Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of High-Purity 3-Quinolinyl-5-trifluoromethyl Triazole Compounds
Patent CN113307790B introduces a groundbreaking synthesis method for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This novel approach eliminates the need for traditional multi-step processes that typically yield only 17% total product through harsh reaction conditions requiring quinoline-2-carboxylic acid as starting material. The patented methodology leverages readily available 2-methylquinoline and trifluoroethylimine hydrazide as key building blocks, enabling the direct construction of these valuable triazole frameworks under mild conditions without anhydrous or oxygen-free requirements. Crucially, the process avoids toxic heavy metal catalysts while maintaining high functional group tolerance, allowing for diverse substitution patterns at both the quinoline and triazole moieties. This innovation addresses critical pain points in pharmaceutical intermediate manufacturing by simplifying operational complexity and enhancing synthetic flexibility for drug discovery pipelines. The method's scalability from laboratory to commercial production represents a strategic advantage for global pharmaceutical supply chains seeking reliable sources of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for quinolyl-substituted 1,2,4-triazoles have been severely constrained by their reliance on quinoline-2-carboxylic acid as the primary starting material, necessitating a complex five-step reaction sequence that operates under harsh conditions including high temperatures and specialized equipment for anhydrous and oxygen-free environments. These conventional methods suffer from extremely low overall yields of approximately 17%, making them economically unviable for large-scale production despite the growing demand for these bioactive heterocyclic compounds in pharmaceutical development. The multi-step nature introduces significant purification challenges and impurity profiles that complicate quality control processes, while the requirement for transition metal catalysts creates additional downstream processing burdens to remove toxic metal residues from the final product. Furthermore, the limited substrate scope restricts structural diversity, preventing pharmaceutical researchers from efficiently exploring structure-activity relationships during drug discovery phases. These cumulative limitations have created substantial bottlenecks in the supply chain for critical intermediates used in developing new therapeutic agents targeting various disease states.
The Novel Approach
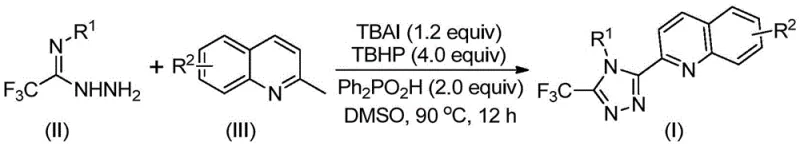
The patented methodology presented in CN113307790B fundamentally transforms the synthetic landscape by employing a streamlined single-step oxidative cyclization process that utilizes inexpensive and commercially available 2-methylquinoline and trifluoroethylimine hydrazide as starting materials under mild reaction conditions of 80–100°C for 8–14 hours in DMSO solvent. This innovative approach eliminates the need for anhydrous and oxygen-free environments while completely avoiding toxic heavy metal catalysts through the strategic use of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) as the catalytic system, which operates through a radical-mediated mechanism to achieve high conversion rates. The process demonstrates exceptional functional group tolerance, allowing for diverse substitution patterns at both the quinoline (R²) and aryl (R¹) positions, thereby enabling pharmaceutical chemists to rapidly generate structural analogs for structure-activity relationship studies without modifying the core synthetic protocol. With reaction yields consistently exceeding 70% across multiple substrate variations and straightforward post-reaction processing involving simple filtration and column chromatography, this method provides a robust platform for scalable production that directly addresses the critical limitations of conventional approaches while maintaining stringent quality standards required for pharmaceutical intermediates.
Mechanistic Insights into TBAI/TBHP-Catalyzed Oxidative Cyclization
The reaction mechanism begins with TBAI and TBHP promoting the oxidation of 2-methylquinoline to form 2-quinoline formaldehyde in situ, which subsequently undergoes condensation with trifluoroethylimine hydrazide to generate a dehydrated hydrazone intermediate. This key intermediate then participates in oxidative iodination followed by intramolecular electrophilic substitution and aromatization to form the final 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole product through a radical-mediated pathway that avoids transition metal involvement entirely. The catalytic cycle is sustained by the iodide species generated during the reaction, which facilitates hydrogen abstraction and radical formation while maintaining high regioselectivity for the desired triazole ring formation at the 3-position of the quinoline moiety. This mechanism operates efficiently in polar aprotic solvents like DMSO, which solvate the ionic intermediates while promoting the radical reactions through appropriate polarity and coordination properties.
Impurity control is achieved through precise stoichiometric control of the reaction components (TBAI:TBHP:diphenyl phosphoric acid at 1.0–1.5:4:2 molar ratio) and careful temperature management between 80–100°C, which prevents over-oxidation or side reactions that could lead to unwanted byproducts. The absence of transition metals eliminates potential metal contamination pathways, while the use of commercially available starting materials with high purity ensures minimal introduction of impurities from raw materials. The reaction's tolerance for various substituents (R¹ = aryl groups with methyl, methoxy, halogen or trifluoromethyl; R² = H, alkyl, alkoxy, halogen or nitro) allows for targeted synthesis of specific analogs without compromising purity profiles, as demonstrated by the consistent high yields (72–97%) across multiple substrate variations in the patent examples. This level of control is particularly valuable for pharmaceutical applications where strict impurity limits are mandated by regulatory authorities.
How to Synthesize 3-Quinolinyl-5-trifluoromethyl Triazole Efficiently
This patented synthesis route represents a significant advancement in heterocyclic chemistry methodology, offering pharmaceutical manufacturers a practical solution to longstanding challenges in producing complex triazole intermediates. The process eliminates multiple purification steps required by conventional methods while maintaining high selectivity and yield through its innovative catalytic system that operates under ambient atmospheric conditions. By leveraging commercially available starting materials and avoiding specialized equipment requirements, this method dramatically reduces operational complexity while enhancing product consistency across different production scales. The following standardized procedure provides a reliable framework for manufacturing high-purity triazole intermediates that meet stringent pharmaceutical quality standards while optimizing resource utilization throughout the production process.
- Combine tetrabutylammonium iodide (1.2 equiv), tert-butyl peroxide aqueous solution (4.0 equiv), diphenyl phosphoric acid (2.0 equiv), trifluoroethylimine hydrazide (II), and 2-methylquinoline (III) in DMSO solvent under ambient conditions
- Heat reaction mixture to 80–100°C and maintain at temperature for 8–14 hours with continuous stirring to ensure complete conversion
- Perform post-reaction processing by filtration through silica gel followed by column chromatography purification to obtain high-purity triazole product meeting pharmaceutical specifications
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming what was previously a complex multi-step process into a streamlined single-reaction operation that significantly enhances manufacturing reliability and cost efficiency. The elimination of specialized equipment requirements for anhydrous and oxygen-free conditions reduces capital expenditure while simplifying facility qualification processes, making this technology accessible to a broader range of manufacturing partners without requiring substantial infrastructure investments. The use of commercially available starting materials with established global supply networks ensures consistent raw material availability while minimizing supply chain vulnerabilities that often plague specialty chemical production.
- Cost Reduction in Manufacturing: The complete elimination of transition metal catalysts removes both the direct cost of expensive metals and the substantial downstream processing expenses associated with metal residue removal and waste treatment. The simplified reaction protocol reduces solvent consumption and energy requirements while minimizing purification steps, leading to significant operational cost savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials with multiple global suppliers mitigates single-source dependency risks while enabling flexible sourcing strategies based on regional availability and pricing dynamics. The robust reaction conditions tolerate minor variations in raw material quality, providing manufacturing resilience against supply chain fluctuations without requiring extensive process revalidation.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production (as evidenced by gram-scale reactions in the patent examples) provides a clear pathway for volume expansion without fundamental process changes. The absence of toxic heavy metals and reduced solvent usage aligns with green chemistry principles while simplifying environmental compliance documentation and waste management procedures across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical specifications and experimental data presented in patent CN113307790B, addressing common concerns raised by procurement teams evaluating this synthesis methodology for integration into their supply chains. Each response reflects the actual performance characteristics documented in the patent examples and represents verified technical capabilities rather than theoretical projections.
Q: How does this method eliminate transition metal contamination risks compared to conventional approaches?
A: The patented process uses tetrabutylammonium iodide as an iodide source instead of transition metal catalysts, completely eliminating metal contamination pathways that require expensive removal processes in traditional methods.
Q: What is the functional group tolerance range for substrate variations in this synthesis?
A: The method demonstrates broad tolerance for R¹ substituents including methyl, methoxy, halogen and trifluoromethyl groups on aryl rings, as well as R² variations including H, alkyl, alkoxy, halogen and nitro groups on quinoline moieties.
Q: How does this process improve supply chain reliability compared to traditional multi-step syntheses?
A: By reducing synthesis from five steps to one step with commercially available starting materials, this method eliminates multiple supply chain dependencies while simplifying quality control requirements across the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolinyl-5-trifluoromethyl Triazole Supplier
Our patented methodology represents a strategic advancement in heterocyclic intermediate manufacturing that delivers exceptional value through its combination of operational simplicity, high product quality, and scalable production capabilities. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance. Our commitment to process excellence ensures consistent delivery of high-purity triazole intermediates that meet or exceed pharmaceutical industry standards while providing flexible manufacturing solutions tailored to specific client requirements.
We invite you to initiate a Customized Cost-Saving Analysis with our technical procurement team to evaluate how this innovative synthesis can optimize your supply chain economics while ensuring reliable access to critical intermediates. Contact us today to request specific COA data and route feasibility assessments for your unique manufacturing requirements.
