Advanced N-N Axis Chiral Bis-Indole Phosphine Ligands for Asymmetric Catalysis and Commercial Scale-Up
Advanced N-N Axis Chiral Bis-Indole Phosphine Ligands for Asymmetric Catalysis and Commercial Scale-Up
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel ligand architectures that surpass the limitations of traditional scaffolds. Patent CN116041393B discloses a groundbreaking class of N-N axis chiral bis-indole phosphine ligands, specifically represented by Formula 4, which exhibit exceptional stereoselective control in transition metal-catalyzed reactions. Unlike conventional ligands that rely on carbon-carbon axial chirality, this innovation leverages the unique steric and electronic properties of the N-N bond connecting two indole units. This structural motif provides a robust platform for fine-tuning catalytic environments, addressing the critical needs of R&D directors seeking higher purity and selectivity in complex molecule synthesis. The ligand's design allows for precise modulation of the metal center's coordination sphere, making it a powerful tool for the construction of chiral pharmaceutical intermediates.
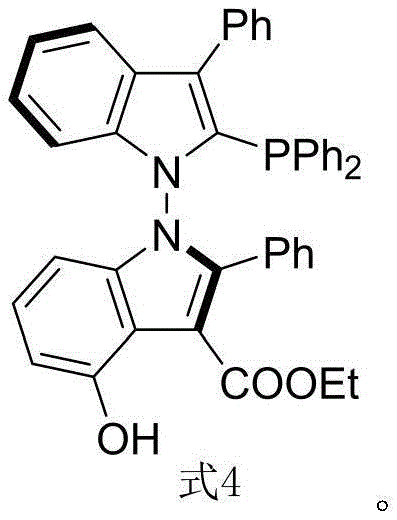
For procurement managers and supply chain heads, the significance of this technology extends beyond mere performance metrics; it represents a pathway to cost reduction in pharmaceutical intermediate manufacturing. The synthesis protocol outlined in the patent utilizes readily available starting materials such as indole derivatives and 2,3-diketone esters, avoiding the supply chain bottlenecks often associated with exotic chiral pool resources. Furthermore, the reaction conditions are remarkably mild and operationally simple, utilizing common solvents and reagents that facilitate commercial scale-up of complex fine chemicals. By adopting this advanced ligand technology, manufacturers can achieve high-purity OLED material precursors or active pharmaceutical ingredients with reduced waste generation and improved process safety, aligning with modern green chemistry principles and regulatory compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric catalysis has been dominated by axially chiral ligands derived from the binaphthyl skeleton, such as BINAP and its derivatives. While these ligands have served the industry well for decades, they possess inherent structural rigidity that limits the tunability of the dihedral angle between the aromatic rings. This rigidity can restrict the conformational flexibility required to accommodate bulky substrates or to induce specific transition states necessary for high enantioselectivity in challenging transformations. Additionally, binaphthyl-based systems often lack sufficient hydrogen bond donor or acceptor sites near the catalytic center, limiting their ability to activate substrates through secondary interactions. These limitations frequently result in suboptimal yields or poor stereocontrol when applied to complex, multifunctional molecules common in modern drug discovery, necessitating the development of more versatile ligand frameworks.
The Novel Approach
The novel approach presented in patent CN116041393B overcomes these hurdles by introducing an N-N axis chiral bis-indole scaffold. This architecture offers a significantly wider space for dihedral angle regulation compared to its binaphthyl counterparts, allowing chemists to fine-tune the steric environment around the metal center with unprecedented precision. The presence of nitrogen atoms within the indole rings introduces additional hydrogen bond activation sites and provides greater electronic adjustment space, enabling the ligand to interact more effectively with a diverse range of substrates. This enhanced flexibility and interaction capability translate directly into superior enantioselective control, as demonstrated by the high ee values achieved in the patented examples. Consequently, this new class of ligands opens up new possibilities for synthesizing chiral compounds that were previously difficult or impossible to access with high optical purity.
Mechanistic Insights into Chiral Phosphoramide Catalyzed Cyclization
The synthesis of the target ligand involves a sophisticated organocatalytic cascade that establishes the crucial N-N axial chirality with high fidelity. The process begins with the reaction between an indole-derived enamine (Formula 1) and a 2,3-diketone ester (Formula 2), catalyzed by a chiral phosphoramide (Formula 5). This step is critical as it sets the stereochemical foundation for the entire molecule. The chiral phosphoramide acts as a Brønsted acid catalyst, activating the electrophilic diketone while simultaneously organizing the nucleophilic enamine through a well-defined hydrogen-bonding network. The use of hexafluoroisopropanol (HFIP) as an additive further enhances this activation by stabilizing charged intermediates and improving the solubility of polar species. Molecular sieves are employed as a dehydrating agent to drive the equilibrium towards the formation of the N-N bonded bis-indole derivative (Formula 3), ensuring high conversion rates.
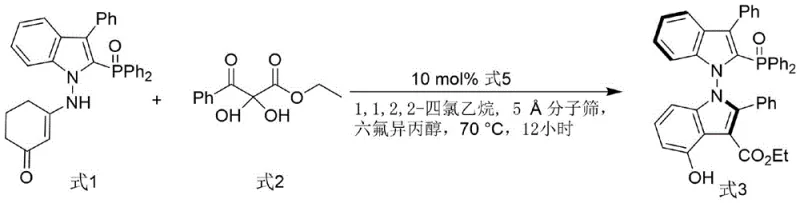
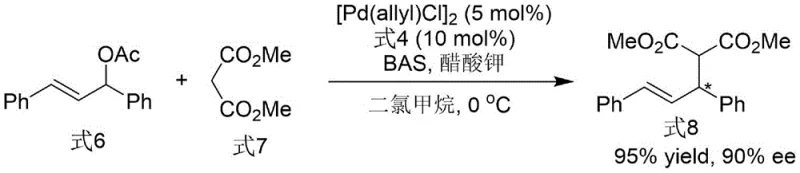
Following the establishment of the chiral axis, the intermediate Formula 3 undergoes a deoxygenation reaction to yield the final phosphine ligand (Formula 4). This transformation is achieved using trichlorosilane and triethylamine in toluene at elevated temperatures. The mechanism likely involves the reduction of the phosphine oxide moiety to the corresponding phosphine, a step that must be carefully controlled to prevent racemization of the sensitive N-N axis. The success of this step is evidenced by the retention of high enantiomeric excess (99% ee) in the final product, demonstrating the robustness of the chiral axis under the reaction conditions. The resulting ligand features a phosphine group positioned strategically to coordinate with transition metals, creating a chiral pocket that dictates the facial selectivity of subsequent catalytic reactions, such as the asymmetric allylation shown in the patent applications.
How to Synthesize N-N Axis Chiral Bis-Indole Phosphine Ligand Efficiently
The preparation of this high-value ligand is designed for reproducibility and scalability, utilizing standard laboratory equipment and commercially sourced reagents. The process is divided into two distinct stages: the construction of the chiral backbone and the final functionalization to the phosphine. The first stage relies on precise temperature control and the exclusion of moisture to ensure the chiral phosphoramide catalyst functions optimally. The second stage requires careful handling of silane reagents but proceeds cleanly to afford the target molecule in high yield. For detailed operational parameters, stoichiometry, and purification protocols, please refer to the standardized guide below which outlines the critical process parameters for GMP-compliant production.
- React indole-derived enamine with 2,3-diketone ester using a chiral phosphoramide catalyst, molecular sieves, and hexafluoroisopropanol in 1,1,2,2-tetrachloroethane at 70°C to form the N-N axis chiral bisindole derivative.
- Purify the intermediate via silica gel column chromatography and recrystallization to achieve high enantiomeric excess.
- Perform deoxygenation of the intermediate using trichlorosilane and triethylamine in toluene at 120°C to yield the final phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-N axis chiral bis-indole phosphine ligand offers substantial strategic benefits for chemical manufacturers aiming to optimize their supply chains and reduce production costs. The synthesis route avoids the use of precious transition metal catalysts during the ligand construction phase, relying instead on organocatalysis which is inherently cheaper and easier to remove from the final product. This elimination of heavy metals simplifies the purification process, reducing the need for expensive scavenging resins or complex extraction procedures, thereby lowering the overall cost of goods sold. Furthermore, the starting materials are commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of supply disruptions and ensuring reliable pharmaceutical intermediate supplier status for partners adopting this technology.
- Cost Reduction in Manufacturing: The process utilizes mild reaction conditions (70°C and 120°C) and common solvents like 1,1,2,2-tetrachloroethane and toluene, which are significantly less expensive than specialized fluorinated solvents often required in high-end asymmetric synthesis. The absence of expensive noble metals in the ligand synthesis step drastically reduces the raw material cost burden. Additionally, the high stereoselectivity achieved minimizes the formation of unwanted diastereomers, reducing the material loss associated with recycling or discarding off-spec batches, leading to substantial cost savings in the long run.
- Enhanced Supply Chain Reliability: The reliance on robust, non-sensitive reagents such as trichlorosilane and triethylamine ensures that the manufacturing process is less susceptible to variations in raw material quality or storage conditions. The synthetic route is linear and concise, involving fewer unit operations compared to multi-step resolutions of racemic mixtures. This simplicity translates to shorter lead times and greater predictability in production scheduling, allowing supply chain managers to respond more agilely to market demands for high-purity fine chemical intermediates without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up in standard stainless steel reactors, avoiding the need for specialized high-pressure or cryogenic equipment. The workup procedures involve standard filtration and extraction techniques that are easily automated and scaled. Moreover, the atom economy of the organocatalytic step is favorable, and the use of molecular sieves allows for the efficient management of water byproducts. These factors contribute to a reduced environmental footprint, facilitating easier regulatory approval and alignment with sustainability goals, which is increasingly critical for maintaining a license to operate in the global chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these aspects is crucial for evaluating the feasibility of integrating this ligand into existing manufacturing workflows and for assessing its potential impact on product quality and process efficiency.
Q: What are the advantages of N-N axis chiral ligands over traditional binaphthyl ligands?
A: N-N axis chiral bis-indole ligands offer a wider space for dihedral angle regulation, more hydrogen bond activation sites, and greater electronic tunability compared to binaphthyl-derived ligands, leading to superior enantioselectivity.
Q: What are the key reaction conditions for synthesizing this ligand?
A: The synthesis involves mild conditions: an initial organocatalytic step at 70°C in 1,1,2,2-tetrachloroethane with hexafluoroisopropanol, followed by a deoxygenation step at 120°C in toluene using trichlorosilane.
Q: Can this ligand be used for large-scale pharmaceutical production?
A: Yes, the method utilizes commercially available raw materials, avoids expensive transition metals in the ligand synthesis itself, and employs standard purification techniques like column chromatography, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-N Axis Chiral Bis-Indole Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands like the N-N axis bis-indole phosphine in driving innovation in pharmaceutical and fine chemical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or intermediate delivered meets the highest international standards for enantiomeric excess and chemical purity required by top-tier global clients.
We invite you to collaborate with our technical team to explore how this cutting-edge ligand technology can optimize your specific synthetic routes. By leveraging our expertise in process optimization and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and pilot trial proposals, ensuring that your supply chain is fortified with the most advanced and reliable chemical solutions available in the market.
