Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for a diverse array of bioactive natural products and therapeutic agents. As highlighted in recent patent literature, specifically CN112694430B, the demand for efficient access to this heterocyclic system is driven by its presence in potent antibiotics like althiomycin, hypoglycemic agents such as glimepiride, and anticancer candidates like isomalyngamide A. Traditional synthetic approaches to these complex molecules often suffer from multi-step sequences, low atom economy, and the requirement for hazardous gaseous reagents. However, the emergence of transition-metal catalyzed carbonylation has opened new avenues for streamlining these syntheses. This report analyzes a novel palladium-catalyzed bis-carbonylation protocol that utilizes readily available propargylamines and benzyl chlorides to construct the pyrrol-2-one core in a single operational step.
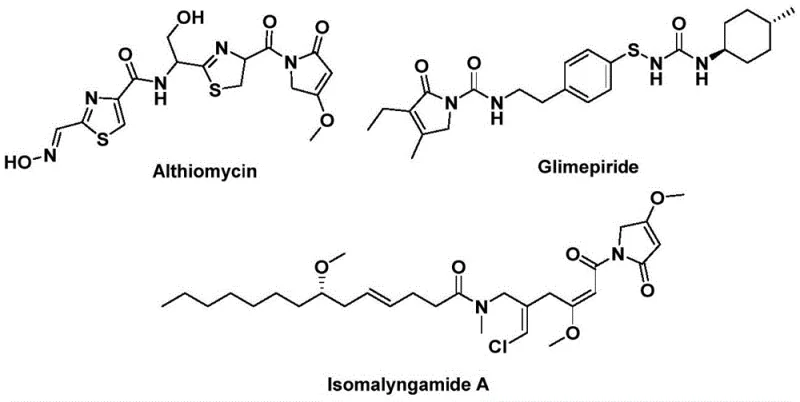
The significance of this technological advancement cannot be overstated for pharmaceutical manufacturers seeking reliable pharmaceutical intermediate suppliers. By shifting from classical condensation reactions to a catalytic carbonylative cyclization, producers can achieve higher purity profiles and significantly reduced waste generation. The methodology described in the patent leverages a solid carbon monoxide surrogate, which not only enhances safety profiles by eliminating the need for high-pressure gas manifolds but also improves the reproducibility of the reaction on a commercial scale. For R&D directors and process chemists, this represents a critical opportunity to re-evaluate legacy routes for pyrrol-2-one containing APIs, potentially unlocking substantial cost reductions and supply chain resilience through a more robust and scalable manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one ring system has relied heavily on the condensation of amines with activated carboxylic acid derivatives or the cyclization of unsaturated precursors under acidic or basic conditions. These conventional pathways frequently encounter significant bottlenecks, including the necessity for pre-functionalized starting materials that are expensive and difficult to source in bulk quantities. Furthermore, traditional methods often require stoichiometric amounts of coupling reagents, leading to poor atom economy and generating substantial quantities of chemical waste that complicate downstream purification and environmental compliance. The reliance on harsh reaction conditions, such as strong acids or elevated temperatures without catalytic control, can also result in the formation of complex impurity profiles, necessitating rigorous and costly chromatographic separations to meet the stringent purity specifications required for active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the innovative method disclosed in patent CN112694430B introduces a streamlined palladium-catalyzed bis-carbonylation strategy that fundamentally alters the synthetic landscape for these intermediates. This approach utilizes simple, commercially available benzyl chlorides and propargylamines as starting materials, reacting them in the presence of a palladium catalyst and a solid carbon monoxide source known as TFBen (1,3,5-tricarboxylic acid phenol ester). The reaction proceeds through a sophisticated cascade involving oxidative addition, dual carbon monoxide insertion, and intramolecular cyclization to forge the five-membered lactam ring efficiently. This one-pot transformation not only simplifies the operational workflow by reducing the number of unit operations but also exhibits exceptional substrate compatibility, tolerating a wide range of electronic and steric environments on the aromatic rings. Consequently, this novel route offers a direct path to high-purity intermediates with minimal byproduct formation, addressing many of the inefficiencies inherent in older synthetic technologies.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The success of this synthesis lies in the intricate catalytic cycle mediated by the palladium complex, which orchestrates the assembly of the molecular framework with high precision. The mechanism initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generating a reactive benzyl-palladium(II) intermediate. Subsequently, carbon monoxide, which is released in situ from the thermal decomposition of the TFBen surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargylamine, followed by a second insertion of carbon monoxide, which is a critical step that distinguishes this bis-carbonylation process from standard carbonylations. The resulting six-membered palladium intermediate then undergoes reductive elimination to release the final 1,5-dihydro-2H-pyrrol-2-one product and regenerate the active catalyst, completing the cycle.
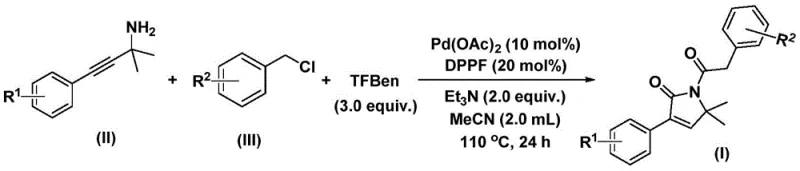
From an impurity control perspective, the use of a well-defined ligand system, specifically 1,1'-bis(diphenylphosphino)ferrocene (DPPF), plays a pivotal role in stabilizing the palladium center and directing the regioselectivity of the cyclization. This ligand choice minimizes side reactions such as homocoupling of the benzyl chloride or polymerization of the alkyne moiety, which are common pitfalls in uncatalyzed or poorly optimized systems. The reaction conditions, typically maintained at 110°C in acetonitrile, provide sufficient energy to drive the dual CO insertion steps while maintaining the stability of the sensitive intermediates. For quality assurance teams, understanding this mechanism is vital, as it explains the high yields observed across various substrates, ranging from electron-rich methoxy-substituted benzyl chlorides to electron-deficient trifluoromethyl analogs, ensuring consistent product quality regardless of the specific derivative being manufactured.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
Implementing this advanced synthetic route requires careful attention to reagent ratios and reaction parameters to maximize efficiency and yield. The process begins by charging a reaction vessel with the palladium catalyst, the DPPF ligand, the solid CO surrogate, a base such as triethylamine, and the specific propargylamine and benzyl chloride substrates in an organic solvent like acetonitrile. The mixture is then heated to facilitate the catalytic cycle, allowing the transformation to proceed to completion over a defined period. While the general procedure is robust, optimization of temperature and reaction time may be necessary for specific sterically hindered substrates to ensure full conversion. For detailed operational protocols, please refer to the standardized synthesis guide below.
- Combine palladium acetate, DPPF ligand, triethylamine, TFBen (CO source), propargylamine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate oxidative addition and carbonyl insertion.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this palladium-catalyzed technology offers compelling strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the raw material supply chain, as the process relies on commodity chemicals like benzyl chlorides and propargylamines which are produced globally in massive volumes, ensuring long-term availability and price stability. Unlike methods requiring specialized gaseous reagents or exotic organometallics, this route utilizes a solid CO source that is easier to handle, store, and transport, thereby reducing logistical complexities and safety overheads associated with hazardous material management. This shift towards safer, more accessible inputs directly translates to enhanced supply chain reliability and reduced risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of multiple synthetic steps and the reduction of waste disposal costs associated with stoichiometric reagents. By consolidating the synthesis into a single catalytic pot, manufacturers can drastically reduce solvent consumption, energy usage, and labor hours required for intermediate isolations. Furthermore, the high atom economy of the carbonylation reaction ensures that a greater proportion of the starting mass is converted into valuable product, minimizing the cost of goods sold per kilogram. Although specific percentage savings depend on the scale, the qualitative reduction in processing time and material intensity provides a clear pathway for margin improvement in competitive API markets.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against variations in substrate electronics means that a single manufacturing platform can be adapted to produce a wide library of derivatives without extensive re-validation. This flexibility allows supply chain planners to respond rapidly to changes in demand for different drug candidates that share the pyrrol-2-one core. Additionally, the use of non-proprietary, off-the-shelf catalysts and ligands reduces dependency on single-source suppliers for critical reagents, mitigating the risk of supply disruptions and fostering a more resilient procurement strategy for long-term commercial production campaigns.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, replacing high-pressure carbon monoxide gas with a solid surrogate significantly lowers the barrier for scaling this reaction to multi-ton quantities. This modification simplifies the engineering requirements for the reactor setup, removing the need for specialized high-pressure autoclaves and complex gas handling infrastructure. The resulting process generates less hazardous waste and operates under milder conditions, aligning with modern green chemistry principles and facilitating easier regulatory approval for commercial manufacturing sites. This ease of scale-up ensures that the transition from laboratory discovery to industrial production is seamless and compliant with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, offering clarity on the practical aspects of adopting this route for pharmaceutical intermediate production. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the key advantages of this carbonylation method over traditional cyclization?
A: This method utilizes a palladium-catalyzed bis-carbonylation strategy that operates in a single pot, eliminating the need for pre-functionalized acid chlorides or harsh cyclization conditions often required in conventional routes.
Q: Is the carbon monoxide source safe for large-scale manufacturing?
A: Yes, the process employs 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid CO surrogate, which releases carbon monoxide in situ, thereby avoiding the safety hazards associated with handling high-pressure CO gas cylinders.
Q: What is the substrate scope for the benzyl chloride component?
A: The reaction demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the successful commercialization of new therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is executed with precision and safety. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-one intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the one described in CN112694430B, we empower our partners to accelerate their drug development timelines while maintaining the highest standards of quality and regulatory compliance.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for an existing program or need to evaluate the feasibility of a new target, our experts are ready to provide specific COA data and route feasibility assessments. Contact us today to explore how our manufacturing expertise can support your supply chain goals and drive value for your organization through superior chemical solutions.
