Advanced Resolution Technology for Axially Chiral Oxindole Styrenes: Scalable Synthesis and Commercial Viability
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for accessing high-value chiral scaffolds, particularly those exhibiting axial chirality which are pivotal for advanced catalytic applications. Patent CN111848322B introduces a groundbreaking approach for the synthesis and resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in current organic synthesis literature. This technology leverages a highly efficient kinetic resolution strategy mediated by chiral phosphoric acid catalysts, enabling the transformation of readily available racemic mixtures into optically pure intermediates with exceptional stereocontrol. For R&D directors and process chemists, this represents a critical advancement, as it bypasses the limitations of traditional asymmetric synthesis which often requires complex chiral pool starting materials or expensive transition metal complexes. The ability to generate structural diversity while maintaining high enantiomeric excess positions this methodology as a cornerstone for developing next-generation chiral ligands and organocatalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has been fraught with synthetic challenges that hinder widespread industrial adoption. Conventional strategies predominantly rely on the nucleophilic addition to alkynes or the coupling of aryl halides with alkenes, processes that frequently demand harsh reaction conditions and specialized reagents. For instance, previous literature describes the use of L-proline for catalyzing nucleophilic additions or chiral palladium species for coupling reactions, yet these methods often suffer from narrow substrate scopes and moderate stereoselectivity. Furthermore, the reliance on transition metals introduces significant downstream processing burdens, including the necessity for rigorous metal scavenging to meet stringent pharmaceutical purity standards. These factors collectively escalate production costs and complicate supply chain logistics, making the reliable sourcing of high-purity axially chiral intermediates a persistent bottleneck for many manufacturing entities seeking cost reduction in chiral catalyst manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes an organocatalytic kinetic resolution that operates under remarkably mild and practical conditions. By employing a chiral phosphoric acid catalyst in dichloromethane at temperatures ranging from 0°C to 10°C, the process achieves excellent enantioselectivity without the need for cryogenic conditions or inert atmospheres typically associated with sensitive organometallic chemistry. The reaction tolerates a wide array of functional groups on both the styrene and azlactone components, allowing for the generation of structurally diverse products with high yields. This operational simplicity not only enhances safety profiles but also significantly streamlines the workflow, making it an ideal candidate for the commercial scale-up of complex chiral intermediates. The versatility of this approach ensures that manufacturers can access a broad library of chiral building blocks efficiently.
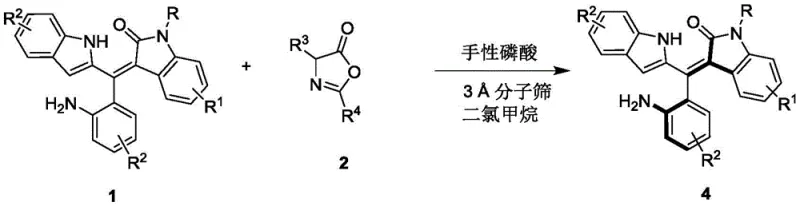
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
The success of this resolution strategy hinges on the precise molecular recognition facilitated by the chiral phosphoric acid catalyst, specifically derivatives based on binaphthyl, octahydrobinaphthyl, or spiro ring skeletons. These catalysts function through a dual hydrogen-bonding activation mode, where the acidic proton of the phosphate group activates the electrophilic azlactone while the basic phosphoryl oxygen interacts with the nucleophilic substrate. This cooperative activation creates a highly organized chiral environment that differentiates between the enantiomers of the racemic styrene substrate. The steric bulk introduced by substituents such as 2-naphthyl groups on the catalyst backbone further restricts the conformational freedom of the transition state, thereby enforcing high facial selectivity during the bond-forming event. Understanding this mechanistic nuance is vital for process optimization, as slight modifications to the catalyst structure can dramatically influence the enantiomeric outcome.

Impurity control is another critical aspect where this mechanistic understanding pays dividends. The high specificity of the chiral catalyst minimizes the formation of diastereomeric byproducts and homocoupling impurities that often plague less selective methods. By operating at low temperatures (preferably 0°C), the reaction kinetics are tuned to favor the desired enantiomer while suppressing non-selective background reactions. This inherent selectivity reduces the burden on downstream purification steps, such as silica gel column chromatography, leading to higher overall recovery of the target material. For quality assurance teams, this translates to a more consistent impurity profile and reduced risk of batch failure, ensuring that the final axially chiral products meet the rigorous specifications required for use in sensitive catalytic applications or as advanced pharmaceutical intermediates.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for executing this resolution on a laboratory or pilot scale. The procedure begins with the preparation of a reaction mixture containing the racemic styrene substrate and the azlactone reagent in dry dichloromethane, supplemented with 3A molecular sieves to maintain anhydrous conditions which are crucial for catalyst activity. The addition of the chiral phosphoric acid catalyst, typically at a loading of 10 mol%, initiates the kinetic resolution process. Maintaining the reaction temperature at 0°C is essential for maximizing enantioselectivity, as evidenced by the experimental data showing up to 96% ee under these conditions. Following the reaction monitoring via TLC, the workup involves a simple filtration to remove the molecular sieves, followed by concentration and purification. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety.
- Prepare the reaction mixture by combining the racemic styrene substrate (Formula 1) and the azlactone reagent (Formula 2) in dichloromethane solvent with 3A molecular sieves.
- Add the chiral phosphoric acid catalyst (Formula 3, preferably 10 mol%) to the mixture and stir at a controlled low temperature of 0°C to 10°C.
- Monitor the reaction progress via TLC until completion, then filter off the molecular sieves, concentrate the filtrate, and purify the resulting axially chiral product (Formula 4) using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial strategic benefits that extend beyond mere technical feasibility. The shift towards organocatalysis eliminates the dependency on scarce and volatile transition metals like palladium or titanium, which are subject to significant price fluctuations and supply chain disruptions. By utilizing robust organic catalysts that can be synthesized from abundant feedstocks, manufacturers can achieve significant cost savings and enhance supply chain reliability. Furthermore, the mild reaction conditions reduce energy consumption associated with heating or extreme cooling, contributing to a lower carbon footprint and aligning with modern sustainability goals. These factors collectively make the adoption of this resolution method a financially sound decision for long-term production planning.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated ligand systems directly lowers the raw material costs per kilogram of product. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and waste disposal costs. The high atom economy of the resolution process ensures that a significant portion of the starting material is converted into valuable chiral product, minimizing waste generation. These efficiencies cumulatively drive down the cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity axially chiral styrenes.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including the racemic styrenes and azlactones, are derived from commodity chemicals that are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and ensures continuity of supply even during market volatility. The stability of the chiral phosphoric acid catalysts also allows for easier storage and handling compared to air-sensitive metal complexes, reducing logistical complexities. Consequently, procurement managers can secure reliable long-term contracts with reduced risk of production delays caused by material shortages.
- Scalability and Environmental Compliance: The use of dichloromethane as a solvent, while requiring proper management, is a well-established practice in the industry with mature recovery and recycling infrastructure. The absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for extensive testing for residual metal contaminants in the final product. This facilitates faster regulatory approvals and market entry for downstream applications. Moreover, the process is inherently scalable, as the reaction parameters do not change significantly when moving from gram to kilogram scales, ensuring that reducing lead time for high-purity chiral ligands is achievable without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. They are designed to provide clarity on the operational parameters and potential applications of the axially chiral products. Understanding these details is essential for stakeholders evaluating the integration of this method into their existing manufacturing portfolios.
Q: What represents the primary advantage of this resolution method over traditional synthesis?
A: Unlike traditional methods relying on harsh nucleophilic additions or metal-catalyzed couplings which often suffer from limited substrate scope, this organocatalytic resolution operates under extremely mild conditions (0°C) and achieves high enantioselectivity (up to 96% ee) across a diverse range of substrates.
Q: Can the resolved styrene compounds be utilized for further catalytic applications?
A: Yes, the resolved axially chiral oxindole-substituted styrenes serve as versatile precursors. As demonstrated in the patent, they can be derivatized into chiral tertiary amine catalysts (Formula 5) which exhibit superior performance in asymmetric [4+2] cyclization reactions compared to commercial alternatives.
Q: Is this process suitable for large-scale industrial manufacturing?
A: The process is highly amenable to scale-up due to its use of common solvents like dichloromethane, commercially available starting materials, and the absence of sensitive transition metals. The simple workup involving filtration and standard column chromatography further supports robust commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of advanced pharmaceuticals and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of axially chiral oxindole-substituted styrene meets the highest international standards. Our commitment to excellence extends beyond mere supply; we act as a strategic partner in your innovation journey, providing the technical support necessary to optimize these complex syntheses for your specific needs.
We invite you to explore the potential of this cutting-edge resolution technology for your upcoming projects. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis tailored to your production volumes and quality requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your time-to-market while optimizing your manufacturing economics. Together, we can unlock the full potential of chiral chemistry for a healthier future.
