Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
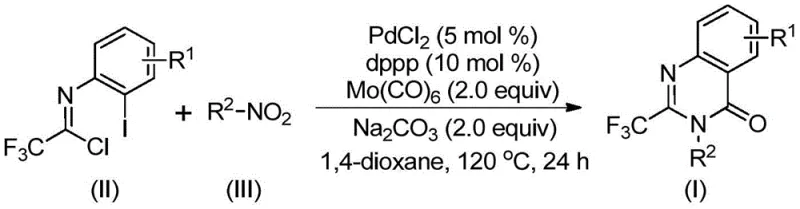
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in patent CN112480015B, which discloses a novel multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. Quinazolinone compounds represent a crucial class of condensed nitrogen-containing six-membered heterocycles, widely recognized for their presence in numerous functional molecules and drugs exhibiting antifungal, antibacterial, antiviral, anti-inflammatory, and anticancer properties. The strategic incorporation of a trifluoromethyl group into these scaffolds is particularly valuable, as fluorine atoms uniquely improve physicochemical properties such as metabolic stability and lipophilicity. This patent presents a transformative approach that leverages transition metal palladium catalysis to streamline the construction of these complex heterocycles from readily available starting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has been fraught with significant technical and economic challenges that hinder large-scale production. Traditional methods often rely on harsh reaction conditions, such as high-pressure carbon monoxide environments, which necessitate specialized and expensive reactor infrastructure, posing substantial safety risks in a manufacturing setting. Furthermore, many existing protocols require the use of expensive ruthenium or platinum catalysts, or substrates that need rigorous pre-activation, such as 2-bromoformylaniline or acid anhydrides. These factors collectively contribute to low overall yields, narrow substrate compatibility, and a complex purification burden. The reliance on pre-functionalized starting materials not only increases the step count but also generates additional waste streams, making these conventional routes less attractive for the cost reduction in pharmaceutical intermediate manufacturing that modern supply chains demand.
The Novel Approach
In stark contrast, the methodology described in CN112480015B offers a streamlined, one-pot solution that bypasses these historical bottlenecks. By utilizing cheap and easily accessible nitro compounds alongside trifluoroethylimidoyl chloride, this process eliminates the need for pre-activated substrates and high-pressure gas cylinders. The reaction employs a robust catalytic system consisting of palladium chloride, a dppp ligand, and molybdenum hexacarbonyl as a solid carbon monoxide source. This innovation allows the reaction to proceed efficiently at 120°C in common organic solvents like dioxane. The result is a highly versatile protocol capable of tolerating a wide range of functional groups, enabling the synthesis of diverse 2-trifluoromethyl-substituted quinazolinone compounds with impressive efficiency. This shift represents a major leap forward for reliable pharmaceutical intermediate supplier networks seeking to optimize their production capabilities.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors aiming to replicate or modify the process for specific analogs. The reaction is believed to initiate with the reduction of the nitro compound to an amine by molybdenum hexacarbonyl, which simultaneously serves as the carbon monoxide source. This generated amine then undergoes a base-promoted intermolecular carbon-nitrogen bond coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the intermediate, forming a divalent palladium species. As the temperature rises, the molybdenum complex releases carbon monoxide, which inserts into the carbon-palladium bond to create an acyl-palladium intermediate. This intricate dance of organometallic steps culminates in the formation of a seven-membered cyclic palladium intermediate, followed by reductive elimination to yield the final 2-trifluoromethyl substituted quinazolinone.
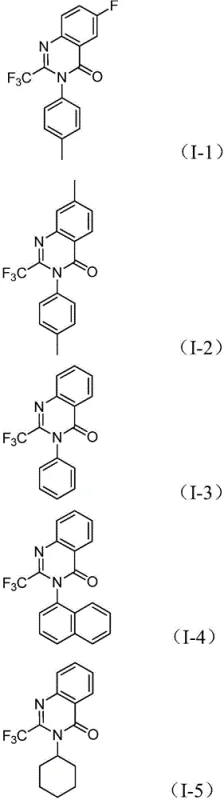
From an impurity control perspective, the one-pot nature of this reaction is advantageous. By avoiding the isolation of unstable intermediates, the process minimizes exposure to air and moisture, which often leads to degradation in multi-step sequences. The use of sodium carbonate as a mild base helps neutralize acidic byproducts without promoting excessive hydrolysis of the sensitive imidoyl chloride or the final lactam ring. The high substrate compatibility demonstrated in the patent, accommodating groups like halogens, alkyls, and trifluoromethyls on both the aromatic ring and the nitrogen substituent, suggests a robust catalytic cycle that resists poisoning. This level of control ensures high-purity pharmaceutical intermediates, reducing the burden on downstream purification processes and ensuring consistent quality for clinical applications.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires careful attention to reagent ratios and thermal management to maximize yield and minimize side reactions. The patent outlines a standardized procedure where palladium chloride, the dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and the specific nitro compound are combined in a Schlenk tube. The choice of solvent is critical, with 1,4-dioxane identified as the optimal medium for dissolving all components and facilitating the reaction kinetics. The mixture is heated to 120°C for a duration ranging from 16 to 30 hours, depending on the specific electronic nature of the substrates. Following the reaction, a simple workup involving filtration and silica gel treatment, followed by column chromatography, affords the pure product. For detailed operational parameters and specific stoichiometric adjustments, refer to the guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to complete.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By shifting from expensive, pre-activated precursors to commodity nitro compounds and stable imidoyl chlorides, manufacturers can significantly reduce raw material costs and mitigate supply chain volatility. The elimination of high-pressure carbon monoxide gas removes a major safety hazard and regulatory hurdle, allowing for production in standard glass-lined reactors rather than specialized autoclaves. This flexibility enhances supply chain reliability and reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of several cost drivers inherent in traditional synthesis. By avoiding the use of noble metals like ruthenium or platinum in favor of a more economical palladium system with a reusable ligand framework, the direct catalyst cost is lowered. Furthermore, the one-pot design consolidates multiple synthetic steps into a single operation, which drastically reduces labor hours, solvent consumption, and energy usage associated with intermediate isolations and drying. This consolidation translates directly into substantial cost savings in API manufacturing, making the final drug substance more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Nitro compounds and trifluoroethylimidoyl chlorides are commodity chemicals produced by numerous vendors globally, reducing the risk of single-source dependency. The robustness of the reaction conditions means that slight variations in raw material quality can be tolerated without catastrophic failure of the batch, ensuring consistent output. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines required by multinational pharmaceutical clients, thereby strengthening the partnership between suppliers and drug developers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the absence of hazardous high-pressure gases and the use of standard heating protocols. The simplified workup procedure, which relies on filtration and standard chromatography rather than complex distillations or extractions, generates less hazardous waste. This aligns with modern green chemistry principles and environmental regulations, reducing the cost and complexity of waste disposal. The ability to scale up complex pharmaceutical intermediates efficiently ensures that the technology remains viable as production volumes increase to meet commercial demand, supporting long-term business growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this technology for their own production lines.
Q: What are the primary advantages of this one-pot synthesis method over traditional routes?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap nitro compounds and achieves high yields (up to 96%) under relatively mild thermal conditions, significantly simplifying the operational complexity and reducing safety risks associated with high-pressure reactors.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates that the method can be expanded to the gram level and is suitable for industrial production. The use of commercially available catalysts and solvents, combined with a straightforward workup procedure involving filtration and chromatography, supports robust commercial scale-up of complex pharmaceutical intermediates.
Q: How does the trifluoromethyl group impact the biological activity of the resulting compounds?
A: The introduction of a trifluoromethyl group significantly enhances the physicochemical properties of the parent molecule. It improves electronegativity, bioavailability, metabolic stability, and lipophilicity, which are critical parameters for developing potent antifungal, antiviral, and anticancer agents within the quinazolinone class.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112480015B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards for pharmaceutical applications. We are committed to delivering excellence in every aspect of the manufacturing process.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable expertise in fine chemical synthesis.
