Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
In the rapidly evolving landscape of medicinal chemistry, the incorporation of fluorine atoms into drug candidates has become a cornerstone strategy for enhancing metabolic stability and bioavailability. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical safety and efficiency bottlenecks in current synthetic methodologies. This innovation leverages a base-promoted cyclization strategy that bypasses the traditional reliance on toxic azides and heavy metal catalysts, offering a streamlined pathway for constructing these vital heterocyclic scaffolds. For R&D teams focused on developing next-generation therapeutics, this technology represents a significant leap forward in process safety and environmental compliance. By utilizing cheap and readily available diazo compounds and trifluoroethylimidoyl chlorides, the method ensures a robust supply chain for high-purity pharmaceutical intermediates. The ability to access diverse substitution patterns at the 1 and 4 positions of the triazole ring further expands the chemical space available for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has been dominated by copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation steps. These traditional routes suffer from severe drawbacks, primarily the requirement for hazardous organic azides which are known for their potential explosiveness and toxicity, posing significant safety risks in both laboratory and industrial settings. Furthermore, the reliance on transition metal catalysts necessitates rigorous downstream purification processes to remove trace metal residues, which is a critical regulatory requirement for active pharmaceutical ingredients (APIs). The multi-step nature of these conventional protocols often leads to lower overall yields and increased waste generation, complicating the commercial scale-up of complex heterocycles. Additionally, the use of specialized trifluoromethylating reagents can be cost-prohibitive and may introduce compatibility issues with sensitive functional groups present in advanced intermediates. These cumulative factors create substantial barriers to efficient manufacturing, driving up costs and extending lead times for high-purity intermediates needed in drug development pipelines.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in CN113121462B employs a simple, efficient, and metal-free strategy that fundamentally reshapes the synthetic landscape for these compounds. As illustrated in the reaction overview below, the new method utilizes a base-promoted reaction between trifluoroethylimidoyl chlorides and diazo compounds, completely circumventing the need for explosive azides or expensive copper catalysts. 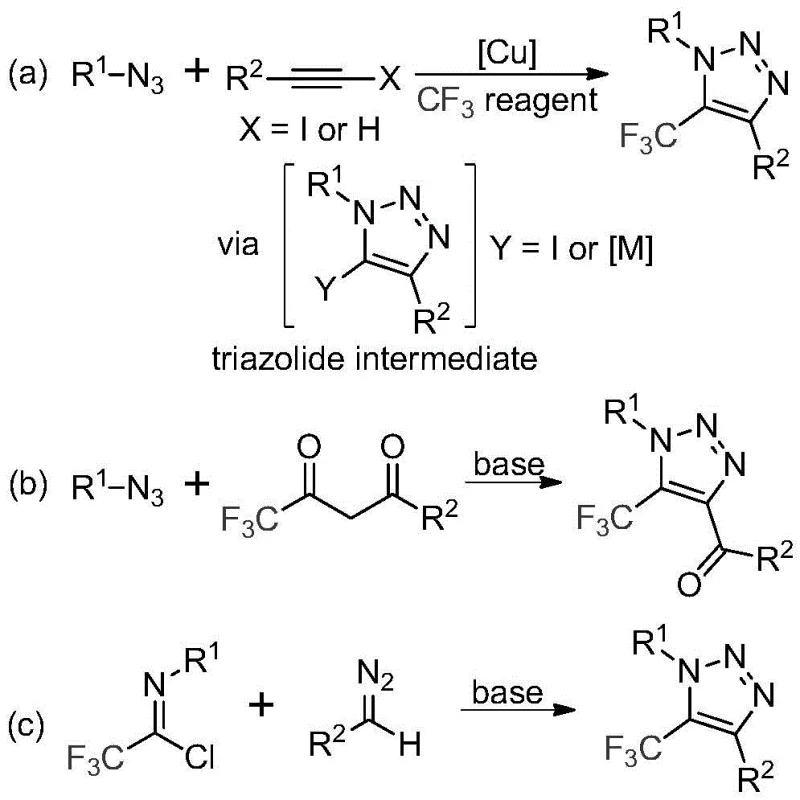 This direct construction of the triazole ring not only simplifies the operational procedure but also drastically improves the safety profile of the manufacturing process. The reaction conditions are mild, typically proceeding at moderate temperatures in common organic solvents like acetonitrile, which facilitates easy handling and scalability. By eliminating the metal catalyst, the process inherently avoids the formation of metal-complexed impurities, thereby reducing the burden on purification teams and ensuring higher quality outputs. This approach exemplifies a shift towards greener chemistry principles, aligning with the industry's growing demand for sustainable and cost-effective synthetic routes for reliable pharmaceutical intermediate supplier networks.
This direct construction of the triazole ring not only simplifies the operational procedure but also drastically improves the safety profile of the manufacturing process. The reaction conditions are mild, typically proceeding at moderate temperatures in common organic solvents like acetonitrile, which facilitates easy handling and scalability. By eliminating the metal catalyst, the process inherently avoids the formation of metal-complexed impurities, thereby reducing the burden on purification teams and ensuring higher quality outputs. This approach exemplifies a shift towards greener chemistry principles, aligning with the industry's growing demand for sustainable and cost-effective synthetic routes for reliable pharmaceutical intermediate supplier networks.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation involves a sophisticated yet elegant sequence of bond-forming events initiated by the base. The reaction likely commences with a base-promoted intermolecular nucleophilic addition of the diazo compound to the electrophilic carbon of the trifluoroethylimidoyl chloride. This step is crucial as it establishes the initial carbon-carbon bond connectivity required for the subsequent ring closure, driven by the elimination of the chloride leaving group. Following this addition-elimination sequence, the intermediate undergoes an intramolecular 5-endo-dig cyclization, which effectively closes the five-membered triazole ring system. This specific cyclization mode is favored under the reaction conditions provided, leading to the exclusive formation of the 5-trifluoromethyl substituted isomer with high regioselectivity. The use of cesium carbonate as the promoter is particularly effective due to its optimal basicity and solubility profile in aprotic solvents, which helps to drive the equilibrium towards product formation without degrading the sensitive diazo functionality. Understanding this mechanism allows chemists to fine-tune reaction parameters to maximize yield and minimize side reactions, ensuring the production of high-purity pharmaceutical intermediates suitable for stringent regulatory standards.
From an impurity control perspective, the absence of transition metals eliminates a major source of contamination that often plagues heterocyclic synthesis. In traditional copper-catalyzed routes, metal-ligand complexes can persist through workup, requiring specialized scavengers or chromatography to meet ppm-level specifications. In this metal-free protocol, the primary byproducts are inorganic salts derived from the base and the eliminated chloride, which are easily removed during the aqueous workup or filtration steps. The mild thermal conditions (50-70°C) further suppress thermal decomposition pathways that could lead to complex impurity profiles. Moreover, the broad functional group tolerance of the diazo and imidoyl chloride starting materials means that sensitive moieties such as esters, ketones, and halides remain intact throughout the transformation. This robustness is essential for late-stage functionalization strategies where the triazole motif is introduced into complex molecular architectures. Consequently, this method offers a cleaner reaction profile that simplifies the isolation process and enhances the overall purity of the final API intermediate.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and reaction monitoring to ensure optimal performance. The patent outlines a straightforward protocol where cesium carbonate, trifluoroethylimidoyl chloride, and the diazo compound are combined in an organic solvent such as acetonitrile, often with the addition of molecular sieves to manage moisture. The reaction is typically heated to a moderate temperature range of 50-70°C and stirred for a period of 8 to 16 hours to achieve full conversion. Detailed standard operating procedures for this synthesis, including precise molar ratios and workup techniques, are provided in the technical guide below to assist process chemists in replication. 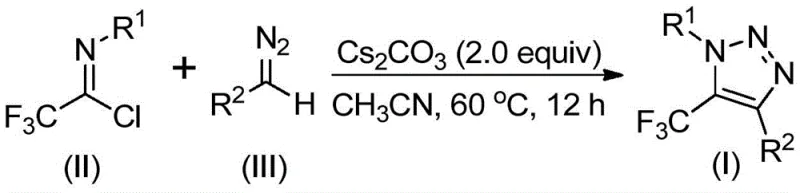 Adhering to these standardized steps ensures reproducibility and safety, allowing teams to reliably produce the target triazole derivatives. The simplicity of the setup, requiring only standard glassware and heating equipment, makes this method accessible for both small-scale discovery and larger pilot plant operations. By following the established guidelines, manufacturers can achieve consistent results while maintaining strict control over the quality attributes of the synthesized material.
Adhering to these standardized steps ensures reproducibility and safety, allowing teams to reliably produce the target triazole derivatives. The simplicity of the setup, requiring only standard glassware and heating equipment, makes this method accessible for both small-scale discovery and larger pilot plant operations. By following the established guidelines, manufacturers can achieve consistent results while maintaining strict control over the quality attributes of the synthesized material.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, diazo compound, and 4Å molecular sieves in an organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the high-purity triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of expensive transition metal catalysts and specialized ligands results in a significant reduction in raw material costs, while the simplified purification workflow reduces solvent consumption and waste disposal expenses. This streamlining of the manufacturing process translates into substantial cost savings in API manufacturing, making the final drug substance more competitive in the global market. Furthermore, the reliance on commercially available and stable starting materials mitigates supply chain risks associated with sourcing hazardous or proprietary reagents. The robustness of the reaction conditions ensures high batch-to-batch consistency, which is critical for maintaining uninterrupted supply lines to downstream formulation units. By reducing the complexity of the synthesis, companies can also accelerate the timeline from process development to commercial production, enabling faster time-to-market for new therapeutic candidates.
- Cost Reduction in Manufacturing: The removal of copper catalysts and the avoidance of toxic azides eliminate the need for costly metal scavenging resins and specialized safety containment infrastructure. This qualitative shift in process design removes several high-cost unit operations from the production flow, leading to a leaner and more economical manufacturing process. Additionally, the use of inexpensive bases like cesium carbonate and common solvents like acetonitrile further drives down the variable costs per kilogram of product. The overall efficiency gains mean that resources can be reallocated to other critical areas of drug development, maximizing the return on investment for the project.
- Enhanced Supply Chain Reliability: Sourcing stable diazo compounds and imidoyl chlorides is significantly less risky than managing the supply of explosive organic azides, which often require special permits and storage conditions. This stability ensures a continuous flow of materials even during periods of market volatility or logistical disruptions. The ability to synthesize the target molecules from readily available precursors reduces dependency on single-source suppliers for exotic reagents. Consequently, this enhances the resilience of the supply chain, ensuring that production schedules are met without unexpected delays caused by material shortages or regulatory hurdles associated with hazardous chemicals.
- Scalability and Environmental Compliance: The metal-free nature of this reaction aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge in pharmaceutical wastewater. Scaling this process from gram to kilogram levels does not introduce new safety hazards related to exothermic metal-catalyzed reactions or azide accumulation. The simplified workup involving filtration and standard chromatography is easily adaptable to large-scale industrial equipment. This ease of scale-up facilitates the commercial scale-up of complex heterocycles, allowing manufacturers to meet growing market demand without compromising on environmental stewardship or worker safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology. Understanding these details is crucial for making informed decisions about integrating this route into existing manufacturing portfolios. The answers reflect the practical realities of running this chemistry in a GMP environment, focusing on safety, quality, and efficiency.
Q: Does this synthesis method require toxic azide reagents?
A: No, unlike conventional copper-catalyzed methods, this novel base-promoted approach utilizes stable diazo compounds and imidoyl chlorides, completely eliminating the need for hazardous and explosive organic azides.
Q: What are the cost advantages of this metal-free route?
A: By avoiding expensive transition metal catalysts like copper and complex ligands, and removing the costly downstream purification steps required to remove heavy metal residues, this method significantly lowers the overall production cost.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates that the reaction proceeds efficiently under mild conditions with readily available starting materials, making it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our expertise in process optimization allows us to adapt this novel chemistry to your specific needs, guaranteeing a reliable supply of high-purity pharmaceutical intermediates that comply with global regulatory standards. Partnering with us means gaining access to a robust supply chain capable of supporting your long-term commercial goals.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the economic impact of switching to this metal-free protocol. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules. Let us help you optimize your supply chain and accelerate your drug development timeline with our superior manufacturing capabilities and technical support.
