Advanced Resolution of Axially Chiral Oxindole-Substituted Styrenes for High-Purity Pharmaceutical Intermediates
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral intermediates that meet stringent regulatory standards in the pharmaceutical industry. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a critical gap in the availability of efficient, metal-free synthetic routes. This technology leverages the power of chiral phosphoric acid organocatalysis to transform racemic mixtures into optically pure building blocks with exceptional stereocontrol. For R&D directors and process chemists, this represents a significant advancement over traditional metal-catalyzed approaches, offering a pathway to complex molecular architectures without the burden of heavy metal contamination. The ability to access these structurally diverse styrenes opens new avenues for developing next-generation ligands and catalysts, positioning this technology as a cornerstone for modern fine chemical manufacturing.
Furthermore, the versatility of this resolution method extends beyond simple isolation; it provides a robust platform for the subsequent derivation of highly active chiral tertiary amine catalysts. These downstream products exhibit superior performance in asymmetric transformations, such as [4+2] cyclizations, outperforming many commercially available counterparts. By integrating this patented resolution strategy into your supply chain, organizations can secure a reliable source of high-value chiral intermediates that are essential for the synthesis of advanced therapeutic agents. The combination of mild reaction conditions, high atom economy, and broad substrate scope makes this approach not only scientifically elegant but also commercially viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral styrene compounds has relied heavily on transition metal catalysis, such as palladium or titanium-mediated coupling reactions, or nucleophilic additions to alkynes. While effective in certain contexts, these conventional methods suffer from significant drawbacks that hinder their widespread adoption in GMP-compliant manufacturing environments. The use of expensive and toxic transition metals necessitates rigorous purification steps to remove trace metal residues, which can drastically increase production costs and extend lead times. Moreover, many of these metal-catalyzed processes require harsh reaction conditions, including elevated temperatures or strong bases, which can compromise the stability of sensitive functional groups and lead to the formation of unwanted byproducts. The limited substrate scope of some traditional methods also restricts the structural diversity of the accessible chiral scaffolds, forcing chemists to rely on lengthy multi-step syntheses to achieve the desired molecular complexity.
The Novel Approach
In stark contrast, the methodology disclosed in CN111848322B utilizes a chiral phosphoric acid catalyst to facilitate the kinetic resolution of racemic styrene precursors under remarkably mild conditions. This organocatalytic approach eliminates the need for transition metals entirely, thereby removing the risk of metal contamination and simplifying the purification workflow. The reaction proceeds efficiently at low temperatures, typically between 0°C and 10°C, which preserves the integrity of sensitive moieties and minimizes thermal degradation. By employing readily available azlactones as reactants in conjunction with the styrene precursors, this method achieves high levels of stereocontrol, delivering products with enantiomeric excess values reaching up to 96%. This shift from metal-based to organocatalytic resolution represents a paradigm shift in the manufacturing of chiral intermediates, offering a cleaner, safer, and more sustainable alternative for the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
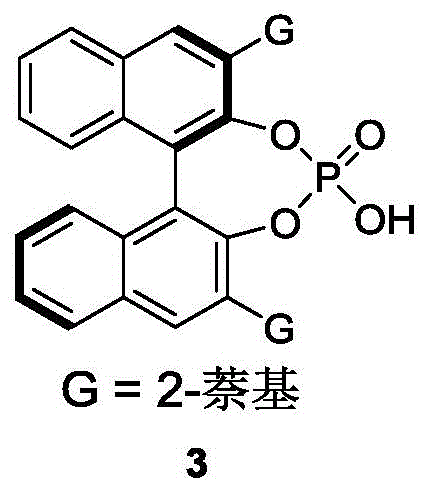
The core of this innovative process lies in the precise stereochemical control exerted by the chiral phosphoric acid catalyst, specifically derivatives based on binaphthyl, octahydrobinaphthyl, or spiro ring skeletons. As illustrated in the patent, the preferred catalyst (Formula 3) features a bulky 2-naphthyl group that creates a well-defined chiral environment around the phosphoric acid moiety. During the reaction, the catalyst likely activates the azlactone electrophile through hydrogen bonding while simultaneously interacting with the styrene nucleophile, guiding the approach of the reactants in a highly specific orientation. This dual activation mechanism ensures that the reaction proceeds through a single favored transition state, resulting in the selective formation of one enantiomer over the other. The use of 3Å molecular sieves as an additive further enhances the reaction efficiency by sequestering water, which could otherwise deactivate the catalyst or promote hydrolysis of the sensitive azlactone intermediate.
From an impurity control perspective, this mechanism offers distinct advantages. The high enantioselectivity inherent to the catalyst design means that the formation of the undesired enantiomer is suppressed from the outset, reducing the burden on downstream purification processes. Furthermore, the mild acidic nature of the phosphoric acid catalyst prevents the occurrence of base-mediated side reactions that are common in traditional coupling protocols. The reaction tolerates a wide range of substituents on both the indole and phenyl rings, including electron-donating and electron-withdrawing groups, without significant loss of selectivity. This robustness ensures consistent product quality across different batches, a critical factor for maintaining supply chain reliability. The ability to fine-tune the catalyst structure by modifying the substituents on the binaphthyl backbone provides an additional layer of control, allowing for optimization of the process for specific substrate classes.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
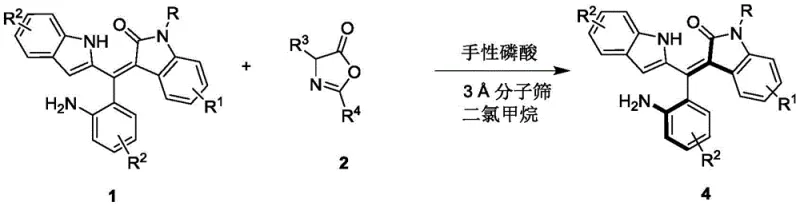
The synthesis of these valuable chiral intermediates is streamlined into a concise operational protocol that balances efficiency with ease of execution. The process begins with the preparation of the reaction mixture in dichloromethane, a solvent chosen for its ability to dissolve both organic reactants while maintaining compatibility with the catalyst system. The stoichiometry is carefully controlled, with a molar ratio of the styrene precursor to the azlactone reactant maintained at approximately 1:0.58 to optimize conversion and selectivity. The addition of the chiral phosphoric acid catalyst at a loading of 10 mol% initiates the transformation, which is allowed to proceed at a controlled temperature of 0°C. Monitoring the reaction progress via thin-layer chromatography (TLC) ensures that the reaction is quenched at the optimal point to maximize yield and purity. Following completion, the mixture is filtered to remove the molecular sieves, concentrated, and subjected to silica gel column chromatography to isolate the final axially chiral product.
- Prepare the reaction mixture by combining the racemic styrene precursor (Formula 1) and the azlactone reactant (Formula 2) in dichloromethane solvent with a molar ratio of approximately 1: 0.58.
- Add 3Å molecular sieves as an additive and introduce the chiral phosphoric acid catalyst (Formula 3, preferably 10 mol%) to the mixture.
- Stir the reaction at a controlled low temperature of 0°C to 10°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel column chromatography to isolate the axially chiral product (Formula 4).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organocatalytic resolution technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts from the synthetic route fundamentally alters the cost structure of the manufacturing process. Without the need for expensive palladium or titanium reagents, raw material costs are significantly reduced. More importantly, the absence of heavy metals removes the necessity for specialized scavenging resins and extensive analytical testing for residual metals, which are often bottlenecks in the release of pharmaceutical intermediates. This simplification of the purification train leads to substantial cost savings in both materials and labor, enhancing the overall economic viability of the project. The mild reaction conditions also contribute to energy efficiency, as the process does not require energy-intensive heating or cooling cycles beyond standard refrigeration.
- Cost Reduction in Manufacturing: The transition to an organocatalytic process eliminates the dependency on precious metal catalysts, which are subject to volatile market pricing and supply constraints. By replacing these with stable, recyclable organic acids, the direct material cost of the catalyst system is drastically lowered. Furthermore, the simplified workup procedure, which avoids complex metal removal steps, reduces the consumption of solvents and purification media. This leaner manufacturing approach results in a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates. The high yields and selectivity reported in the patent further minimize waste generation, contributing to a more sustainable and cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method against variations in substrate structure ensures a consistent supply of critical intermediates. Unlike metal-catalyzed reactions that can be sensitive to trace impurities or moisture, this organocatalytic system demonstrates high tolerance, reducing the risk of batch failures. The use of commercially available starting materials and reagents mitigates the risk of supply disruptions associated with specialized custom synthesis. Additionally, the scalability of the process from gram to kilogram scale has been validated, providing confidence in the ability to meet increasing demand without compromising quality. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturing.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles and regulatory expectations. The absence of toxic heavy metals simplifies waste disposal and reduces the environmental footprint of the manufacturing facility. The use of dichloromethane as a solvent is standard in the industry, and the overall atom economy of the reaction is high, minimizing the generation of hazardous byproducts. As regulatory agencies increasingly scrutinize the environmental impact of pharmaceutical manufacturing, adopting this cleaner technology positions the supply chain for long-term compliance. The potential for catalyst recovery and reuse further enhances the sustainability credentials of the process, making it an attractive option for companies committed to corporate social responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of using chiral phosphoric acid for resolving axially chiral styrenes compared to metal catalysis?
A: The primary advantage is the elimination of toxic heavy metal residues, which simplifies downstream purification and ensures compliance with strict pharmaceutical impurity standards. Additionally, the organocatalytic process operates under significantly milder conditions (0°C) compared to many metal-catalyzed coupling reactions.
Q: What level of enantiomeric excess (ee) can be achieved with this resolution method?
A: According to the patent data, this method consistently achieves high enantioselectivity, with specific embodiments demonstrating ee values up to 96%, ensuring the production of high-purity chiral intermediates suitable for sensitive catalytic applications.
Q: Can the resolved styrene compounds be further functionalized into active catalysts?
A: Yes, the resolved axially chiral styrenes (Formula 4) serve as versatile precursors. They can be derivatized into chiral tertiary amine catalysts (Formula 5), which have been shown to effectively catalyze asymmetric [4+2] cyclization reactions with superior performance compared to commercial alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of advanced therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN111848322B can be seamlessly translated into industrial reality. We are equipped with state-of-the-art facilities and rigorous QC labs capable of meeting stringent purity specifications, including the demanding enantiomeric excess requirements of axially chiral compounds. Our commitment to quality assurance guarantees that every batch of axially chiral oxindole-substituted styrene delivered meets the highest standards of consistency and performance, supporting your R&D and manufacturing goals with unwavering reliability.
We invite you to collaborate with us to explore the full potential of this innovative resolution technology for your specific applications. Whether you require custom synthesis of novel derivatives or large-scale supply of the resolved intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our expertise can accelerate your timeline and reduce your overall development costs. Let us be your partner in navigating the complexities of chiral synthesis and securing a competitive edge in the marketplace.
