Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Purity Pharmaceutical Intermediates
Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Purity Pharmaceutical Intermediates
The landscape of asymmetric synthesis is continuously evolving to meet the stringent demands of the pharmaceutical industry for high-purity chiral intermediates. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in current synthetic capabilities. This technology leverages a sophisticated organocatalytic system based on chiral phosphoric acids to achieve exceptional enantioselectivity under remarkably mild conditions. For R&D directors and process chemists, this represents a pivotal shift away from traditional transition metal-dependent routes, offering a cleaner, more sustainable pathway to complex chiral scaffolds. The ability to access these structurally diverse molecules with high optical purity opens new avenues for the development of next-generation therapeutic agents and advanced catalytic ligands.
Furthermore, the versatility of this resolution strategy extends beyond simple isolation, as the resulting axially chiral products serve as precursors for potent chiral tertiary amine catalysts. This dual utility enhances the value proposition for procurement teams seeking multifunctional intermediates that can streamline supply chains. By establishing a robust protocol for generating these challenging stereogenic axes, the patent provides a reliable foundation for scaling up the production of high-value fine chemicals. The integration of this technology into existing manufacturing workflows promises to reduce reliance on scarce metal resources while maintaining rigorous quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has been fraught with significant synthetic challenges, primarily relying on nucleophilic additions to alkynes or cross-coupling reactions involving aryl halides. These conventional pathways often necessitate the use of expensive and toxic transition metal catalysts, such as palladium or titanium complexes, which introduce substantial complications regarding metal residue removal and environmental disposal. Moreover, the ligand systems required to induce chirality in these metal-catalyzed processes are frequently complex to synthesize and sensitive to air and moisture, leading to inconsistent batch-to-batch reproducibility. The harsh reaction conditions typically associated with these methods, including elevated temperatures and strong bases, can also limit substrate scope and lead to the degradation of sensitive functional groups, thereby reducing overall process efficiency and yield.
The Novel Approach
In stark contrast, the methodology disclosed in CN111848322B employs a metal-free organocatalytic resolution strategy that operates under exceptionally mild conditions, typically between 0°C and 10°C in dichloromethane. This approach utilizes readily available chiral phosphoric acid derivatives, such as binaphthyl-based catalysts, to differentiate between enantiomers of the racemic starting material through a highly organized hydrogen-bonding network. The reaction proceeds with high atom economy and avoids the generation of hazardous heavy metal waste, aligning perfectly with green chemistry principles. As illustrated in the reaction scheme below, the transformation of racemic Formula 1 and azlactone Formula 2 into the axially chiral Formula 4 is achieved with remarkable precision, offering a streamlined alternative to legacy synthesis routes.
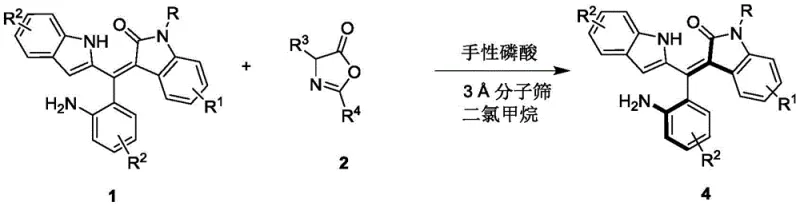
This novel route not only simplifies the operational complexity but also significantly broadens the substrate scope, accommodating various substituents on the aromatic rings without compromising stereocontrol. The use of 3A molecular sieves as an additive further enhances reaction efficiency by sequestering water, ensuring the catalyst remains active throughout the process. For supply chain managers, this translates to a more robust and predictable manufacturing process that is less susceptible to the fluctuations in metal catalyst availability and pricing. The ability to produce these complex chiral architectures in fewer steps with higher purity directly contributes to cost reduction in pharmaceutical intermediate manufacturing and accelerates the timeline from discovery to commercial production.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
The core of this technological advancement lies in the precise mechanistic interaction between the chiral phosphoric acid catalyst and the substrate molecules. The catalyst, typically a binaphthyl-derived phosphoric acid with bulky substituents like 2-naphthyl groups, acts as a bifunctional activator. It simultaneously activates the electrophilic azlactone through hydrogen bonding with the carbonyl oxygen and stabilizes the nucleophilic attack via the acidic proton of the phosphate group. This dual activation creates a rigid chiral environment that dictates the trajectory of the incoming nucleophile, effectively discriminating between the pro-chiral faces of the intermediate species. The steric bulk of the catalyst's backbone prevents unfavorable interactions, forcing the reaction to proceed through a single, energetically favored transition state that leads to the desired axial chirality.
Impurity control is inherently built into this mechanism due to the high specificity of the organocatalyst. Unlike metal catalysts which may promote side reactions such as homocoupling or over-reduction, the chiral phosphoric acid selectively facilitates the desired addition-elimination pathway. The mild reaction temperature of 0°C further suppresses thermal degradation and non-selective background reactions, ensuring that the final product profile is clean and dominated by the target enantiomer. Post-reaction purification is straightforward, often requiring only standard silica gel chromatography, as the catalyst and byproducts are easily separated from the highly lipophilic product. This level of control is critical for R&D teams aiming to minimize impurity profiles in early-stage drug candidates, thereby reducing the regulatory burden during later clinical phases.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the preparation of a dry reaction vessel containing dichloromethane and activated 3A molecular sieves to ensure anhydrous conditions. The racemic oxindole precursor and the azlactone coupling partner are introduced in a specific molar ratio, typically ranging from 1:0.5 to 1:0.6, to optimize the kinetic resolution efficiency. Upon addition of the chiral phosphoric acid catalyst at a loading of 10 mol%, the mixture is stirred at a controlled temperature of 0°C, allowing the stereoselective transformation to proceed over several hours. Detailed standardized synthesis steps follow below.
- Prepare the reaction mixture by combining the racemic starting material (Formula 1) and the azlactone derivative (Formula 2) in dichloromethane solvent with 3A molecular sieves.
- Add the chiral phosphoric acid catalyst (Formula 3, preferably 10 mol%) to the mixture and stir at a controlled low temperature between 0°C and 10°C.
- Monitor the reaction progress via TLC until completion, then filter off the molecular sieves, concentrate the filtrate, and purify the resulting axially chiral product (Formula 4) using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic resolution technology offers distinct strategic advantages over traditional metal-catalyzed processes. The elimination of precious metals like palladium and titanium from the synthesis route removes a major source of cost volatility and supply risk, as these metals are subject to geopolitical fluctuations and mining constraints. Furthermore, the absence of heavy metals simplifies the downstream purification process, removing the need for expensive scavenging resins or complex extraction protocols designed to meet strict residual metal limits. This streamlining of the workflow results in substantial cost savings and a reduced environmental footprint, making the process more attractive for large-scale industrial applications where waste disposal costs are a significant factor.
- Cost Reduction in Manufacturing: The shift to organocatalysis drastically reduces raw material costs by replacing expensive transition metal complexes with more affordable chiral phosphoric acids. Additionally, the mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, leading to lower utility expenses. The simplified workup procedure, which avoids complex metal removal steps, further decreases processing time and labor costs, contributing to a more economically viable manufacturing model for high-value chiral intermediates.
- Enhanced Supply Chain Reliability: By relying on stable, commercially available organic catalysts and common solvents like dichloromethane, the supply chain becomes more resilient to disruptions. The robustness of the reaction conditions ensures consistent output quality regardless of minor variations in ambient conditions, reducing the rate of batch failures. This reliability allows for better inventory planning and shorter lead times for high-purity pharmaceutical intermediates, ensuring that downstream drug production schedules are met without delay.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable at low temperatures and the absence of hazardous reagents. The green chemistry profile, characterized by low toxicity and minimal waste generation, facilitates easier regulatory approval and compliance with increasingly stringent environmental regulations. This makes the technology suitable for commercial scale-up of complex pharmaceutical intermediates, supporting sustainable growth and long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What represents the primary advantage of this resolution method over traditional transition metal catalysis?
A: Unlike traditional methods relying on palladium or titanium catalysts which often require harsh conditions and complex ligand synthesis, this organocatalytic approach utilizes chiral phosphoric acids under mild temperatures (0-10°C), eliminating heavy metal contamination risks and simplifying purification protocols for pharmaceutical grade intermediates.
Q: Can the resolved axially chiral styrenes be further functionalized into active catalysts?
A: Yes, the patent demonstrates that the resolved compound (Formula 4a) can be efficiently derivatized into a chiral tertiary amine catalyst (Formula 5) through a thiourea linkage, which subsequently exhibits superior performance in asymmetric [4+2] cyclization reactions compared to commercial benchmarks.
Q: What is the typical enantiomeric excess (ee) achievable with this protocol?
A: The methodology consistently delivers high optical purity, with experimental data showing enantiomeric excess values reaching up to 96% ee across a diverse range of substrates, ensuring the production of high-purity chiral building blocks essential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral building blocks for the development of innovative therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in organocatalytic processes allows us to optimize the resolution of axially chiral oxindole-substituted styrenes, guaranteeing consistent supply and superior optical purity for your most demanding projects.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us support your journey towards more sustainable and efficient chemical manufacturing.
