Advanced N-N Axis Chiral Bis-Indole Phosphine Ligands for Asymmetric Catalysis
Advanced N-N Axis Chiral Bis-Indole Phosphine Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continuously evolving, driven by the demand for ligands that offer superior stereocontrol and operational simplicity. A significant breakthrough in this domain is documented in patent CN116041393B, which introduces a novel class of N-N axis chiral bis-indole phosphine ligands, specifically exemplified by the structure designated as Formula 4. Unlike traditional ligands that rely on carbon-carbon or phosphorus-carbon axial chirality, this innovation leverages the unique rotational barrier and electronic properties of the nitrogen-nitrogen single bond within a bis-indole scaffold. This structural motif not only expands the library of available chiral environments but also provides distinct advantages in terms of tunability and interaction with metal centers. For research and development teams seeking to optimize difficult asymmetric transformations, this ligand represents a powerful new tool capable of delivering exceptional enantiomeric excess.
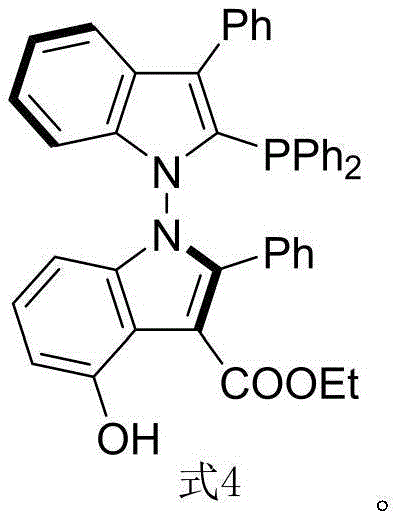
For procurement specialists and supply chain managers, the introduction of such specialized ligands signals a shift towards more efficient and cost-effective manufacturing pathways. The ability to source high-purity chiral ligands that function under mild conditions directly translates to reduced energy consumption and simplified downstream processing in pharmaceutical production. As a reliable pharmaceutical intermediates supplier, understanding the nuances of these next-generation catalysts is crucial for maintaining a competitive edge in the global market. The following analysis delves into the technical superiority of this N-N axis system, contrasting it with conventional methods and highlighting its potential for commercial scale-up in the synthesis of complex organic molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric catalysis has been dominated by axially chiral ligands derived from the binaphthyl skeleton, such as BINAP and BINOL derivatives. While these compounds have served as workhorses for decades, they possess inherent limitations that can hinder optimization in modern synthetic challenges. The rigid biaryl framework of binaphthyl ligands often restricts the conformational flexibility required to accommodate bulky substrates or to fine-tune the steric environment around the metal center. Furthermore, the synthesis of these traditional ligands frequently involves harsh conditions, expensive resolving agents, or multi-step sequences that generate significant waste. From a process chemistry perspective, the lack of additional functional handles, such as hydrogen bond donors, limits the ability of these ligands to engage in secondary interactions that could enhance substrate activation and stereocontrol.
The Novel Approach
The methodology described in patent CN116041393B overcomes these hurdles by introducing an N-N axis chiral bis-indole framework. This novel architecture provides a wider space for dihedral angle regulation compared to binaphthyl-derived phosphine ligands, allowing for a more precise fit within the catalytic pocket. Additionally, the indole nitrogen atoms serve as potential hydrogen bond activation sites, offering an extra dimension of electronic adjustment that is absent in purely hydrocarbon-based axes. The synthesis route is designed for efficiency, utilizing readily available indole derivatives and diketone esters. This approach not only simplifies the construction of the chiral backbone but also ensures that the resulting ligands can be produced with high enantioselectivity, addressing the critical need for purity in fine chemical manufacturing without the prohibitive costs associated with traditional resolution techniques.
Mechanistic Insights into Organocatalytic Coupling and Reduction
The synthesis of the target ligand (Formula 4) proceeds through a sophisticated two-step sequence that exemplifies modern green chemistry principles. The first stage involves the construction of the N-N axis via an organocatalytic coupling reaction between an indole-derived enamine (Formula 1) and a 2,3-diketone ester (Formula 2). This transformation is mediated by a chiral phosphoramide catalyst (Formula 5) in the presence of hexafluoroisopropanol (HFIP) and molecular sieves. The role of HFIP is particularly critical; as a strong hydrogen bond donor, it likely activates the carbonyl electrophile and stabilizes the transition state, thereby facilitating the formation of the sterically congested N-N bond with high fidelity. The reaction is conducted in 1,1,2,2-tetrachloroethane at a moderate temperature of 70°C, yielding the intermediate bis-indole derivative (Formula 3) with excellent stereochemical integrity.
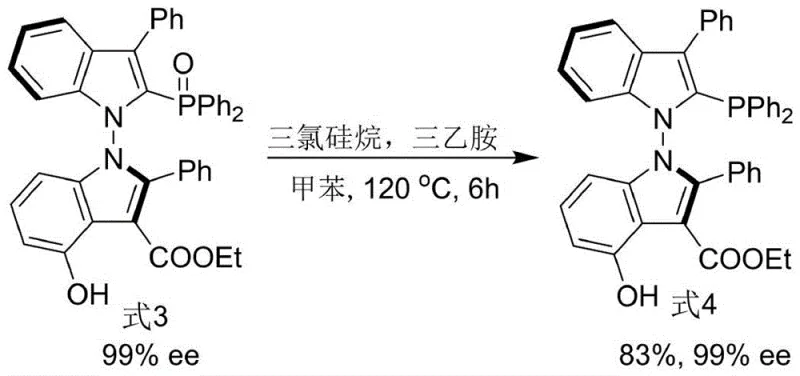
Following the formation of the chiral backbone, the second step involves the deoxygenative reduction of the phosphine oxide moiety to the active phosphine. This is achieved using trichlorosilane and triethylamine in toluene at 120°C. This reduction protocol is robust and avoids the use of hazardous reagents often associated with phosphine oxide reduction, such as chlorosilanes in combination with more dangerous reducing agents. The preservation of the N-N axial chirality during this thermal treatment is a testament to the stability of the indole scaffold. The final product, Formula 4, is obtained in high yield and maintains the high enantiomeric excess established in the first step, demonstrating the robustness of the overall synthetic strategy. This mechanistic pathway ensures that impurities are minimized, a key factor for R&D directors focused on impurity profiles.
Beyond its synthesis, the utility of this ligand is demonstrated in transition metal-catalyzed asymmetric allylation reactions. When employed with a palladium catalyst, the ligand facilitates the nucleophilic attack of malonates on allylic acetates with remarkable stereocontrol. The unique steric bulk and electronic properties of the bis-indole system create a chiral environment that effectively differentiates between the enantiotopic faces of the pi-allyl palladium intermediate. This results in the formation of quaternary carbon centers with high enantiomeric excess, a transformation that is notoriously difficult to achieve with high selectivity. The ability to drive such challenging reactions under mild conditions (0°C to room temperature) underscores the ligand's potential for broad application in the synthesis of complex pharmaceutical intermediates.
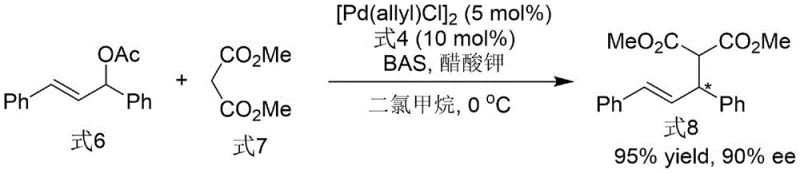
How to Synthesize N-N Axis Chiral Bis-Indole Phosphine Ligand Efficiently
The preparation of this advanced ligand is streamlined for reproducibility and scalability, making it accessible for both laboratory research and pilot plant operations. The process relies on standard unit operations such as stirring, heating, filtration, and column chromatography, which are familiar to any process chemist. The initial coupling step requires careful control of water content, managed effectively by the inclusion of molecular sieves, to drive the equilibrium towards the product. Subsequent purification via silica gel chromatography ensures the removal of any diastereomeric impurities before the final reduction step. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Condense indole-derived enamine with 2,3-diketone ester using chiral phosphoramide catalyst and molecular sieves in 1,1,2,2-tetrachloroethane at 70°C.
- Purify the intermediate N-N axis chiral bisindole derivative via silica gel column chromatography to ensure high enantiomeric excess.
- Reduce the phosphine oxide intermediate using trichlorosilane and triethylamine in toluene at 120°C to yield the final phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-N axis chiral bis-indole phosphine ligand offers substantial strategic benefits for chemical manufacturing enterprises. The synthesis route described in the patent utilizes commodity chemicals and avoids the need for exotic or prohibitively expensive starting materials. This accessibility of raw materials significantly mitigates supply chain risks, ensuring a stable and continuous flow of production inputs. Furthermore, the reaction conditions are relatively mild, operating at temperatures that do not require extreme cooling or high-pressure equipment. This translates to lower capital expenditure for reactor infrastructure and reduced energy costs per kilogram of product, directly impacting the bottom line for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the use of catalytic amounts of chiral inducers drastically simplify the production workflow. By achieving high enantioselectivity directly through organocatalysis, the need for costly chiral separation technologies is removed. Additionally, the reagents used, such as trichlorosilane and triethylamine, are bulk industrial chemicals with well-established supply chains, preventing price volatility. The overall atom economy of the process is favorable, minimizing waste disposal costs and enhancing the sustainability profile of the manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent quality output, which is critical for maintaining long-term contracts with downstream API manufacturers. The tolerance of the reaction to standard laboratory and plant conditions means that technology transfer from R&D to production is seamless. There are no sensitive steps that require inert atmosphere gloveboxes or cryogenic conditions, reducing the likelihood of batch failures due to operational variances. This reliability allows supply chain heads to plan inventory with greater confidence and reducing lead time for high-purity chiral ligands.
- Scalability and Environmental Compliance: The process is inherently scalable, moving from milligram to kilogram scales without fundamental changes to the chemistry. The solvents used, while requiring appropriate handling, are common in the industry and can be recovered and recycled efficiently. The absence of heavy metal catalysts in the ligand synthesis itself (prior to application) reduces the burden of heavy metal clearance in the final product, simplifying regulatory compliance. This aligns with global trends towards greener chemistry, making the ligand an attractive choice for companies aiming to improve their environmental, social, and governance (ESG) metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this ligand into their existing synthetic portfolios.
Q: What distinguishes N-N axis ligands from traditional BINAP derivatives?
A: N-N axis chiral bis-indole ligands offer a wider dihedral angle regulation space and additional hydrogen bond activation sites compared to binaphthyl-derived ligands, leading to potentially superior enantioselectivity in specific transition metal-catalyzed reactions.
Q: What are the critical reaction conditions for the ligand synthesis?
A: The synthesis requires mild conditions, specifically utilizing hexafluoroisopropanol as an additive and 1,1,2,2-tetrachloroethane as a solvent at 70°C for the coupling step, followed by a reduction at 120°C using trichlorosilane.
Q: Can this ligand be used for large-scale pharmaceutical manufacturing?
A: Yes, the patent highlights that the method uses low-cost reagents and mild conditions suitable for industrialized large-scale production, making it viable for commercial API intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bis-Indole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands like the N-N axis bis-indole phosphine in driving innovation in drug discovery and process chemistry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of this specific ligand or adaptation of the technology for your proprietary processes, our CDMO capabilities are ready to support your goals.
We invite you to collaborate with us to explore how this technology can optimize your synthetic routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and enhance your competitive position in the global marketplace.
