Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those bearing fluorine motifs which enhance metabolic stability and bioavailability. Patent CN113121462B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical limitations in existing synthetic pathways. This technology leverages a base-promoted cyclization strategy that bypasses the need for toxic azides and expensive transition metal catalysts, representing a significant leap forward in green chemistry for high-purity pharmaceutical intermediates. By utilizing readily available trifluoroethylimidoyl chlorides and diazo compounds, this process offers a streamlined route to valuable scaffolds found in beta3 adrenergic receptor agonists and various fungicides. For R&D directors and procurement specialists alike, understanding this innovation is key to securing a reliable pharmaceutical intermediate supplier capable of delivering complex molecules with superior impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present substantial operational and safety hurdles for large-scale manufacturing. The first conventional approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides to form triazole-metal complexes, which subsequently react with trifluoromethyl reagents. This pathway is fraught with dangers due to the inherent instability and explosive nature of organic azides, posing severe safety risks during storage and handling in a production environment. Additionally, the reliance on copper catalysts introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet stringent regulatory limits for active pharmaceutical ingredients. The second traditional method employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones, which similarly suffers from the safety liabilities associated with azide usage. These legacy methods often result in lower atom economy and generate significant hazardous waste, complicating environmental compliance and driving up the overall cost of goods sold for fine chemical intermediates.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the methodology described in patent CN113121462B introduces a metal-free, azide-free synthesis that fundamentally reshapes the risk profile of triazole production. As illustrated in the comparative reaction schemes below, this novel route utilizes stable diazo compounds and trifluoroethylimidoyl chlorides as starting materials, promoted simply by an inorganic base.
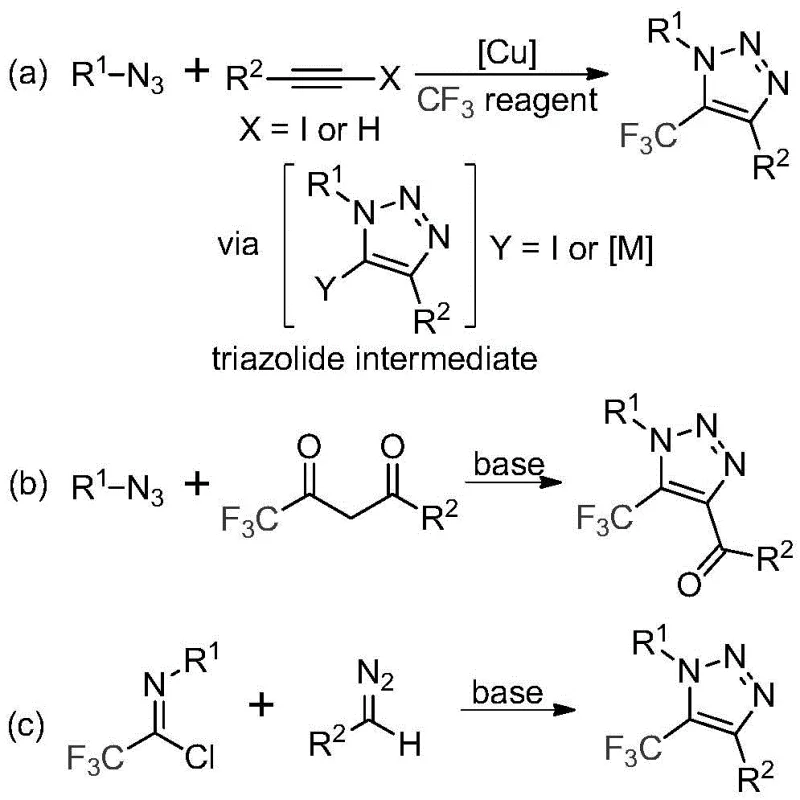
This shift eliminates the need for explosive azide precursors entirely, thereby drastically simplifying safety protocols and reducing insurance and containment costs for manufacturing facilities. Furthermore, the absence of transition metals like copper means that the downstream purification process is significantly simplified, removing the need for specialized scavengers or complex extraction procedures to reduce metal residues. The reaction conditions are remarkably mild, typically proceeding in common aprotic solvents like acetonitrile at moderate temperatures, which enhances energy efficiency. This approach not only improves the safety and environmental footprint of the synthesis but also broadens the substrate scope, allowing for the efficient incorporation of diverse functional groups that might be incompatible with harsher metal-catalyzed conditions, ultimately facilitating the rapid discovery of new drug candidates.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic elegance of this transformation lies in its ability to forge the triazole ring through a cascade of nucleophilic attacks and cyclizations without external metal activation. The reaction initiates with the base, typically cesium carbonate, promoting the activation of the diazo compound. Although diazo compounds are often viewed primarily as carbene precursors, under these specific basic conditions, they participate in an intermolecular nucleophilic addition-elimination process with the electrophilic trifluoroethylimidoyl chloride. This initial step facilitates the formation of a critical carbon-carbon bond, setting the stage for the subsequent ring closure. Following this addition, the intermediate undergoes an intramolecular 5-endo-dig cyclization, a kinetically favorable process that constructs the five-membered 1,2,3-triazole core with high regioselectivity. This mechanism ensures that the trifluoromethyl group is installed specifically at the 5-position of the triazole ring, a structural feature crucial for the biological activity of many target molecules.
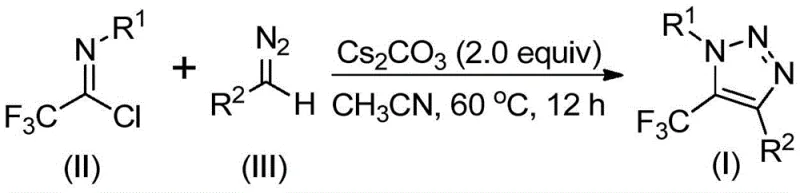
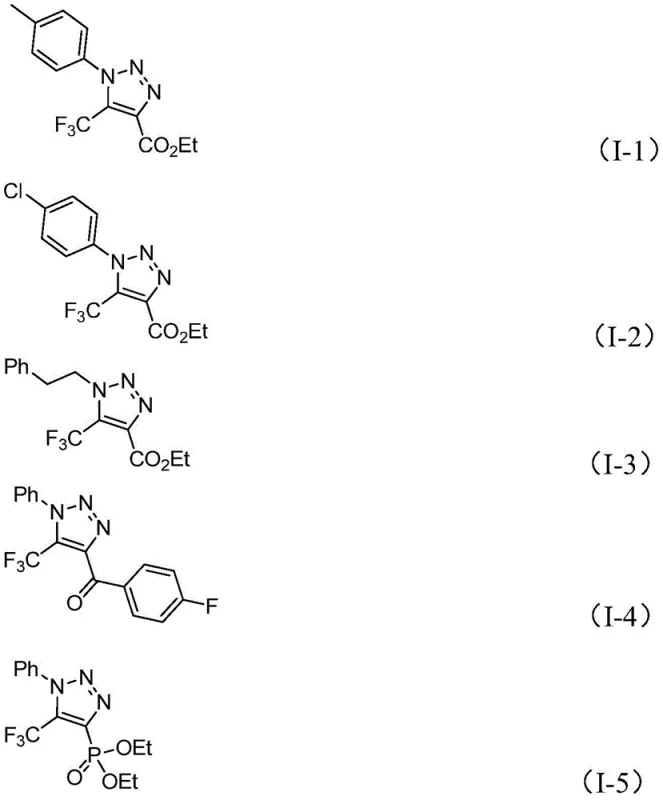
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-mediated pathways. The ionic nature of the base-promoted reaction tends to produce cleaner reaction profiles with fewer side products derived from radical recombination or metal-complex decomposition. The use of cesium carbonate, a mild yet effective base, helps to minimize the degradation of sensitive functional groups on the aromatic rings of the substrates, such as esters, halogens, or phosphonates. As demonstrated by the diverse array of successful substrates shown below, the reaction tolerates electron-withdrawing and electron-donating groups alike, maintaining high efficiency across a broad chemical space. This robustness is essential for process chemists aiming to lock in a synthesis route early in drug development, as it reduces the likelihood of having to re-optimize the chemistry when scaling up or modifying the lead structure.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The protocol is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes or specialized high-pressure equipment, which makes it accessible for most standard organic synthesis facilities. The following section outlines the standardized procedure derived from the patent examples, ensuring reproducibility and consistency for commercial scale-up of complex pharmaceutical intermediates.
- Prepare the reaction mixture by adding cesium carbonate (2.0 equivalents), 4 Å molecular sieves, trifluoroethylimidoyl chloride, and the corresponding diazo compound into an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50°C to 70°C (optimally 60°C) and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Upon completion, filter the mixture to remove solids, concentrate the filtrate, and purify the crude residue via silica gel column chromatography to isolate the pure 5-trifluoromethyl-1,2,3-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible operational efficiencies and risk mitigation. The shift away from hazardous azides and precious metal catalysts fundamentally alters the cost structure and supply security of producing these valuable heterocyclic building blocks. By simplifying the raw material portfolio and reducing the complexity of waste management, manufacturers can achieve a more resilient and cost-effective supply chain.
- Cost Reduction in Manufacturing: The elimination of copper catalysts removes a significant cost driver associated with both the purchase of noble metals and the subsequent removal processes required to meet ppm-level specifications. Furthermore, replacing explosive azides with stable diazo compounds and imidoyl chlorides reduces the need for specialized blast-proof infrastructure and expensive safety monitoring systems. The use of cesium carbonate, a relatively inexpensive inorganic base, further drives down reagent costs compared to specialized organocatalysts or ligand systems. These factors combine to substantially lower the overall cost of goods, allowing for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including trifluoroethylimidoyl chlorides and various diazo compounds, are commercially available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike custom-synthesized azides which often have long lead times and strict transport regulations, these precursors can be sourced reliably and stored safely for extended periods. This availability ensures continuous production schedules and minimizes the risk of delays caused by raw material shortages, providing a stable supply of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The reaction operates under mild thermal conditions (50-70°C) in common solvents like acetonitrile, which are easily recovered and recycled, aligning with modern green chemistry principles. The absence of heavy metals simplifies wastewater treatment and reduces the environmental burden of the manufacturing process, facilitating easier regulatory approval in jurisdictions with strict environmental laws. The protocol has been demonstrated to scale effectively from milligram to gram levels with consistent yields, indicating a clear path toward multi-kilogram and ton-scale production without the need for extensive re-engineering of the process parameters.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for technical teams evaluating this route for their own production needs.
Q: What are the safety advantages of this new triazole synthesis method compared to traditional routes?
A: Unlike conventional copper-catalyzed azide-alkyne cycloadditions that require hazardous and potentially explosive organic azides, this novel method utilizes stable diazo compounds and imidoyl chlorides. Furthermore, it eliminates the need for toxic transition metal catalysts, significantly reducing heavy metal contamination risks in the final API intermediate.
Q: Can this synthesis protocol be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states that the method is easily scalable to the gram level and beyond. The use of inexpensive inorganic bases like cesium carbonate and common solvents like acetonitrile, combined with mild reaction temperatures (60°C), makes it highly suitable for commercial scale-up in fine chemical manufacturing.
Q: What is the substrate scope for the R1 and R2 groups in this triazole formation?
A: The reaction demonstrates broad functional group tolerance. R1 can be alkyl, substituted or unsubstituted aryl groups (including phenethyl and various substituted phenyls), while R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for the design of diverse molecular scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the fast-evolving landscape of drug discovery and agrochemical development. Our team of expert process chemists has thoroughly analyzed the technology disclosed in patent CN113121462B and is fully prepared to leverage this metal-free, azide-free route for your specific project requirements. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediate we deliver meets the highest global standards for quality and safety.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and purity requirements. We encourage you to contact us today to request specific COA data for our catalog compounds or to initiate a discussion on route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply of these critical building blocks for your future success.
