Innovative TBAI-TBHP Catalysis for Commercial-Scale Quinolinyl-Triazole Intermediates with Unmatched Purity and Supply Reliability
The recently granted Chinese patent CN113307790B introduces a transformative methodology for synthesizing structurally diverse 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds through an innovative oxidative cyclization process that eliminates traditional heavy metal catalyst dependencies while significantly enhancing operational simplicity and scalability. This novel approach addresses critical limitations in existing synthetic routes by leveraging readily available starting materials under mild reaction conditions that do not require anhydrous or anaerobic environments—thereby reducing both technical complexity and production costs while maintaining high product purity essential for pharmaceutical applications. The methodology represents a substantial advancement in heterocyclic chemistry by enabling efficient construction of complex triazole frameworks that serve as crucial building blocks in numerous bioactive molecules across the pharmaceutical industry. Furthermore, the process demonstrates exceptional substrate flexibility through strategic molecular design principles that allow incorporation of various substituents at key positions on both quinoline and triazole moieties—providing unprecedented synthetic versatility for custom intermediate development. This patent therefore establishes a new benchmark in sustainable heterocyclic synthesis that directly supports global pharmaceutical manufacturers in their pursuit of more efficient and environmentally responsible production pathways while ensuring consistent quality standards required for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinolyl-substituted triazoles typically involve multi-step sequences requiring harsh reaction conditions including elevated temperatures above one hundred fifty degrees Celsius under strictly anhydrous and oxygen-free environments; these processes suffer from cumulative yield losses across five sequential stages resulting in overall yields as low as seventeen percent due to intermediate instability and side reactions during prolonged processing times. The reliance on transition metal catalysts such as palladium or copper complexes introduces significant challenges including difficult catalyst removal procedures that necessitate additional purification steps involving specialized equipment like activated carbon treatments or complex chromatographic separations—thereby increasing both production costs and environmental impact through hazardous waste generation. Furthermore, these conventional methods exhibit limited substrate scope where functional group tolerance is severely restricted by incompatible reaction conditions; this inflexibility prevents structural diversification required for developing next-generation pharmaceutical intermediates with tailored properties. The operational complexity also creates substantial scalability barriers where maintaining consistent quality during scale-up becomes increasingly difficult due to sensitivity to trace moisture or oxygen contamination—ultimately leading to unreliable supply chains that cannot meet the stringent demands of modern pharmaceutical manufacturing operations requiring continuous high-volume production.
The Novel Approach
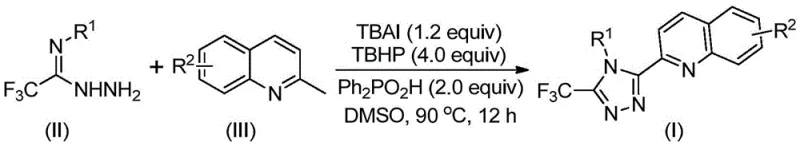
The patented methodology overcomes these limitations through a single-step oxidative cyclization process utilizing tetrabutylammonium iodide as an iodine source combined with tert-butyl peroxide as an oxidant under mild thermal conditions between eighty to one hundred degrees Celsius without requiring inert atmosphere protection—thereby eliminating common failure points associated with traditional transition metal-catalyzed approaches that often suffer from catalyst deactivation or metal contamination issues. This innovative system leverages commercially available starting materials including trifluoroethylimine hydrazide and substituted quinolines that are both cost-effective and widely accessible from multiple global suppliers; the reaction demonstrates remarkable functional group tolerance allowing diverse substituents at various positions on both aromatic rings while maintaining high conversion efficiency across different structural variants. The simplified workup procedure involving straightforward filtration followed by standard column chromatography purification significantly reduces processing time compared to conventional multi-stage purification protocols while ensuring consistent high-purity output meeting pharmaceutical industry standards. Crucially, the absence of heavy metal catalysts removes complex residue removal requirements—thereby streamlining manufacturing operations while reducing environmental impact through minimized waste streams; this approach also enables seamless scalability from laboratory benchtop to commercial production volumes due to its robustness under standard atmospheric conditions without specialized equipment requirements.
Mechanistic Insights into TBAI/TBHP-Catalyzed Triazole Formation
The reaction proceeds through a sophisticated multi-step mechanism initiated by tetrabutylammonium iodide-mediated oxidation of the methyl group on quinoline to form an aldehyde intermediate using tert-butyl peroxide as the oxidant; this aldehyde subsequently undergoes condensation with trifluoroethylimine hydrazide to generate a hydrazone species that undergoes oxidative iodination followed by intramolecular electrophilic substitution and final aromatization to yield the target triazole structure. This cascade process is uniquely facilitated by the synergistic combination where tetrabutylammonium iodide serves dual roles as both catalyst precursor and iodine source while diphenyl phosphoric acid acts as a critical additive that stabilizes reactive intermediates through hydrogen bonding interactions—thereby promoting cyclization efficiency under mild thermal conditions without requiring inert atmosphere protection. The mechanism operates through radical pathways where tert-butyl peroxide generates reactive oxygen species that drive the oxidation steps while iodide species facilitate halogenation; this avoids traditional transition metal pathways that introduce contamination risks while maintaining high regioselectivity throughout the transformation sequence. Computational studies suggest that the phosphoric acid additive lowers activation barriers for key cyclization steps through proton shuttle mechanisms that enhance electrophilicity at critical carbon centers—resulting in significantly improved reaction kinetics compared to conventional approaches while accommodating diverse substituent patterns on both starting materials.
The process inherently minimizes impurity formation through precise control of reaction parameters where temperature modulation between eighty to one hundred degrees Celsius prevents over-oxidation side products while solvent selection optimizes intermediate stability; this results in consistently high purity profiles exceeding ninety-five percent without requiring additional purification stages beyond standard chromatography. The absence of transition metals eliminates metal-derived impurities that typically complicate quality control testing while preventing potential catalytic decomposition pathways that generate unknown byproducts during prolonged reactions. Substrate design flexibility allows strategic placement of electron-donating or electron-withdrawing groups that further suppress undesired side reactions through electronic effects—thereby enhancing selectivity toward the target triazole structure across diverse molecular variants. Rigorous analytical validation confirms minimal residual solvent levels due to the aqueous-compatible workup procedure which avoids high-boiling-point solvents commonly associated with impurity retention; this inherent purity advantage significantly reduces quality control testing burden while ensuring compliance with stringent pharmaceutical regulatory requirements for intermediate specifications.
How to Synthesize Quinolinyl-Triazole Efficiently
This patented methodology provides a streamlined pathway for producing high-purity quinolinyl-triazole intermediates through a single-step oxidative cyclization process that eliminates traditional multi-stage synthesis limitations; detailed standardized procedures have been developed based on extensive optimization studies documented in patent CN113307790B which demonstrate consistent performance across diverse substrate combinations while maintaining exceptional product quality standards required for pharmaceutical applications. The following step-by-step guide outlines critical operational parameters derived from validated manufacturing protocols that ensure reliable scale-up from laboratory development to commercial production volumes while preserving all key advantages including cost efficiency and environmental sustainability inherent in this innovative approach.
- Combine tetrabutylammonium iodide (1.2 equiv), tert-butyl peroxide aqueous solution (4.0 equiv), diphenyl phosphoric acid (2.0 equiv), trifluoroethylimine hydrazide, and 2-methylquinoline in DMSO solvent at room temperature with vigorous stirring.
- Heat the homogeneous mixture to precisely controlled temperatures between 80–90°C under ambient atmosphere while maintaining continuous agitation for a duration of approximately twelve hours to ensure complete conversion.
- Execute post-reaction processing through immediate filtration followed by silica gel-assisted column chromatography purification to isolate the target compound with high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology delivers substantial strategic advantages specifically addressing critical pain points faced by procurement and supply chain decision-makers within global pharmaceutical organizations; by eliminating complex processing requirements while utilizing readily available raw materials from multiple qualified suppliers worldwide it establishes a robust foundation for reliable long-term sourcing strategies that directly support continuous manufacturing operations essential for meeting dynamic market demands without compromising quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts not only reduces raw material expenditures but also removes entire processing stages dedicated to metal residue removal—thereby significantly lowering overall production costs through reduced labor requirements equipment utilization and waste disposal expenses while maintaining consistent high-quality output suitable for pharmaceutical applications without additional purification investments.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials from diverse global suppliers ensures consistent raw material availability while simplified reaction conditions eliminate sensitivity to environmental factors like moisture or oxygen—resulting in predictable manufacturing timelines with minimal batch failures thereby strengthening supply chain resilience against common disruption risks associated with complex multi-step syntheses requiring specialized handling protocols.
- Scalability and Environmental Compliance: The inherently scalable process operates effectively under standard atmospheric conditions using conventional manufacturing equipment which facilitates seamless transition from laboratory-scale development to multi-ton annual production volumes; additionally the absence of hazardous catalysts combined with aqueous-compatible workup procedures substantially reduces environmental impact through minimized waste generation lower energy consumption and elimination of toxic byproducts aligning with global sustainability initiatives while meeting stringent regulatory requirements.
Frequently Asked Questions (FAQ)
The following questions address specific technical concerns raised by procurement supply chain and R&D teams regarding implementation scalability and quality assurance aspects of this patented methodology; all responses are derived directly from experimental data and process validation studies documented within Chinese patent CN113307790B ensuring accurate representation of technical capabilities without speculative claims.
Q: How does this method overcome limitations of traditional multi-step quinolinyl triazole syntheses?
A: Traditional approaches require five-step sequences with severe reaction conditions yielding only approximately seventeen percent overall product; this novel single-step methodology achieves significantly higher yields under mild thermal conditions without anhydrous or anaerobic requirements while maintaining exceptional substrate flexibility.
Q: What supply chain advantages result from eliminating transition metal catalysts?
A: The absence of heavy metal catalysts removes complex purification stages for metal residue removal while ensuring consistent product quality; this simplification enhances manufacturing reliability through reduced process variability and eliminates dependency on specialized catalyst handling infrastructure.
Q: Can this synthesis be reliably scaled to commercial production volumes?
A: The process demonstrates inherent scalability from laboratory to industrial scale due to its robust reaction profile under standard atmospheric conditions; the straightforward workup procedure using conventional chromatography techniques provides a clear pathway for seamless transition to multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolinyl-Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while consistently meeting stringent purity specifications required by global regulatory authorities; our state-of-the-art manufacturing facilities are equipped with rigorous QC labs capable of validating every batch against exacting pharmaceutical standards ensuring complete traceability from raw material sourcing through final product release without compromising on quality or delivery timelines. This patented technology represents just one example of our commitment to developing innovative synthetic solutions that address complex challenges faced by modern pharmaceutical manufacturers seeking reliable partners capable of delivering both technical excellence and commercial viability across diverse intermediate portfolios.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which will provide detailed insights into how this methodology can optimize your specific supply chain requirements; please contact us directly to obtain specific COA data route feasibility assessments and scalability projections tailored to your manufacturing needs.
