically viable for broader applications.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable organic reagents ensures a consistent supply of raw materials, unaffected by the geopolitical instabilities that often impact the mining and refining of rare earth metals. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in environmental parameters. This reliability is critical for maintaining continuous production lines and meeting the stringent delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The use of dichloromethane as a solvent, combined with the absence of toxic metal waste, simplifies the environmental compliance burden associated with waste disposal. The reaction's high atom economy and selectivity minimize the formation of byproducts, reducing the overall volume of chemical waste generated per kilogram of product. These green chemistry attributes facilitate easier regulatory approval and support sustainability goals, positioning the technology favorably for future scale-up initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this resolution technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation. Understanding these nuances is vital for process chemists evaluating the feasibility of integrating this method into existing workflows. The answers provided reflect the current state of the art as described in the intellectual property.
Q: What is the primary advantage of this resolution method over traditional metal-catalyzed couplings?
A: This organocatalytic method eliminates the need for expensive and toxic transition metals like palladium or titanium, operating under much milder conditions (0°C) while achieving high enantiomeric excess (up to 96% ee).
Q: Can the resolved styrene compounds be further functionalized?
A: Yes, the resolved axially chiral styrenes (Formula 4) serve as precursors for chiral tertiary amine catalysts (Formula 5), which demonstrate superior performance in asymmetric [4+2] cyclization reactions compared to commercial alternatives.
Q: What represents the optimal reaction temperature for this resolution process?
A: The patent data indicates that a reaction temperature of 0°C is preferred to maximize enantioselectivity and yield, although the process remains effective within a range of 0°C to 10°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral intermediates play in accelerating drug discovery pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN111848322B can be seamlessly transitioned from the laboratory to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of axially chiral oxindole-substituted styrene meets the highest international standards. Our commitment to quality assurance ensures that our partners receive materials that are ready for immediate use in sensitive catalytic applications.
We invite you to collaborate with us to explore the full potential of this resolution technology for your specific project needs. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements and process constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your supply chain.
