Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN112694430B, which discloses a novel preparation method for 1,5-dihydro-2H-pyrrole-2-one compounds. This structural backbone is not merely an academic curiosity; it is a critical pharmacophore embedded in numerous bioactive molecules and natural products with profound therapeutic potential. For instance, the antibiotic althiomycin, the hypoglycemic agent glimepiride, and the anticancer candidate isomalyngamide A all share this privileged structural motif, highlighting its immense value in drug discovery and development pipelines.
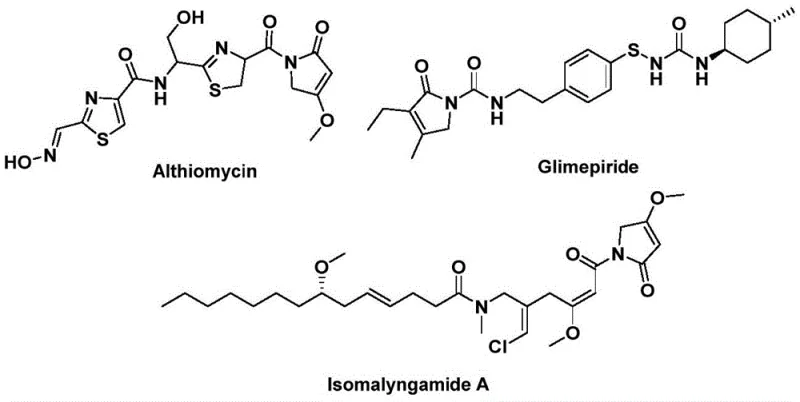
Traditionally, accessing these valuable cores often required multi-step sequences involving harsh conditions or unstable reagents. However, the methodology outlined in the patent leverages a sophisticated palladium-catalyzed bis-carbonylation strategy. By utilizing readily available propargyl amines and benzyl chlorides as starting materials, coupled with a solid carbon monoxide substitute, this process achieves the construction of the five-membered lactam ring in a single operational step. This represents a paradigm shift for reliable pharmaceutical intermediate suppliers seeking to streamline their catalog offerings while maintaining rigorous quality standards. The ability to synthesize these complex architectures directly from simple precursors reduces the cumulative waste and energy consumption typically associated with linear synthetic routes, aligning perfectly with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing 1,5-dihydro-2H-pyrrol-2-one derivatives have frequently been plagued by significant operational hurdles that impede large-scale commercialization. Conventional pathways often rely on the use of gaseous carbon monoxide, which poses severe safety risks due to its high toxicity and flammability, necessitating specialized high-pressure autoclaves and rigorous safety protocols that drive up capital expenditure. Furthermore, traditional cyclization methods may require pre-functionalized substrates that are expensive to procure or synthesize, leading to inflated raw material costs. Many existing protocols also suffer from poor atom economy, generating substantial amounts of stoichiometric waste, and often exhibit limited substrate scope, failing to tolerate sensitive functional groups that are common in advanced drug candidates. These factors collectively result in longer lead times and higher production costs, creating bottlenecks for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the innovative process described in patent CN112694430B overcomes these barriers through an elegant catalytic design. The method employs a palladium catalyst system, specifically palladium acetate paired with a DPPF ligand, to facilitate a dual carbonyl insertion sequence. Crucially, it replaces hazardous gaseous CO with 1,3,5-tricarboxylic acid phenol ester (TFBen), a stable solid that releases CO in situ under thermal conditions. This modification drastically simplifies the reactor setup, allowing the reaction to proceed in standard glassware or stainless steel reactors at atmospheric pressure equivalents. The reaction operates efficiently in acetonitrile at 110°C, demonstrating remarkable tolerance for a wide array of substituents including halogens, alkyl groups, and trifluoromethyl moieties. This robustness ensures that the synthesis of high-purity pharmaceutical intermediates is not only feasible but highly reproducible across different batches, providing a stable foundation for supply chain continuity.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors aiming to optimize the process for specific analogs. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride derivative, forming a reactive benzyl-palladium intermediate. Subsequently, a molecule of carbon monoxide, liberated from the thermal decomposition of TFBen, inserts into the palladium-carbon bond to generate an acyl-palladium species. This electrophilic acyl complex then undergoes nucleophilic attack by the amino group of the propargyl amine substrate. Following this coordination, the alkyne moiety participates in an intramolecular insertion, closing the five-membered ring to form a cyclic palladium intermediate. A second insertion of carbon monoxide occurs, expanding the coordination sphere before the final reductive elimination step releases the desired 1,5-dihydro-2H-pyrrole-2-one product and regenerates the active palladium catalyst.
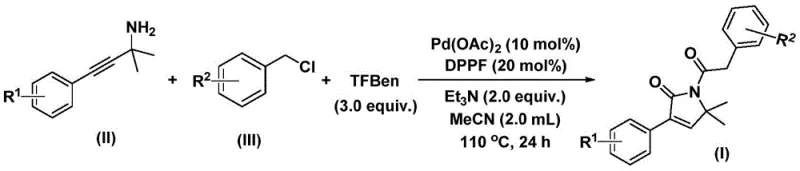
This intricate dance of bond formations is meticulously controlled by the choice of ligand and base. The use of DPPF (1,1'-bis(diphenylphosphino)ferrocene) is critical for stabilizing the palladium center throughout the multiple insertion steps, preventing catalyst deactivation or the formation of palladium black. Furthermore, the presence of triethylamine serves to neutralize the hydrochloric acid byproduct generated during the oxidative addition, driving the equilibrium forward. From an impurity control perspective, this mechanism is highly favorable because the sequential nature of the insertions minimizes side reactions such as homocoupling of the benzyl chloride or polymerization of the alkyne. The result is a clean reaction profile that simplifies downstream purification, a key factor in achieving the stringent purity specifications required for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
Implementing this synthesis in a production environment requires precise adherence to the optimized parameters established in the patent data. The process is designed to be operationally simple, requiring the combination of all reagents in a single vessel followed by heating, which minimizes unit operations and labor costs. The following guide outlines the standardized procedure derived from the exemplary embodiments, ensuring consistent results for technical teams looking to adopt this methodology.
- Combine palladium acetate, DPPF ligand, triethylamine, TFBen (CO source), propargyl amine, and benzyl chloride in acetonitrile solvent within a reaction vessel.
- Heat the reaction mixture to 110°C and maintain stirring for 24 hours to allow for complete bis-carbonylation and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Benzyl chlorides and propargyl amines are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. Moreover, the replacement of gaseous carbon monoxide with a solid surrogate eliminates the logistical complexities and regulatory burdens associated with transporting and storing toxic gases. This shift significantly enhances site safety profiles and reduces insurance and compliance costs, contributing to overall cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The one-pot nature of this reaction eliminates the need for intermediate isolation and purification steps that are typical in multi-step syntheses. By consolidating bond-forming events into a single thermal process, manufacturers can significantly reduce solvent consumption, energy usage for heating and cooling cycles, and labor hours. The high catalytic efficiency means that expensive palladium is used in relatively low loadings (10 mol%), and the high yields reported (up to 92%) ensure that raw material waste is minimized. This efficient atom economy translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions—specifically the tolerance for air-stable reagents and the use of common solvents like acetonitrile—makes the process highly resilient to supply chain disruptions. Unlike methods requiring cryogenic conditions or exotic reagents, this protocol can be executed in standard facilities with minimal retrofitting. The broad substrate scope means that a single manufacturing line can be adapted to produce a library of different analogs simply by swapping the starting benzyl chloride or propargyl amine. This flexibility allows suppliers to respond rapidly to changing customer demands, effectively reducing lead time for high-purity intermediates and ensuring continuous availability for downstream API production.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this exothermic-controlled carbonylation is well-suited for batch reactors. The absence of high-pressure gas feeds simplifies the engineering controls required for scale-up from kilogram to tonne scale. Furthermore, the use of a solid CO source reduces the emission of volatile organic compounds and toxic gases, aligning with increasingly strict environmental regulations. The simplified workup, involving filtration and standard chromatography or crystallization, generates less hazardous waste compared to traditional methods, supporting corporate sustainability goals and reducing waste disposal costs.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common questions based on the patent specifications. These insights address the practical concerns of technical buyers and process engineers evaluating this route for integration into their supply chains.
Q: What is the primary advantage of using TFBen as a carbon monoxide substitute?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a safe and solid surrogate for gaseous carbon monoxide, eliminating the need for high-pressure gas equipment and significantly enhancing operational safety in standard laboratory or pilot plant settings.
Q: Does this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl, without compromising reaction efficiency.
Q: What are the typical yields achieved with this catalytic system?
A: Experimental data indicates high reaction efficiency, with isolated yields ranging from 70% to 92% across various substituted substrates, demonstrating the robustness of the palladium/DPPF catalytic system for scalable production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
The technological advancements detailed in patent CN112694430B represent a significant opportunity for the pharmaceutical industry to access high-value heterocyclic building blocks more efficiently. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such chemical innovations, translating cutting-edge academic and patent literature into robust, commercial-grade manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1,5-dihydro-2H-pyrrole-2-one meets the exacting standards required for drug substance manufacturing.
We invite global partners to leverage our expertise in palladium-catalyzed carbonylations to secure a stable and cost-effective supply of these critical intermediates. Whether you require custom synthesis of novel analogs or bulk production of established compounds, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this advanced synthetic method can optimize your supply chain and accelerate your drug development timelines.
