Advanced Synthesis Of N-N Axis Chiral Bis-indole Phosphine Ligands For Industrial Catalysis
Advanced Synthesis Of N-N Axis Chiral Bis-indole Phosphine Ligands For Industrial Catalysis
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel ligand architectures that surpass traditional limitations. Patent CN116041393B discloses a groundbreaking N-N axis chiral bis-indole phosphine ligand, designated as Formula 4, which represents a paradigm shift from conventional binaphthyl-based systems. This innovative chemical structure offers unprecedented control over stereoselectivity in transition metal-catalyzed reactions, addressing critical pain points in the synthesis of high-value pharmaceutical intermediates. Unlike existing solutions that often struggle with rigid conformational constraints, this new ligand class provides a versatile platform for tuning dihedral angles and electronic properties. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the technical nuances of this patent is essential for securing a competitive edge in complex molecule manufacturing.
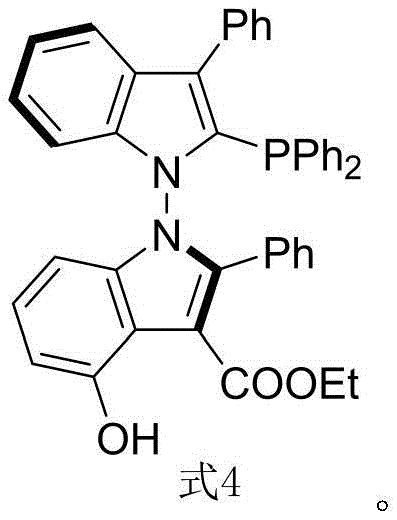
The structural uniqueness of Formula 4 lies in its N-N axial chirality, which fundamentally alters the steric environment around the catalytic center. This architecture allows for a broader range of dihedral angle adjustments compared to the fixed geometry of binaphthyl backbones. Furthermore, the presence of multiple hydrogen bond activation sites within the indole framework enhances substrate binding affinity, leading to improved reaction kinetics and selectivity. The patent highlights that these ligands are not merely theoretical constructs but are synthesized via a robust, scalable pathway that avoids the use of precious metals during the ligand construction phase. This distinction is vital for cost reduction in chiral ligand manufacturing, as it eliminates the need for expensive palladium or rhodium catalysts in the initial steps, relying instead on efficient organocatalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of axially chiral phosphine ligands has been dominated by binaphthyl derivatives, which, while effective, suffer from inherent structural rigidity. These traditional ligands often lack the conformational flexibility required to accommodate bulky or electronically diverse substrates, resulting in suboptimal enantioselectivity for certain transformations. Moreover, the synthesis of conventional ligands frequently involves harsh reaction conditions, including high temperatures and the use of toxic heavy metal catalysts, which complicate purification and increase environmental compliance costs. The reliance on resolution techniques to separate enantiomers further drives up production expenses and reduces overall yield. For supply chain managers, these factors translate into longer lead times and higher volatility in the availability of high-purity OLED material precursors and active pharmaceutical ingredients. The inability to finely tune the steric bulk without synthesizing entirely new scaffolds limits the adaptability of these legacy systems to modern drug discovery pipelines.
The Novel Approach
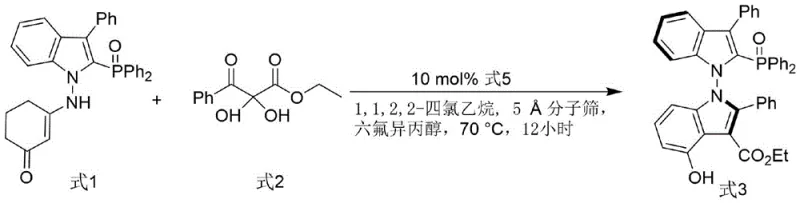
The methodology outlined in the patent introduces a sophisticated yet practical synthetic route that leverages organocatalysis to construct the chiral backbone. The process begins with the coupling of an indole-derived enamine (Formula 1) and a 2,3-diketone ester (Formula 2) under the influence of a chiral phosphoramide catalyst (Formula 5). This reaction proceeds under remarkably mild conditions at 70°C in 1,1,2,2-tetrachloroethane, utilizing 5Å molecular sieves as a dehydrating agent to drive the equilibrium forward. The use of hexafluoroisopropanol as an additive further enhances the reaction efficiency through hydrogen-bonding interactions. This approach not only achieves high enantioselectivity (up to 82% ee in the intermediate step) but also simplifies the workflow by avoiding metal contamination. The subsequent reduction of the phosphine oxide intermediate to the final phosphine ligand (Formula 4) is achieved using trichlorosilane and triethylamine in toluene at 120°C, a standard and scalable protocol.
Mechanistic Insights into Organocatalytic Axial Chirality Construction
The core innovation of this technology lies in the mechanism of axial chirality induction via organocatalysis. The chiral phosphoramide catalyst acts as a Brønsted acid activator, protonating the carbonyl oxygen of the diketone ester to increase its electrophilicity. Simultaneously, the catalyst's chiral pocket directs the approach of the indole enamine nucleophile, ensuring that the bond formation occurs with high facial selectivity. The N-N bond formation is the critical stereodetermining step, where the steric bulk of the catalyst shields one face of the reacting species, forcing the creation of a specific atropisomer. The inclusion of hexafluoroisopropanol is mechanistically significant; it stabilizes the transition state through a network of hydrogen bonds, lowering the activation energy barrier and suppressing non-selective background reactions. This dual-activation mode ensures that the resulting N-N axis chiral bisindole derivative (Formula 3) possesses the correct spatial arrangement necessary for high-performance catalysis.
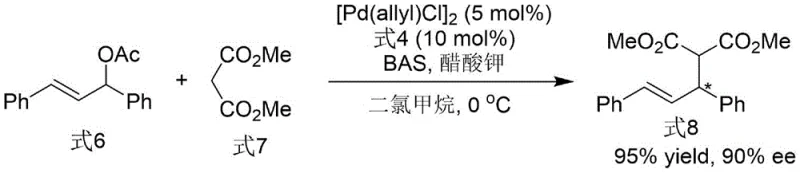
Following the construction of the chiral axis, the conversion of the phosphine oxide to the active phosphine ligand involves a deoxygenation mechanism that preserves the fragile axial chirality. The use of trichlorosilane generates a reactive silyl species that coordinates with the oxygen atom of the P=O bond, facilitating its removal as a siloxane byproduct in the presence of triethylamine. Crucially, this reduction occurs without racemization, maintaining the 99% ee achieved after recrystallization of the intermediate. The final ligand, with its free phosphine group, is then poised to coordinate with transition metals such as palladium. In application, as demonstrated in the asymmetric allylation reaction, the ligand forms a chiral pocket around the metal center that discriminates between the prochiral faces of the allylic substrate. This results in the formation of carbon-carbon bonds with exceptional stereocontrol, yielding products with up to 90% ee, which is critical for the synthesis of bioactive molecules.
How to Synthesize N-N Axis Chiral Bis-indole Phosphine Ligand Efficiently
The synthesis of this advanced ligand is designed for operational simplicity and scalability, making it accessible for both laboratory research and industrial production. The process is divided into two distinct stages: the organocatalytic assembly of the chiral scaffold and the subsequent functional group transformation. The first stage requires precise control of moisture levels, necessitating the use of activated molecular sieves and anhydrous solvents to ensure high yields. The reaction temperature of 70°C is easily maintainable in standard jacketed reactors, and the progress can be monitored conveniently via TLC. Following the isolation of the intermediate, the second stage involves a straightforward reduction protocol using commercially available reagents. The entire workflow minimizes the generation of hazardous waste and avoids the complexities associated with handling air-sensitive metal catalysts during the ligand synthesis itself. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- React indole-derived enamine with 2,3-diketone ester using chiral phosphoramide catalyst and molecular sieves at 70°C.
- Purify the intermediate N-N axis chiral bisindole derivative via silica gel column chromatography.
- Reduce the phosphine oxide intermediate using trichlorosilane and triethylamine in toluene at 120°C to obtain the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-N axis chiral bis-indole phosphine ligand offers substantial strategic benefits for procurement and supply chain operations. The primary advantage stems from the shift towards organocatalysis in the ligand synthesis, which drastically simplifies the supply chain by removing the dependency on scarce and expensive transition metals during the manufacturing phase. This transition not only mitigates the risk of price volatility associated with precious metals but also streamlines the purification process, as there is no need for rigorous heavy metal scavenging steps. Consequently, this leads to significant cost savings in raw material acquisition and waste disposal. Furthermore, the use of common industrial solvents like toluene and tetrachloroethane ensures that the process can be seamlessly integrated into existing manufacturing infrastructure without requiring specialized equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in the ligand synthesis step directly translates to lower variable costs per kilogram of product. By utilizing an organic small-molecule catalyst (Formula 5) that can potentially be recovered or used in low loadings (10 mol%), the process avoids the high capital expenditure associated with palladium or rhodium complexes. Additionally, the high enantioselectivity achieved (99% ee) reduces the need for costly downstream chiral separation processes, thereby improving the overall mass balance and yield of the final active ingredient. This efficiency gain is compounded by the mild reaction conditions, which reduce energy consumption compared to high-temperature or high-pressure alternatives typically found in traditional ligand synthesis.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including indole derivatives and diketone esters, are readily available from global chemical suppliers, reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate standard laboratory and plant environments, ensures consistent batch-to-batch quality, a critical factor for maintaining regulatory compliance in pharmaceutical manufacturing. By diversifying the ligand portfolio with this N-N axis architecture, companies can reduce their reliance on single-source binaphthyl ligands, thereby enhancing supply chain resilience. The scalability of the process, demonstrated by the use of standard solvents and simple workup procedures, facilitates rapid scale-up from gram to ton quantities to meet fluctuating market demands.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing the use of toxic reagents and generating less hazardous waste. The absence of heavy metals in the synthesis phase simplifies effluent treatment and reduces the environmental footprint of the manufacturing facility. The high atom economy of the coupling reaction, driven by the dehydrating action of molecular sieves, ensures that a maximum proportion of reactants is converted into the desired product. This efficiency is crucial for meeting increasingly stringent environmental regulations and sustainability goals. Furthermore, the ability to achieve high purity through simple recrystallization and column chromatography reduces the solvent load associated with extensive purification, contributing to a more sustainable and cost-effective production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of its practical utility. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this ligand into their current synthetic routes. The answers highlight the specific advantages in terms of selectivity, operational ease, and applicability to key industrial transformations.
Q: What are the advantages of N-N axis chiral ligands over traditional binaphthyl ligands?
A: N-N axis chiral bis-indole ligands provide a wider space for dihedral angle regulation and more hydrogen bond activation sites compared to binaphthyl-derived ligands, leading to superior enantioselectivity in asymmetric reactions.
Q: Is the synthesis method suitable for large-scale production?
A: Yes, the method utilizes mild reaction conditions (70°C and 120°C), common solvents like toluene and tetrachloroethane, and avoids expensive transition metals in the ligand synthesis step, making it highly scalable and cost-effective.
Q: What level of enantiomeric excess (ee) can be achieved?
A: The patented process demonstrates excellent stereocontrol, achieving up to 99% ee for the final ligand (Formula 4) after recrystallization, and maintaining high selectivity (90% ee) in downstream asymmetric allylation applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bis-indole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the N-N axis chiral bis-indole phosphine ligand in advancing asymmetric synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the high enantiomeric excess values demanded by modern pharmaceutical standards. We are committed to delivering high-quality chiral ligands that empower your R&D teams to achieve breakthrough results in drug discovery and process development.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and purity requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral chemistry can drive efficiency and innovation in your supply chain.
