Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
Advanced Synthesis of N-N Axis Chiral Pyrrole Derivatives for Pharmaceutical Applications
The landscape of asymmetric synthesis is continually evolving to meet the rigorous demands of modern drug discovery, particularly for complex heterocyclic scaffolds. A groundbreaking advancement in this field is detailed in patent CN114524701B, which discloses a highly efficient method for constructing N-N axis chiral pyrrole derivatives. These compounds are not merely structural curiosities; they represent a critical class of pharmaceutical intermediates with demonstrated potent cytotoxic activity against QGP-1 tumor cells. The innovation lies in the strategic use of chiral phosphoric acid organocatalysts to facilitate an in-situ ring formation strategy, a approach that significantly diverges from traditional dynamic kinetic resolution methods. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology offers a pathway to access high-purity chiral building blocks that were previously difficult to synthesize with such high stereocontrol.
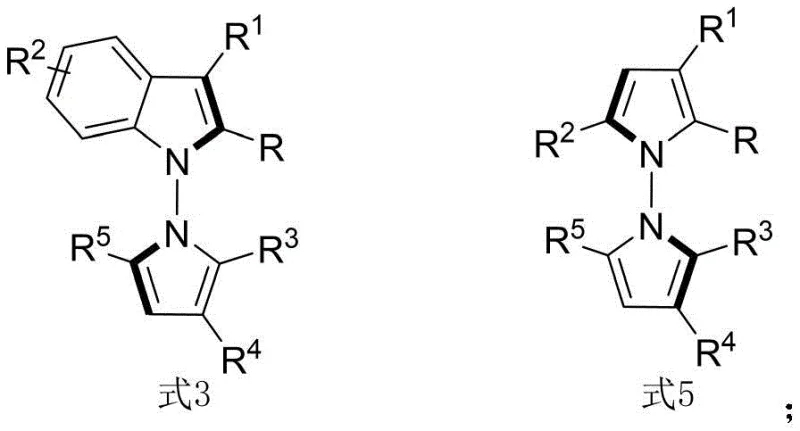
The structural versatility of these derivatives, as illustrated by the general formulas where various R groups can be modified, allows for extensive structure-activity relationship (SAR) studies. This flexibility is paramount for medicinal chemists aiming to optimize lead compounds for specific biological targets. By expanding the range of available N-N axis chiral pyrrole derivatives, this synthesis method opens new avenues for exploring chemical space in oncology drug development. The ability to introduce diverse substituents such as halogens, alkoxy groups, and aryl rings without compromising stereoselectivity underscores the robustness of the catalytic system. Consequently, this patent represents a significant value proposition for companies focused on cost reduction in API manufacturing, as it streamlines the production of high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral compounds has been fraught with challenges, often relying on resolution techniques that inherently limit yield to a maximum of 50% or requiring stoichiometric amounts of chiral auxiliaries that generate significant waste. Conventional methods for accessing N-N axis chirality have been particularly scarce, typically restricted to dynamic kinetic resolution or desymmetrization reactions which demand precise control over reaction parameters and often involve expensive transition metal catalysts. These traditional pathways frequently suffer from narrow substrate scope, meaning that slight modifications to the starting material can lead to catastrophic drops in enantioselectivity or yield. Furthermore, the removal of heavy metal residues from the final product adds a layer of complexity and cost to the downstream processing, posing a significant hurdle for supply chain heads concerned with regulatory compliance and purity specifications. The lack of direct, catalytic asymmetric methods for constructing these specific N-N bonds has created a bottleneck in the availability of diverse libraries for biological screening.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach described in the patent utilizes a direct condensation and cyclization strategy catalyzed by chiral phosphoric acids. This method employs readily available indoleamines or pyrroleamines reacting with 1,4-diketone derivatives to form the target N-N axis chiral pyrroles in a single operational step. The use of carbon tetrachloride as a solvent and 3A molecular sieves as an additive creates an optimal environment for water removal, driving the equilibrium towards product formation without the need for extreme temperatures or pressures. This in-situ ring formation strategy is not only atom-economical but also remarkably tolerant of various functional groups, allowing for the synthesis of a wide array of structurally complex products. By shifting from resolution to direct asymmetric synthesis, the process theoretically doubles the potential yield compared to racemic resolution, offering substantial cost savings and efficiency gains for commercial scale-up of complex pharmaceutical intermediates.
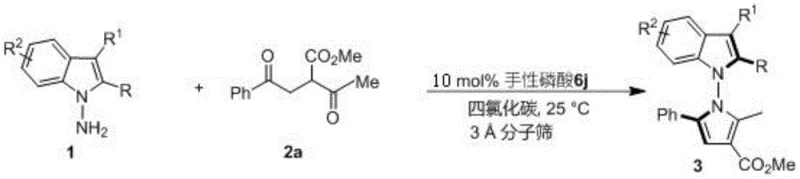
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this transformation hinges on the precise interaction between the chiral phosphoric acid catalyst and the substrates. The catalyst, acting as a Brønsted acid, activates the carbonyl groups of the 1,4-diketone derivative through hydrogen bonding, thereby increasing their electrophilicity towards nucleophilic attack by the amine nitrogen. Simultaneously, the chiral pocket created by the bulky substituents on the catalyst backbone—specifically the spiro-binaphthyl structure of catalyst 6j—exerts steric control over the transition state. This dual activation mode ensures that the cyclization occurs with high facial selectivity, locking the N-N axis into a specific chiral configuration. The rigidity of the spiro-backbone is crucial, as it prevents conformational freedom that could lead to racemization, thus securing the high enantiomeric excess values observed, often exceeding 90% ee. Understanding this mechanistic nuance is vital for process chemists aiming to replicate or further optimize the reaction conditions for specific substrate classes.
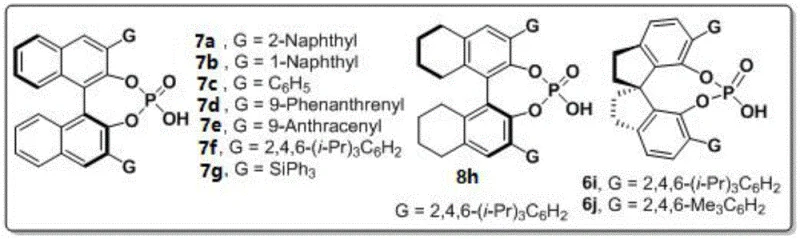
Furthermore, the choice of catalyst 6j, featuring 2,4,6-trimethylphenyl groups, provides an ideal balance of acidity and steric bulk. The electron-donating methyl groups modulate the acidity of the phosphoric acid proton, ensuring it is strong enough to activate the diketone but mild enough to prevent side reactions or decomposition of sensitive intermediates. The ortho-methyl groups create a confined chiral environment that effectively discriminates between the pro-chiral faces of the reacting species. This level of control is what allows the reaction to proceed at room temperature (25°C), a condition that is exceptionally mild for forming such sterically hindered N-N bonds. For R&D teams, this implies that the process is robust and less prone to thermal degradation issues, simplifying the safety profile and equipment requirements for scale-up. The mechanism essentially bypasses the need for external oxidants or reductants, relying solely on the thermodynamic drive of water elimination and the kinetic control of the chiral catalyst.
How to Synthesize N-N Axis Chiral Pyrrole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocols to maximize yield and stereoselectivity. The general procedure involves dissolving the amine substrate and the 1,4-diketone derivative in carbon tetrachloride, followed by the addition of activated 3A molecular sieves to scavenge the water produced during the condensation. The chiral phosphoric acid catalyst is then introduced, typically at a loading of 10 mol%, although this can be tuned based on the specific reactivity of the substrates. The reaction mixture is stirred at ambient temperature, monitored by thin-layer chromatography (TLC) until the starting materials are consumed. Following completion, the mixture is filtered to remove the molecular sieves and catalyst residues, concentrated under reduced pressure, and the crude product is purified via silica gel column chromatography using a petroleum ether and ethyl acetate mixture. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining indoleamine or pyrroleamine with a 1,4-diketone derivative in carbon tetrachloride solvent.
- Add 3A molecular sieves as an additive and introduce 10 mol% of the optimized chiral phosphoric acid catalyst (specifically compound 6j).
- Stir the reaction at 25°C until TLC indicates completion, then filter, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method addresses several critical pain points in the supply chain for chiral intermediates. The reliance on organocatalysis rather than transition metals eliminates the need for costly and time-consuming heavy metal scavenging processes, which are often required to meet stringent pharmaceutical purity standards. This simplification of the downstream processing directly translates to reduced operational expenditures and shorter production cycles. Additionally, the use of common, inexpensive solvents and reagents ensures that the raw material costs remain low, facilitating significant cost reduction in pharmaceutical intermediate manufacturing. The mild reaction conditions also imply lower energy consumption, as there is no need for cryogenic cooling or high-temperature heating, further enhancing the environmental and economic sustainability of the process. For procurement managers, this means a more stable and predictable cost structure for sourcing these high-value chiral building blocks.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated purification steps significantly lowers the overall production cost. By utilizing a catalytic amount of an organic acid that can potentially be recovered or is inexpensive to synthesize, the process avoids the high overheads associated with precious metal chemistry. Furthermore, the high yields and enantioselectivity reduce the amount of wasted starting material and the need for recycling racemic mixtures, optimizing the atom economy. This efficiency allows for a more competitive pricing model for the final API intermediate, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. Since the reaction tolerates a wide range of functional groups and proceeds well at room temperature, the risk of batch failure due to minor fluctuations in temperature or reagent quality is minimized. The availability of diverse substrates means that supply is not dependent on a single, hard-to-source precursor, allowing for greater flexibility in raw material procurement. This resilience is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic filtration and chromatography, makes this method highly amenable to scale-up from gram to kilogram and ton scales. The absence of toxic heavy metals simplifies waste disposal and regulatory compliance, aligning with green chemistry principles. This environmental compatibility reduces the burden on waste treatment facilities and minimizes the regulatory hurdles associated with introducing new manufacturing processes. For supply chain heads, this ensures long-term viability and reduces the risk of production stoppages due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these N-N axis chiral pyrrole derivatives. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical implementation of this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this synthetic route into their existing pipelines.
Q: What is the primary advantage of using chiral phosphoric acid in this synthesis?
A: The use of chiral phosphoric acid, particularly the spiro-backbone derivative 6j, enables extremely high enantioselectivity (up to 96% ee) under mild room temperature conditions, avoiding the need for harsh reagents or transition metals.
Q: What biological activity do these N-N axis chiral pyrrole derivatives exhibit?
A: Biological testing indicates that these derivatives possess strong cytotoxic activity against QGP-1 pancreatic tumor cells, suggesting significant potential for development as anticancer pharmaceutical intermediates.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes conventional reaction conditions, inexpensive solvents like carbon tetrachloride, and simple workup procedures involving filtration and column chromatography, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN114524701B for the development of next-generation anticancer agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of enantiomeric excess via chiral HPLC, which is critical for the biological efficacy of these chiral intermediates. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry.
We invite you to collaborate with us to leverage this advanced chiral phosphoric acid catalysis technology for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target N-N axis chiral pyrrole derivatives, and let us help you accelerate your path to market with confidence and efficiency.
