Revolutionizing Rutaecarpine Production: A Green Electrochemical Approach for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing, driven by the urgent need to reduce environmental footprints and optimize production costs. In this context, the patent CN110453242B, titled "Method for electrochemically synthesizing rutaecarpine," represents a significant technological breakthrough for the production of this potent indolopyridine quinazoline alkaloid. Rutaecarpine, a key bioactive component extracted from Evodia rutaecarpa, has garnered immense attention for its diverse pharmacological properties, including anti-hypertensive, anti-inflammatory, and anti-cancer activities. However, traditional extraction and synthetic methods have long been plagued by low yields, complex multi-step procedures, and the heavy reliance on hazardous reagents. This new electrochemical approach offers a streamlined, green alternative that directly addresses these historical bottlenecks, providing a robust pathway for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rutaecarpine has been a challenging endeavor for process chemists, primarily due to the inefficiency of established routes. As illustrated in the legacy synthetic pathways, traditional methods often rely on isatoic anhydride or anthranilamide as starting materials, necessitating lengthy reaction sequences ranging from four to seven distinct steps. These conventional processes are not only time-consuming but also suffer from disappointingly low overall yields, often reported in literature to be as low as 30% to 34.6%. Furthermore, these routes typically require harsh reaction conditions, including the use of strong acids, strong bases, and stoichiometric amounts of toxic oxidants. The accumulation of byproducts and the difficulty in separating the target molecule from complex reaction mixtures significantly increase the cost of goods sold (COGS) and create substantial waste disposal challenges, making gram-scale production difficult and economically unviable for widespread clinical application.
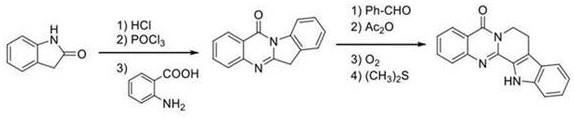
The Novel Approach
In stark contrast to these cumbersome traditional methods, the novel electrochemical cross-dehydrogenation coupling reaction described in the patent offers a remarkably efficient solution. This innovative strategy utilizes electricity as a clean reagent to drive the formation of the critical C=N bond, effectively bypassing the need for external chemical oxidants or expensive transition metal catalysts. By employing a simple setup involving a Reticular Vitreous Carbon (RVC) anode and a platinum cathode, the reaction proceeds under mild conditions—specifically at 70°C in an air atmosphere—to achieve an impressive isolated yield of 80%. This drastic improvement in efficiency not only simplifies the downstream purification process but also aligns perfectly with the principles of green chemistry. For procurement managers and supply chain leaders, this translates to a more reliable source of raw materials with reduced dependency on volatile chemical markets and a significantly lower environmental impact.
Mechanistic Insights into Electrochemical Cross-Dehydrogenation Coupling
The core of this technological advancement lies in the precise control of electron transfer at the electrode surface, which facilitates the oxidative cyclization of the precursor (2-aminophenyl)(1,3,4,9-tetrahydro-2H-pyrido[3,4-b]indol-2-yl)methanone. In this mechanism, the anodic oxidation generates reactive radical intermediates from the amine and the adjacent carbon center without the need for aggressive chemical oxidants that often lead to over-oxidation or side reactions. The use of tetrabutyl ammonium hexafluorophosphate as the supporting electrolyte ensures high conductivity and stability within the acetonitrile solvent system, allowing for a constant current of 10 mA to drive the reaction smoothly. This electrochemical activation is highly selective, promoting the intramolecular coupling required to form the quinazoline ring system of Rutaecarpine while preserving the integrity of other sensitive functional groups within the molecule.

From an impurity control perspective, this mechanism offers distinct advantages over thermal or catalytic methods. Because the reaction potential can be finely tuned by adjusting the current, the formation of unwanted byproducts is minimized, leading to a cleaner crude reaction profile. The absence of metal catalysts eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human use. Consequently, the purification burden is greatly reduced, often requiring only a single flash silica gel column chromatography step to achieve high purity. This mechanistic elegance ensures that the final product meets stringent quality specifications, with characterization data confirming a sharp melting point range of 270.9-273.2°C and consistent NMR spectral data, thereby satisfying the rigorous demands of R&D directors focused on product consistency.
How to Synthesize Rutaecarpine Efficiently
Implementing this electrochemical synthesis route in a laboratory or pilot plant setting requires careful attention to the specific parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the substrate, which can be readily obtained from tryptamine through established literature methods, ensuring a stable supply of starting materials. The electrochemical cell setup is straightforward, utilizing commercially available electrodes and standard power supplies, making it accessible for facilities looking to adopt continuous flow or batch electrochemistry. The following guide summarizes the critical operational steps derived from the patent examples, serving as a foundational protocol for process optimization and scale-up studies.
- Dissolve 0.3 mmol of (2-aminophenyl)(1,3,4,9-tetrahydro-2H-pyrido[3,4-b]indol-2-yl)methanone and 20 mol% tetrabutyl ammonium hexafluorophosphate in 8 mL of acetonitrile.
- Set up the electrolytic cell with a Reticular Vitreous Carbon (RVC) anode and a platinum sheet cathode, then apply a constant current of 10 mA at 70°C under air atmosphere.
- Upon completion monitored by TLC, remove the solvent via rotary evaporation and purify the residue using flash silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the adoption of this electrochemical methodology presents a compelling value proposition centered around cost efficiency and supply security. By fundamentally altering the synthesis landscape, this technology removes several traditional barriers to entry that have historically constrained the availability of Rutaecarpine. The elimination of expensive and hazardous reagents not only lowers the direct material costs but also reduces the overhead associated with safety compliance and waste management. Furthermore, the simplified workflow enhances the agility of the supply chain, allowing manufacturers to respond more quickly to fluctuations in market demand without the lead times associated with complex multi-step syntheses.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the complete removal of stoichiometric oxidants and transition metal catalysts, which are often costly and require specialized disposal protocols. In traditional synthesis, the purification of products from metal residues can account for a substantial portion of the processing budget; by avoiding these metals entirely, the new method drastically simplifies the workup procedure. Additionally, the high yield of 80% means that less raw material is wasted per unit of product produced, directly improving the atom economy and lowering the cost per kilogram. This efficiency gain allows for a more competitive pricing structure, making high-purity Rutaecarpine more accessible for downstream drug development projects.
- Enhanced Supply Chain Reliability: The reliance on electricity as the primary "reagent" decouples production from the volatility of the chemical reagent market. Traditional methods often face disruptions due to the scarcity of specific oxidants or catalysts, but the electrochemical approach relies on stable, commodity-grade materials like acetonitrile and supporting salts. Moreover, the mild reaction conditions (70°C and atmospheric pressure) reduce the risk of thermal runaways or equipment failure, ensuring consistent batch-to-batch production. This reliability is crucial for maintaining uninterrupted supply lines for pharmaceutical clients who require just-in-time delivery of critical intermediates for their own formulation pipelines.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is increasingly feasible with modern flow chemistry technologies, offering a clear path from gram-scale laboratory synthesis to ton-scale commercial production. The process generates minimal hazardous waste, as there are no heavy metal sludge or toxic oxidant byproducts to treat, significantly easing the burden on environmental health and safety (EHS) departments. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the corporate sustainability profile of the supplier, a factor that is becoming increasingly important in vendor selection criteria for major multinational corporations.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled a set of frequently asked questions based on the specific data points and claims within the documentation. These answers address common concerns regarding yield consistency, equipment requirements, and product quality, providing transparency for potential partners evaluating this technology for their supply chains. Understanding these details is essential for making informed decisions about integrating this novel synthesis route into existing manufacturing portfolios.
Q: What are the primary advantages of the electrochemical method over traditional synthesis?
A: The electrochemical method eliminates the need for stoichiometric oxidants and transition metal catalysts, significantly reducing waste and purification costs while achieving an 80% yield compared to traditional yields of 30-35%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of mild conditions (70°C, atmospheric pressure) and inexpensive electrode materials like Reticular Vitreous Carbon makes the process highly scalable and safe for industrial manufacturing.
Q: What is the purity profile of the synthesized Rutaecarpine?
A: The product is obtained as a white solid with high purity after flash column chromatography, characterized by distinct NMR signals and a melting point of 270.9-273.2°C, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rutaecarpine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of complex pharmaceutical intermediates like Rutaecarpine. Our team of expert chemists is well-versed in adapting such cutting-edge patent technologies to meet the rigorous demands of commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Rutaecarpine we deliver meets the highest international standards for safety and efficacy.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this green synthesis technology for their specific needs. Whether you require custom synthesis services or bulk supply of high-purity intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can accelerate your drug development timeline while optimizing your overall production costs.
