Innovative Metal-Free Quinoline Synthesis Commercial Scale-Up High-Purity Pharmaceutical Intermediates Cost Reduction
Patent CN106938984B introduces a novel one-pot synthesis method for 5-substituted arylation/heterocycle 8-acylamino quinoline compounds which serve as critical building blocks in pharmaceutical development pipelines. This breakthrough eliminates transition metal catalysts during the initial halogenation phase thereby preventing contamination that traditionally complicates subsequent coupling reactions while maintaining high regioselectivity at the challenging 5-position of quinoline scaffolds. The process achieves exceptional yields through direct coupling of halogenated intermediates with boronic acids without intermediate isolation significantly reducing processing time compared to conventional multi-step approaches. By operating under moderate thermal conditions (50–160°C) with standard organic solvents this methodology offers immediate compatibility with existing manufacturing infrastructure while addressing long-standing challenges in producing complex quinoline derivatives essential for next-generation drug discovery programs. The elimination of metal residues ensures compliance with increasingly stringent regulatory requirements for residual metals in active pharmaceutical ingredients making this innovation particularly valuable for global pharmaceutical manufacturers seeking robust supply chain solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for quinoline derivatives typically require multi-step procedures involving expensive pre-functionalized aniline precursors that must be synthesized separately creating significant operational complexity and yield loss at each stage. Classical cycloaddition methods suffer from poor regioselectivity at the critical 5-position making targeted arylation particularly difficult while existing one-pot approaches relying on metal-catalyzed halogenation introduce transition metal impurities that poison subsequent coupling reactions necessitating extensive purification steps. These limitations collectively result in lower overall yields higher production costs extended lead times and compromised scalability due to the need for specialized equipment to handle sensitive intermediates. Furthermore residual metals require costly removal processes to meet pharmaceutical purity standards adding both time and expense while increasing environmental waste streams through additional solvent usage during purification protocols.
The Novel Approach
The patented methodology overcomes these challenges through a strategically designed two-step sequence where metal-free halogenation using reagents like NBS or NCS is immediately followed by palladium-catalyzed Suzuki-Miyaura coupling without intermediate isolation or purification. By eliminating transition metals entirely from the initial halogenation phase the process prevents catalyst poisoning during coupling while maintaining high reaction efficiency across diverse substrate combinations as demonstrated by yields ranging from 32% to 87% in patent examples. The one-pot design significantly reduces processing time by eliminating column chromatography between steps while utilizing common solvents like DMF and DMSO ensures seamless integration with existing manufacturing infrastructure. Reaction conditions operate within practical temperature ranges (50–160°C) with controlled reaction times (10–24 hours for halogenation; 4–16 hours for coupling) making this approach readily adaptable to commercial-scale production without requiring specialized equipment or hazardous reagents.
Mechanistic Insights into Metal-Free Halogenation and One-Pot Coupling
The reaction mechanism begins with electrophilic halogenation at the electron-rich 5-position of the 8-amidoquinoline substrate facilitated by halogen sources such as NBS under thermal activation where the amido group directs regioselectivity through hydrogen bonding interactions stabilizing the transition state. This metal-free pathway proceeds without transition metals preventing contamination that would otherwise interfere with subsequent palladium-catalyzed coupling reactions which utilize complexes like tetrakis(triphenylphosphine)palladium(0). The one-pot design enables immediate addition of boronic acid and base after halogenation minimizing intermediate decomposition through oxidative addition of palladium into the carbon-halogen bond followed by transmetalation with boronic acid and reductive elimination forming the new carbon-carbon bond at the critical position. This sequential transformation achieves remarkable regioselectivity due to the directing effect of the amido group while maintaining compatibility across diverse aryl and heterocyclic substrates as evidenced by successful incorporation of phenyl thiophene furan and pyridyl moieties.
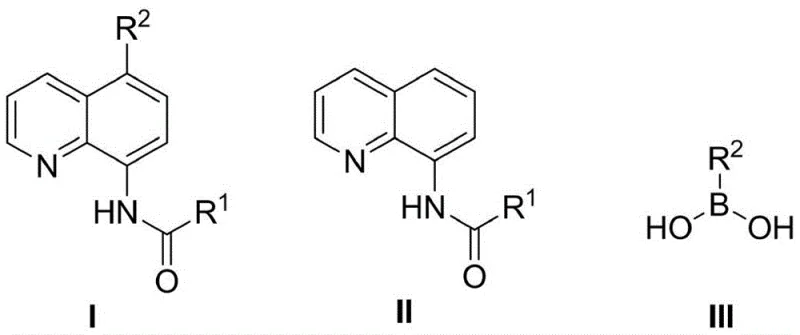
Impurity control is achieved through precise optimization of reaction parameters including temperature modulation (50–160°C) solvent selection (DMF DMSO toluene) and base concentration which prevent side reactions such as over-halogenation or protodeboronation commonly observed in conventional methods. The use of sodium carbonate maintains optimal pH for transmetalation while minimizing hydrolysis of sensitive functional groups ensuring consistent product quality across diverse substrates. Critical to pharmaceutical applications is the elimination of transition metals during halogenation which avoids introducing impurities requiring costly removal processes to meet regulatory specifications for residual metals. Patent examples demonstrate consistent production of high-purity intermediates through this controlled mechanism with column chromatography serving only as a final polishing step rather than a necessity for intermediate purification thereby significantly reducing impurity profiles compared to traditional multi-metal-catalyzed approaches.
How to Synthesize Quinoline Derivatives Efficiently
The patented one-pot methodology represents a significant advancement in quinoline derivative synthesis offering both operational simplicity and enhanced product quality for pharmaceutical manufacturers. By integrating halogenation and coupling into a single continuous process without intermediate isolation this approach eliminates time-consuming purification steps while maintaining high selectivity at the critical 5-position. The following standardized procedure details the precise conditions required to replicate this breakthrough technology across diverse substrate combinations ensuring consistent results from laboratory scale to commercial production. Detailed operational parameters including catalyst loading solvent ratios and temperature profiles are provided below to facilitate seamless technology transfer.
- Perform metal-free halogenation by reacting aminoquinoline compound with halide (NBS/NCS) in DMF at 50–160°C for 10–24 hours under nitrogen atmosphere
- Directly add arylboronic acid, sodium carbonate base, and palladium catalyst without intermediate isolation
- Conduct coupling reaction at 50–160°C for 4–16 hours in DMSO solvent followed by column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis method directly addresses key pain points in pharmaceutical supply chains by transforming complex multi-step processes into streamlined single-vessel operations that reduce raw material consumption while minimizing solvent waste creating substantial environmental benefits alongside cost savings. For procurement teams managing tight budgets and demanding timelines this process offers a compelling solution to traditional bottlenecks in intermediate production that often lead to supply disruptions and price volatility through its reliance on readily available starting materials including common halogenating agents and boronic acids which reduce dependency on specialized reagents prone to supply chain interruptions.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the halogenation step eliminates expensive purification procedures required to remove residual metals significantly reducing both material costs and processing time while minimizing solvent usage through elimination of intermediate isolation steps leading to substantial savings in raw materials labor costs and equipment utilization without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials this method reduces vulnerability to supply chain disruptions caused by specialized reagent dependencies while simplified process flow decreases production cycle times enabling faster response to demand fluctuations through fewer unit operations that maintain consistent quality standards across all production scales from laboratory validation through commercial manufacturing volumes.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability as evidenced by consistent yields across various substrate combinations with reaction conditions easily adaptable to existing manufacturing infrastructure while elimination of toxic metal catalysts reduces hazardous waste generation simplifying environmental compliance through reduced solvent consumption and alignment with green chemistry initiatives increasingly mandated by global regulatory authorities.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this patented technology in commercial pharmaceutical manufacturing environments Each response is grounded in specific technical details from patent CN106938984B to provide actionable insights for technical evaluation teams evaluating this methodology for their production processes.
Q: How does this method eliminate metal contamination compared to conventional approaches?
A: The patented process uses a metal-free halogenation step that avoids transition metals entirely during the initial reaction phase. This prevents catalyst poisoning in subsequent coupling reactions and eliminates the need for costly metal removal procedures required by traditional methods that use metal catalysts or salts.
Q: What makes this synthesis suitable for commercial scale-up despite complex molecular structures?
A: The one-pot design eliminates intermediate isolation steps while maintaining high yields across diverse substrates (32–87% as demonstrated in patent examples). Reaction conditions use common solvents and catalysts compatible with existing manufacturing infrastructure enabling seamless scale-up from laboratory to commercial production volumes.
Q: How does this method address purity requirements for pharmaceutical applications?
A: By removing metal catalysts from the halogenation step the process prevents transition metal impurities that would require extensive purification. Controlled reaction parameters minimize side products resulting in higher purity intermediates meeting stringent pharmaceutical specifications without additional processing steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivative Supplier
Our patented methodology represents a significant leap forward in the synthesis of complex quinoline derivatives offering pharmaceutical manufacturers an efficient pathway to high-purity intermediates essential for next-generation drug development NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs This technology transfer capability ensures seamless implementation of innovative synthetic routes while meeting rigorous quality demands of global regulatory authorities through robust process validation protocols developed over decades of specialized chemical manufacturing experience.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this breakthrough can optimize your specific manufacturing process Contact us today to receive detailed COA data and route feasibility assessments tailored to your production requirements.
