Advanced N-N Axis Chiral Bis-Indole Phosphine Ligand for High-Purity Pharmaceutical Manufacturing
Advanced N-N Axis Chiral Bis-Indole Phosphine Ligand for High-Purity Pharmaceutical Manufacturing
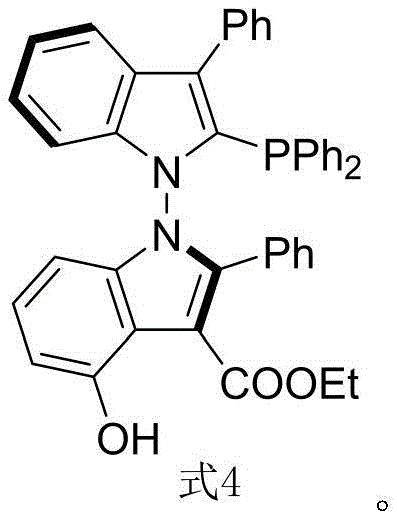
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel ligand architectures that surpass the limitations of traditional scaffolds. Patent CN116041393B discloses a groundbreaking N-N axis chiral bis-indole phosphine ligand, designated as Formula 4, which represents a paradigm shift in stereoselective control for transition metal-catalyzed reactions. This innovation addresses the critical demand for high-purity pharmaceutical intermediates by offering a ligand system that provides broader dihedral angle regulation and enhanced electronic tunability compared to conventional binaphthyl derivatives. For R&D directors and procurement specialists seeking reliable sources for advanced catalytic materials, this technology promises not only superior performance in asymmetric allylation but also a robust pathway for cost-effective manufacturing. The structural uniqueness of this ligand allows for precise modulation of the metal coordination sphere, thereby enabling the synthesis of complex chiral molecules with exceptional enantiomeric excess.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the domain of axial chiral phosphine ligands has been dominated by scaffolds derived from the binaphthyl skeleton, such as BINAP and its variants. While these ligands have served the industry well, they possess inherent structural constraints that limit their efficacy in increasingly complex synthetic challenges. The rigid binaphthyl backbone often restricts the accessible dihedral angles, thereby limiting the steric environment around the catalytic center and potentially compromising enantioselectivity in difficult transformations. Furthermore, the lack of diverse functionalization sites on the binaphthyl core reduces the ability to fine-tune electronic properties or introduce secondary interactions like hydrogen bonding, which are crucial for activating specific substrates. These limitations often necessitate extensive screening of ligand libraries or the use of excessive catalyst loading to achieve acceptable yields, driving up the cost of goods and complicating the supply chain for high-value active pharmaceutical ingredients.
The Novel Approach
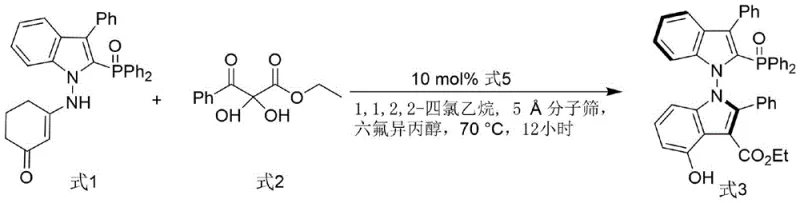
The methodology outlined in the patent introduces a sophisticated synthetic route that constructs an N-N axis chiral bis-indole framework, effectively overcoming the geometric and electronic restrictions of prior art. The process begins with an organocatalytic cyclization between an indole-derived enamine (Formula 1) and a 2,3-diketone ester (Formula 2), facilitated by a chiral phosphoramide catalyst (Formula 5). This step is critical as it establishes the axial chirality with high fidelity under relatively mild thermal conditions. The subsequent transformation involves a deoxygenation step using trichlorosilane and triethylamine to convert the intermediate oxide into the final phosphine ligand. This two-step sequence is remarkably efficient, avoiding the use of precious metals during the ligand synthesis itself, which significantly lowers the barrier to entry for large-scale production. The resulting ligand architecture offers a more open and tunable coordination environment, making it an ideal candidate for demanding asymmetric transformations.
Mechanistic Insights into Organocatalytic Cyclization and Asymmetric Allylation
The core of this technological advancement lies in the intricate mechanism of the organocatalytic cyclization that generates the chiral axis. The reaction employs a chiral phosphoramide catalyst which activates the electrophilic 2,3-diketone ester through hydrogen bonding, while the solvent system, comprising 1,1,2,2-tetrachloroethane and hexafluoroisopropanol (HFIP), plays a pivotal role in stabilizing the transition state. HFIP, known for its strong hydrogen-bond donating ability, enhances the acidity of the catalyst and organizes the solvent cage around the reacting species, thereby enforcing a rigid chiral environment. The presence of 5Å molecular sieves acts as a dehydrating agent, driving the equilibrium towards the formation of the bis-indole derivative (Formula 3) by removing water generated during the condensation. This meticulous control over the reaction microenvironment ensures that the axial chirality is established with high enantiomeric purity, which is subsequently preserved and translated into the final phosphine ligand.
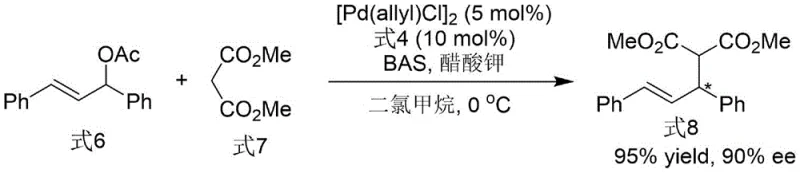
Once synthesized, the ligand demonstrates exceptional utility in palladium-catalyzed asymmetric allylation reactions, a cornerstone transformation in the synthesis of pharmaceutical intermediates. In this application, the ligand coordinates to the palladium center to form a chiral pi-allyl complex that reacts with nucleophiles such as dimethyl malonate. The unique steric bulk and electronic properties of the N-N axis bis-indole scaffold create a highly differentiated chiral pocket, effectively distinguishing between the enantiotopic faces of the allyl intermediate. This results in the formation of the desired chiral product (Formula 8) with outstanding enantiomeric excess, as evidenced by the patent data showing 90% ee in model reactions. The ability to achieve such high stereocontrol with low catalyst loading underscores the ligand's potential for streamlining the production of complex chiral building blocks.
How to Synthesize N-N Axis Chiral Bis-Indole Phosphine Ligand Efficiently
The synthesis of this high-performance ligand is designed for operational simplicity and scalability, utilizing standard laboratory equipment and commercially available reagents. The process is divided into two distinct stages: the construction of the chiral backbone via organocatalysis and the final reduction to the phosphine. The first stage requires careful control of temperature and moisture levels to ensure optimal stereochemical outcomes, while the second stage leverages robust silane chemistry to deliver the final product in high yield. For process chemists looking to implement this route, the detailed standardized synthesis steps provided below outline the precise stoichiometry, solvent choices, and purification protocols necessary to replicate the patent's success. Adhering to these parameters is essential for maintaining the stringent purity specifications required for pharmaceutical applications.
- Perform organocatalytic cyclization of indole-derived enamine and 2,3-diketone ester using chiral phosphoramide catalyst in 1,1,2,2-tetrachloroethane at 70°C.
- Purify the resulting N-N axis chiral bisindole derivative (Formula 3) via silica gel column chromatography and recrystallization to enhance enantiomeric excess.
- Execute deoxygenation of Formula 3 using trichlorosilane and triethylamine in toluene at 120°C to yield the final phosphine ligand (Formula 4).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-N axis chiral bis-indole phosphine ligand offers substantial strategic advantages for procurement managers and supply chain heads focused on cost reduction and reliability. The synthesis route eliminates the need for expensive transition metal catalysts during the ligand manufacturing phase, relying instead on organocatalysis which utilizes more abundant and affordable materials. This shift significantly reduces the raw material costs associated with ligand production and simplifies the waste management profile by avoiding heavy metal contamination. Furthermore, the reaction conditions are mild, operating at moderate temperatures of 70°C and 120°C, which reduces energy consumption and minimizes the risk of thermal runaway incidents, thereby enhancing overall plant safety and operational continuity.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in the ligand synthesis step directly translates to lower production costs and reduced dependency on volatile metal markets. Additionally, the high enantioselectivity achieved in the downstream application means that less catalyst is required to drive the reaction to completion, further optimizing the cost per kilogram of the final API intermediate. The use of standard solvents like toluene and tetrachloroethane, which are readily available in bulk quantities, ensures stable pricing and avoids supply bottlenecks associated with specialized fluorinated solvents often required in other high-end catalytic systems.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including indole derivatives and diketone esters, are commodity chemicals with established global supply chains, ensuring consistent availability and reducing lead times for raw material procurement. The robustness of the synthetic protocol, which tolerates standard workup procedures like silica gel chromatography and recrystallization, facilitates easy technology transfer between manufacturing sites. This flexibility allows companies to diversify their supplier base and mitigate risks associated with single-source dependencies, ensuring a steady flow of critical chiral ligands for continuous pharmaceutical production.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot production without requiring specialized high-pressure or cryogenic equipment. The waste streams generated are primarily organic solvents and salts, which can be managed through standard distillation and treatment facilities, aligning with modern environmental compliance standards. The high atom economy of the cyclization step and the efficient conversion in the reduction step minimize waste generation, supporting sustainability goals and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the operational parameters and performance expectations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this ligand into existing synthetic routes for complex molecule production.
Q: What distinguishes this N-N axis ligand from traditional binaphthyl ligands?
A: Unlike traditional binaphthyl-derived ligands, this N-N axis chiral bis-indole system offers a wider dihedral angle regulation space and additional hydrogen bond activation sites, leading to superior enantioselectivity control in transition metal catalysis.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization requires a chiral phosphoramide catalyst (10 mol%), hexafluoroisopropanol as an additive, and 5Å molecular sieves as a dehydrating agent in 1,1,2,2-tetrachloroethane at 70°C to achieve high stereocontrol.
Q: Can this ligand be scaled for industrial production?
A: Yes, the synthesis utilizes mild reaction conditions, commercially available starting materials, and standard purification techniques like column chromatography, making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bis-Indole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this N-N axis chiral bis-indole phosphine ligand in advancing the synthesis of next-generation pharmaceuticals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand delivered meets the exacting standards required for GMP manufacturing of active pharmaceutical ingredients.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their specific synthetic challenges. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current production volumes and specific route requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and economic efficiency in the competitive global market.
