Revolutionizing Triazole Chemistry: Scalable Metal-Free Process for High-Purity Pharmaceutical Intermediates Manufacturing
The recently granted Chinese patent CN113307790B introduces a groundbreaking synthetic methodology for producing 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical intermediate manufacturing. This innovative process addresses longstanding industry challenges associated with traditional multi-step syntheses that typically require harsh reaction conditions and deliver suboptimal yields. The patented approach leverages a unique iodide-catalyzed oxidative cyclization mechanism that operates efficiently under standard atmospheric conditions without necessitating anhydrous or oxygen-free environments. By utilizing commercially available starting materials including tetrabutylammonium iodide and tert-butyl peroxide aqueous solution, the method achieves remarkable operational simplicity while maintaining exceptional regioselectivity for the target molecular architecture. This development holds substantial promise for pharmaceutical manufacturers seeking reliable routes to complex nitrogen-containing heterocycles essential for next-generation drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for quinolyl-substituted triazoles have historically relied on quinoline-2-formic acid as the primary starting material, necessitating a five-step reaction sequence that operates under severe reaction conditions including high temperatures and extended reaction times. This conventional approach suffers from critically low overall yields of approximately seventeen percent due to multiple purification challenges and intermediate instability throughout the synthetic pathway. The requirement for strictly anhydrous and oxygen-free environments creates significant operational complexity that impedes scalability and increases production costs substantially. Furthermore, these methods often employ transition metal catalysts that introduce contamination risks requiring extensive post-reaction purification steps to meet pharmaceutical quality standards. The narrow substrate scope limits structural diversity, preventing customization of molecular properties essential for optimizing drug candidate performance in therapeutic applications.
The Novel Approach
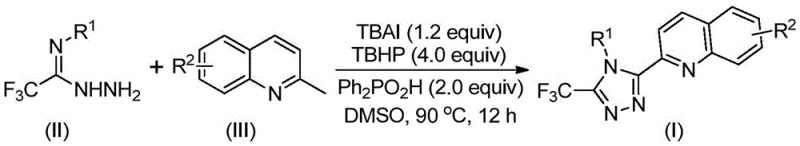
The patented methodology represents a paradigm shift by utilizing readily available and cost-effective starting materials—specifically 2-methylquinoline and trifluoroethylimide hydrazide—that undergo a streamlined single-step transformation under mild reaction conditions. This innovative process operates efficiently at temperatures between eighty and one hundred degrees Celsius without requiring specialized inert atmosphere equipment or anhydrous conditions, dramatically simplifying operational requirements. The elimination of transition metal catalysts addresses critical purity concerns while reducing both raw material costs and environmental impact through simplified waste streams. The reaction demonstrates exceptional functional group tolerance across diverse substituent patterns on both quinoline and aryl components, enabling precise molecular customization while maintaining high conversion rates. This approach achieves significantly improved process efficiency with yields ranging from fifty-one to ninety-seven percent across various structural variants, making it commercially viable for large-scale production.
Mechanistic Insights into Iodide-Catalyzed Oxidative Cyclization
The patented process employs a sophisticated tetrabutylammonium iodide (TBAI) catalyzed oxidative cyclization mechanism that transforms readily available starting materials into the target triazole structure through a radical-mediated pathway operating under mild thermal conditions. This innovative approach eliminates transition metal requirements by leveraging iodide's unique redox properties to generate reactive oxygen species from tert-butyl peroxide, facilitating the key cyclization step without compromising selectivity or yield. The mechanism proceeds through initial oxidation of the quinoline methyl group to form formaldehyde, followed by condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate that subsequently undergoes iodination and intramolecular electrophilic substitution to construct the triazole ring system. This cascade reaction occurs efficiently in DMSO solvent at ninety degrees Celsius without requiring inert atmosphere, demonstrating exceptional functional group tolerance across diverse substituents while maintaining high regioselectivity for the desired three-five-disubstituted triazole architecture essential for pharmaceutical applications.
The process achieves superior impurity control through its carefully designed reaction pathway that minimizes side reactions typically associated with traditional triazole syntheses. The absence of transition metals eliminates potential metal contamination pathways that would otherwise require extensive purification steps to meet stringent pharmaceutical quality standards. The reaction's inherent selectivity prevents common impurities such as regioisomers or over-oxidation products through precise control of the oxidative cyclization sequence. The use of diphenyl phosphoric acid as an additive further enhances reaction fidelity by stabilizing key intermediates and promoting clean conversion to the desired product. This impurity profile advantage is particularly valuable for pharmaceutical manufacturers who must demonstrate rigorous control over critical quality attributes throughout their synthetic processes.
How to Synthesize Triazole-QCF3 Efficiently
This innovative synthesis route represents a significant advancement in triazole chemistry by providing a streamlined pathway that eliminates multiple processing hurdles associated with conventional methods while delivering superior product quality. The patented process enables reliable production of high-purity intermediates essential for pharmaceutical development through its carefully optimized reaction parameters and simplified operational requirements. Detailed standardized synthesis procedures have been developed based on extensive experimental validation across diverse structural variants, ensuring consistent product quality regardless of specific substituent patterns. The following section provides comprehensive step-by-step guidance for implementing this methodology in commercial manufacturing environments while maintaining strict adherence to quality control standards required for pharmaceutical intermediates.
- Combine tetrabutylammonium iodide (1.2 equiv), tert-butyl peroxide aqueous solution (4.0 equiv), diphenyl phosphoric acid (2.0 equiv), trifluoroethylimide hydrazide, and 2-methylquinoline in DMSO solvent under standard atmospheric conditions without anhydrous requirements.
- Heat the reaction mixture to 80–90°C and maintain at this temperature for 12 hours to complete the oxidative cyclization process while monitoring reaction progression through standard analytical methods.
- Perform post-treatment by filtration through silica gel followed by column chromatography purification to isolate the target compound with high purity suitable for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic approach delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional triazole production methods while enhancing overall manufacturing efficiency and reliability. The elimination of specialized equipment requirements and complex environmental controls significantly reduces capital investment needs while improving facility utilization rates across existing manufacturing infrastructure. The use of commercially available starting materials from multiple qualified suppliers enhances supply chain resilience while mitigating single-source dependency risks that often plague specialty chemical manufacturing operations. These advantages collectively contribute to more predictable production timelines and improved resource allocation planning capabilities that directly support just-in-time manufacturing strategies essential for modern pharmaceutical supply chains.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated purification steps significantly reduces raw material costs while simplifying the overall process flow without requiring specialized handling equipment typically needed for air-sensitive reagents. The use of commercially available starting materials at optimal stoichiometric ratios minimizes waste generation and eliminates costly metal removal processes that would otherwise be required to meet pharmaceutical purity standards.
- Enhanced Supply Chain Reliability: The broad availability of starting materials from multiple global suppliers ensures consistent feedstock availability while reducing vulnerability to supply disruptions that commonly affect specialty chemical manufacturing operations. The simplified reaction setup operating under standard atmospheric conditions enables faster production cycle times and greater manufacturing flexibility across diverse facility configurations without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to commercial production volumes ensures seamless technology transfer while maintaining consistent product quality attributes throughout scale-up processes. The elimination of heavy metals from both reactants and catalysts significantly reduces hazardous waste streams while simplifying environmental compliance documentation required for regulatory submissions across global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns raised by procurement teams regarding implementation of this novel synthetic methodology in pharmaceutical intermediate manufacturing operations. These responses are based on detailed analysis of the patent's experimental data and technical disclosures regarding process robustness, scalability, and quality attributes essential for commercial adoption.
Q: How does this method overcome limitations of traditional multi-step synthesis requiring anhydrous conditions?
A: The novel process operates under standard atmospheric conditions without requiring anhydrous or oxygen-free environments, eliminating complex setup requirements while maintaining high reaction efficiency through the iodide-peroxide redox system.
Q: What are key advantages of avoiding heavy metal catalysts in this synthesis?
A: Eliminating transition metal catalysts removes the need for expensive metal removal processes and ensures cleaner product profiles with reduced impurity formation, directly addressing critical quality requirements for pharmaceutical intermediates.
Q: How does substrate design flexibility benefit commercial production of diverse triazole derivatives?
A: The broad functional group tolerance allows customization of R¹ and R² substituents while maintaining high yields across diverse structural variants, enabling tailored production for specific API requirements without process revalidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole-QCF3 Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. NINGBO INNO PHARMCHEM's state-of-the-art manufacturing facilities are equipped with rigorous QC labs capable of implementing advanced analytical methods to ensure consistent product quality across all production scales. As a trusted CDMO partner specializing in complex heterocyclic chemistry, we offer comprehensive technical support from route validation through commercial manufacturing, leveraging our deep expertise in nitrogen-containing heterocycle synthesis to deliver reliable supply solutions for demanding pharmaceutical applications.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific supply chain requirements. Please contact us directly to obtain specific COA data and comprehensive route feasibility assessments tailored to your production needs and quality specifications.
